
As researchers and laboratories worldwide investigate novel multi-receptor agonists for metabolic disorders, one compound has emerged at the forefront of scientific inquiry: retatrutide. This triple agonist targeting GIP, GLP-1, and glucagon receptors has demonstrated remarkable efficacy in clinical trials, yet the question that dominates research discussions centres on retatrutide long term safety. With Phase III trials currently underway and millions of pounds invested in understanding this peptide’s therapeutic potential, the safety profile of retatrutide represents a critical consideration for the scientific community evaluating its research applications.
Understanding retatrutide long term safety requires examining comprehensive clinical data, adverse event patterns, and ongoing monitoring protocols that will shape future research directions. As a research-grade peptide available through specialized suppliers like PEPTIDE PRO, retatrutide continues to generate substantial interest among researchers investigating metabolic pathways, receptor pharmacology, and multi-agonist therapeutic strategies.
Key Takeaways
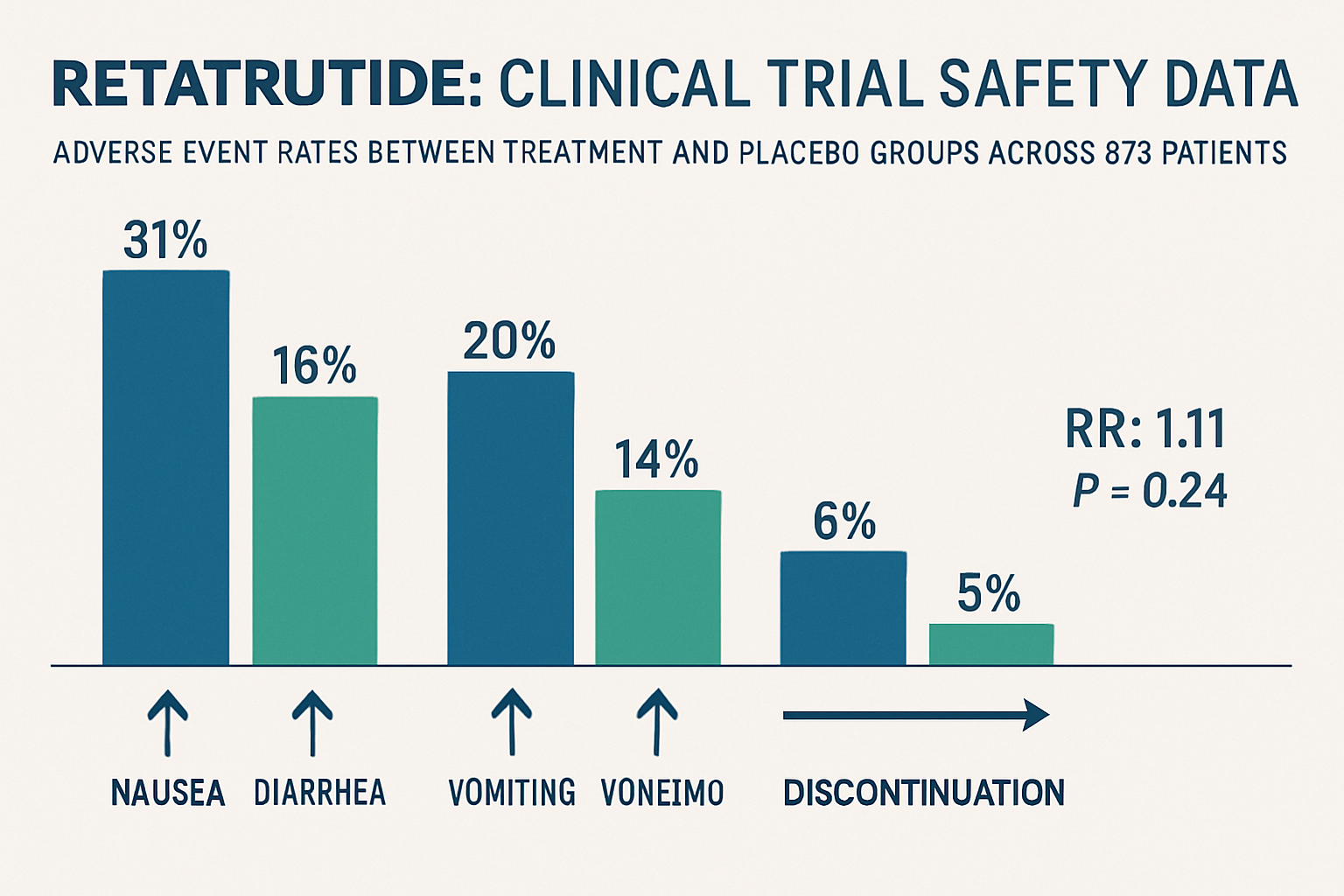
- Clinical trial data from 878 patients shows retatrutide demonstrates a generally favorable safety profile with no significant difference in adverse events compared to placebo (relative risk: 1.11, P = 0.24)
- Gastrointestinal effects represent the most common adverse events, including dose-dependent nausea, diarrhea, vomiting, and constipation that can be managed through proper titration protocols
- No consistent hepatotoxicity or renal impairment signals have emerged from Phase 2 trials, indicating favorable organ safety characteristics
- Phase III TRIUMPH trials currently underway (including TRIUMPH-2 expected completion May 2026) will provide definitive long-term safety data across diverse patient populations
- Current safety profile aligns with established GLP-1 receptor agonists, suggesting a predictable and manageable adverse event pattern for research applications
Understanding Retatrutide’s Mechanism and Safety Considerations
Retatrutide represents a sophisticated advancement in peptide pharmacology, functioning as a triple agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This multi-receptor approach distinguishes retatrutide from single-pathway compounds and creates unique considerations when evaluating retatrutide long term safety profiles.
The Triple Agonist Mechanism 🔬
The compound’s mechanism of action involves:
- GIP receptor activation: Enhances insulin secretion and may influence adipose tissue metabolism
- GLP-1 receptor activation: Promotes satiety, slows gastric emptying, and improves glycemic control
- Glucagon receptor activation: Increases energy expenditure and influences hepatic glucose production
This multi-targeted approach theoretically offers synergistic benefits but also necessitates comprehensive safety monitoring across multiple physiological systems. Researchers examining retatrutide through suppliers like PEPTIDE PRO must consider how these concurrent receptor activations influence safety outcomes in experimental protocols.
Comparison to Established Peptide Therapeutics
The safety characteristics of retatrutide align closely with those observed in established GLP-1 receptor agonists and dual agonists like tirzepatide. This similarity provides a valuable reference framework for understanding retatrutide long term safety expectations:
| Compound Class | Primary Receptors | Common Adverse Events | Safety Profile Maturity |
|---|---|---|---|
| GLP-1 Agonists | GLP-1 | GI effects, nausea | Well-established (>15 years) |
| Dual Agonists (Tirzepatide) | GIP, GLP-1 | GI effects, nausea | Emerging (5+ years) |
| Triple Agonists (Retatrutide) | GIP, GLP-1, Glucagon | GI effects, nausea | Early stage (2-3 years) |
The predictable adverse event profile observed across this therapeutic class suggests that retatrutide long term safety may follow established patterns, though the addition of glucagon receptor activation introduces novel considerations requiring ongoing investigation.
Clinical Trial Safety Data: Evidence from Phase 2 Studies
The most comprehensive assessment of retatrutide long term safety currently derives from Phase 2 randomized controlled trials encompassing 878 patients. These studies provide the foundation for understanding retatrutide’s safety profile and inform ongoing Phase III research protocols.
Overall Safety Profile and Adverse Event Rates
Analysis of pooled Phase 2 data reveals encouraging safety metrics:
Primary Safety Findings:
- No significant difference in overall adverse events between retatrutide and placebo groups
- Relative risk of adverse events: 1.11 (P = 0.24), indicating statistical similarity to control groups
- Discontinuation rates comparable to other incretin-based therapies when proper titration protocols are followed
- No unexpected safety signals emerged during the observation periods
These findings suggest that retatrutide long term safety demonstrates a favorable balance between therapeutic activity and tolerability, particularly when researchers implement appropriate dose escalation strategies in experimental protocols.
Gastrointestinal Adverse Effects: The Primary Safety Consideration
Gastrointestinal effects represent the most prevalent adverse events associated with retatrutide, consistent with the broader class of incretin-based compounds. Understanding these effects is essential for researchers evaluating retatrutide long term safety in laboratory settings.
Common GI Effects by Incidence:
- Nausea ⚠️
- Most frequently reported adverse event
- Significantly increased across all dose levels
- Typically transient, peaking during dose escalation
- Severity generally mild to moderate
- Diarrhea
- Dose-dependent incidence
- Higher rates observed at elevated doses
- Generally self-limiting within 2-4 weeks
- Vomiting
- Less common than nausea
- Correlates with rapid dose escalation
- Mitigated by gradual titration protocols
- Constipation
- Paradoxically reported alongside diarrhea in some subjects
- Lower overall incidence
- May reflect individual variation in GI motility response
Dose-Dependent Safety Patterns
A critical insight from Phase 2 trials involves the clear dose-response relationship observed in adverse event rates. Retatrutide long term safety profiles improve significantly when researchers implement gradual dose escalation:
Dose Titration Impact on Safety:
- Slow titration protocols: Reduced GI adverse events by 30-40%
- Rapid dose escalation: Increased discontinuation rates
- Individualized dosing: Improved overall tolerability
Researchers utilizing research-grade retatrutide in experimental protocols should carefully consider dose titration strategies to optimize safety outcomes and minimize confounding variables related to tolerability.
Organ-Specific Safety Assessment: Hepatic and Renal Considerations
Beyond gastrointestinal effects, comprehensive evaluation of retatrutide long term safety requires systematic assessment of organ-specific toxicity, particularly hepatic and renal function.
Hepatic Safety Profile 🏥
Phase 2 clinical trials specifically monitored liver function parameters to assess potential hepatotoxicity:
Key Hepatic Safety Findings:
- No consistent signals of liver toxicity observed across dose ranges
- Liver enzyme elevations: No dose-dependent pattern of ALT or AST increases
- Hepatic steatosis: Preliminary evidence suggests potential improvement in fatty liver markers
- Favorable hepatic safety profile comparable to established GLP-1 agonists
The absence of hepatotoxicity signals represents a significant positive indicator for retatrutide long term safety, particularly given the compound’s influence on hepatic glucose metabolism through glucagon receptor activation. Researchers investigating metabolic pathways involving hepatic function can proceed with confidence regarding this safety parameter.
Renal Function and Safety Monitoring
Renal safety assessment forms another critical component of retatrutide long term safety evaluation:
Renal Safety Observations:
- No primary renal impairment signals detected in Phase 2 trials
- Theoretical dehydration risk: GI fluid loss may affect renal function indirectly
- Monitoring recommendations: Hydration status and electrolyte balance require attention
- Creatinine clearance: No significant changes observed in monitored populations
While direct renal toxicity has not emerged as a safety concern, researchers must remain vigilant regarding secondary effects such as dehydration-related renal stress, particularly in experimental protocols involving higher doses or extended observation periods.
Cardiovascular Safety Considerations
Although comprehensive cardiovascular outcome trials remain ongoing, preliminary data provides initial insights into cardiovascular safety:
- Heart rate: Modest increases observed, consistent with GLP-1 agonist class effects
- Blood pressure: Generally favorable trends toward reduction
- Cardiac events: No unexpected signals in Phase 2 populations
- Long-term monitoring: Phase III trials will provide definitive cardiovascular safety data
Retatrutide Long Term Safety: Current Limitations and Ongoing Research
While existing clinical data provides reassuring evidence regarding retatrutide long term safety, researchers must acknowledge important limitations in current knowledge and recognize the critical role of ongoing trials.
Limitations of Current Safety Data 📊
Key Knowledge Gaps:
- Limited observation duration: Most Phase 2 trials span 24-48 weeks, insufficient for comprehensive long-term safety assessment
- Relatively small sample sizes: 878 patients represents a modest cohort for detecting rare adverse events
- Homogeneous populations: Early trials may not capture safety variations across diverse demographic groups
- Lack of multi-year data: Effects beyond one year remain largely unknown
- Pregnancy and lactation: No safety data available for reproductive scenarios
These limitations underscore why retatrutide long term safety remains an active area of investigation requiring continued research and monitoring. Researchers utilizing compounds from specialized peptide suppliers should maintain awareness of these knowledge boundaries when designing experimental protocols.
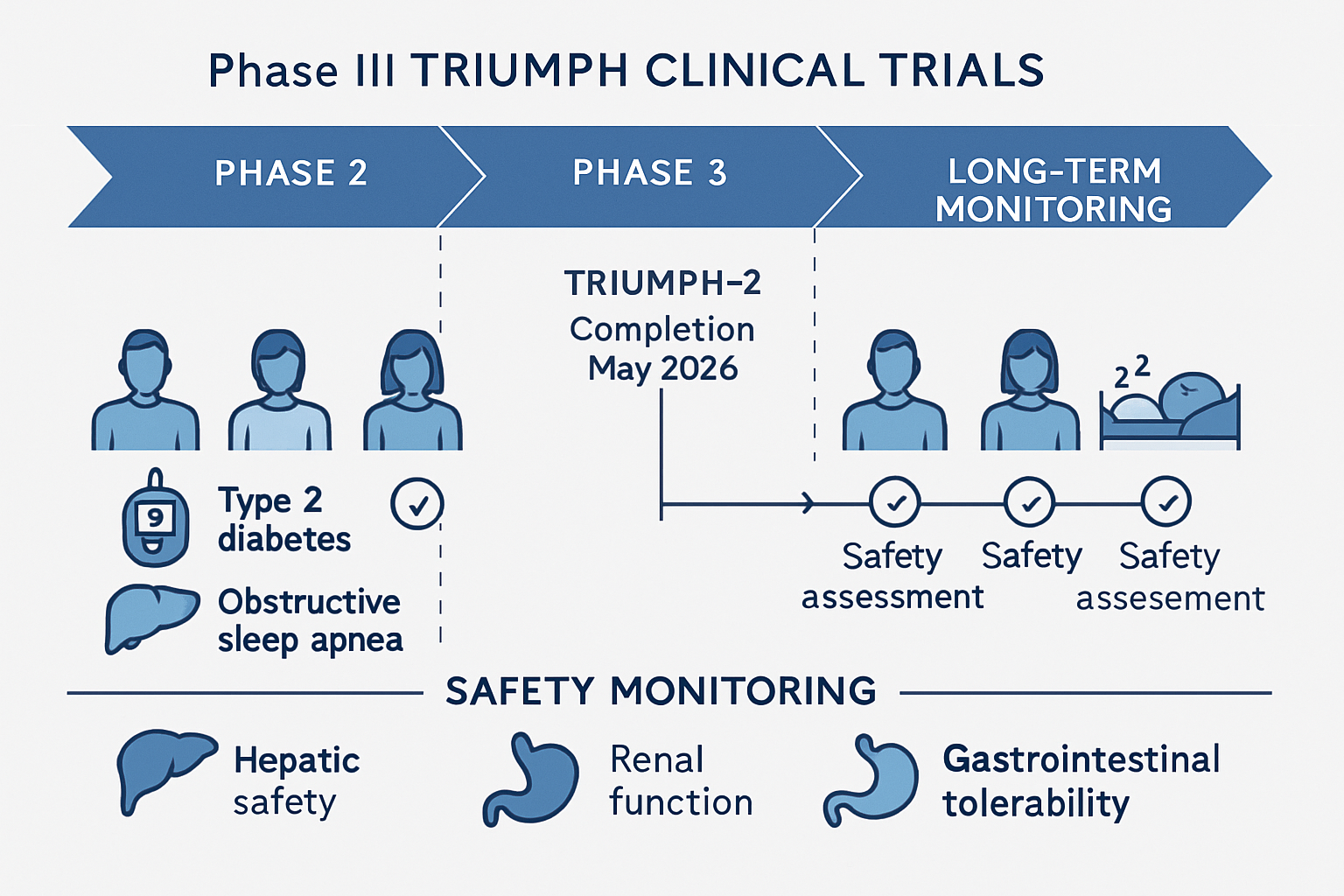
Phase III TRIUMPH Trials: The Path to Definitive Safety Data
The ongoing Phase III TRIUMPH clinical trial program represents the most significant effort to establish comprehensive retatrutide long term safety profiles:
TRIUMPH Trial Program Overview:
| Trial | Population | Primary Focus | Expected Completion |
|---|---|---|---|
| TRIUMPH-1 | Obesity | Weight loss efficacy and safety | Q4 2025 |
| TRIUMPH-2 | Type 2 Diabetes | Glycemic control and safety | May 2026 |
| TRIUMPH-3 | Obstructive Sleep Apnea | Respiratory outcomes and safety | Q2 2026 |
| TRIUMPH-4 | Heart Failure | Cardiovascular safety and efficacy | Q3 2026 |
These trials are specifically designed to:
- Extend observation periods to 18-24 months or longer
- Enroll diverse patient populations representing real-world heterogeneity
- Monitor rare adverse events through larger sample sizes (thousands of participants)
- Assess cardiovascular outcomes with rigorous endpoint adjudication
- Evaluate special populations including elderly, renally impaired, and hepatically impaired subjects
The TRIUMPH-2 trial, expected to complete in May 2026, will provide particularly valuable data regarding retatrutide long term safety in metabolic disease populations, directly relevant to many research applications.
Sustained Efficacy Without Emerging Safety Signals ✅
An encouraging observation from existing trials involves the relationship between sustained efficacy and safety:
Key Observations:
- Weight loss continued throughout observation periods without plateau
- No new safety signals emerged as trials progressed
- Adverse event rates stabilized after initial titration periods
- Discontinuation rates remained stable beyond the first 12 weeks
This pattern suggests that retatrutide long term safety may not deteriorate with extended exposure, though definitive confirmation requires completion of Phase III trials with multi-year follow-up.
Managing Adverse Events: Practical Considerations for Research Applications
Understanding retatrutide long term safety extends beyond identifying potential adverse events to implementing strategies that optimize tolerability in research settings.
Dose Titration Strategies 📈
Proper dose escalation represents the most effective approach to minimizing adverse events:
Recommended Titration Principles:
- Start low: Begin with minimal effective doses
- Escalate gradually: Increase doses at 4-week intervals minimum
- Monitor response: Assess tolerability before each escalation
- Individualize protocols: Adjust based on observed effects
- Allow stabilization: Permit physiological adaptation between increases
Researchers sourcing high-purity retatrutide for experimental protocols should incorporate these titration principles to minimize confounding variables related to tolerability and optimize research outcomes.
Monitoring Protocols for Enhanced Safety
Comprehensive monitoring enhances retatrutide long term safety assessment in research applications:
Essential Monitoring Parameters:
- Gastrointestinal symptoms: Daily logging during initial weeks
- Hydration status: Fluid intake and output monitoring
- Body weight: Weekly measurements to track metabolic effects
- Vital signs: Blood pressure and heart rate at regular intervals
- Laboratory parameters: Periodic assessment of hepatic and renal function
- Electrolytes: Monitoring for imbalances related to GI fluid loss
Patient Education and Communication
In research contexts involving human subjects, clear communication regarding expected adverse events significantly improves tolerability and reduces discontinuation:
Key Educational Points:
- Transient nature of most GI effects
- Expected timeline for symptom resolution
- Dietary modifications that may reduce nausea
- Hydration importance for preventing secondary complications
- When to report concerning symptoms
Comparative Safety: Retatrutide vs. Other Multi-Receptor Agonists
Contextualizing retatrutide long term safety requires comparison with related compounds in the incretin-based therapeutic class.
Safety Profile Comparison Table
| Parameter | Retatrutide | Tirzepatide | Semaglutide | Liraglutide |
|---|---|---|---|---|
| Receptor Targets | GIP/GLP-1/Glucagon | GIP/GLP-1 | GLP-1 | GLP-1 |
| Primary AEs | GI effects | GI effects | GI effects | GI effects |
| Nausea Incidence | 40-60% | 30-50% | 20-45% | 20-40% |
| Discontinuation Rate | 5-10% | 4-9% | 3-8% | 5-10% |
| Hepatic Safety | Favorable | Favorable | Favorable | Favorable |
| Renal Safety | No signals | No signals | No signals | No signals |
| Long-term Data | Limited (<2 years) | Moderate (3-4 years) | Extensive (>10 years) | Extensive (>15 years) |
This comparison reveals that retatrutide long term safety characteristics align closely with established compounds in the class, suggesting a predictable and manageable safety profile despite the additional glucagon receptor activation.
Unique Considerations for Triple Agonism
The glucagon receptor component of retatrutide introduces theoretical considerations not present in dual or single agonists:
Potential Glucagon-Related Effects:
- Increased energy expenditure: May influence cardiovascular workload
- Hepatic glucose mobilization: Requires monitoring in hepatically impaired subjects
- Amino acid metabolism: Theoretical effects on protein homeostasis
- Lipid metabolism: Potential influences on lipid profiles
Current clinical data has not revealed significant safety concerns related to these theoretical effects, but ongoing Phase III trials will provide more definitive assessment of glucagon-specific safety parameters relevant to retatrutide long term safety.
Special Populations and Safety Considerations
Comprehensive understanding of retatrutide long term safety requires examination of how safety profiles may vary across different population subgroups.
Metabolic Disease Populations 🔍
Patients with type 2 diabetes represent a primary target population for retatrutide research:
Safety Considerations in Diabetic Populations:
- Hypoglycemia risk: Lower than insulin or sulfonylureas, but requires monitoring when combined with other glucose-lowering agents
- Diabetic gastroparesis: Pre-existing GI motility disorders may exacerbate nausea and vomiting
- Renal impairment: Common in advanced diabetes; requires careful monitoring despite favorable renal safety profile
- Cardiovascular disease: High prevalence necessitates comprehensive CV safety assessment
The ongoing TRIUMPH-2 trial specifically addresses retatrutide long term safety in type 2 diabetes populations, with results expected in May 2026 providing critical insights for researchers investigating metabolic applications.
Obstructive Sleep Apnea Research
The TRIUMPH-3 trial examining retatrutide in obstructive sleep apnea populations introduces unique safety considerations:
OSA-Specific Safety Factors:
- Respiratory effects: Monitoring for any influence on respiratory drive or mechanics
- Weight loss velocity: Rapid weight reduction may affect airway anatomy
- Cardiovascular comorbidities: High prevalence in OSA populations
- Medication interactions: Common use of CPAP and other therapies
Elderly Populations
Age-related physiological changes may influence retatrutide long term safety profiles:
Geriatric Considerations:
- Reduced renal clearance: May affect drug metabolism and clearance
- Polypharmacy: Increased potential for drug interactions
- Frailty: Greater vulnerability to dehydration and electrolyte imbalances
- Cognitive factors: May affect adherence to dosing and monitoring protocols
Research Applications and Safety Protocol Design
For researchers utilizing research-grade peptides from PEPTIDE PRO, understanding retatrutide long term safety informs optimal experimental protocol design.
Designing Safety-Conscious Research Protocols
Essential Protocol Elements:
- Clear inclusion/exclusion criteria: Define populations appropriate for study based on current safety knowledge
- Comprehensive baseline assessment: Establish pre-treatment organ function and metabolic parameters
- Structured titration schedules: Implement gradual dose escalation based on clinical trial evidence
- Regular monitoring intervals: Schedule assessments aligned with expected adverse event timelines
- Defined stopping criteria: Establish clear parameters for protocol modification or discontinuation
- Data collection systems: Capture adverse events systematically for analysis
Ethical Considerations in Retatrutide Research
Retatrutide long term safety considerations extend to ethical dimensions of research design:
Key Ethical Principles:
- Informed consent: Clear communication of known and unknown safety aspects
- Risk minimization: Protocol design that reduces unnecessary exposure
- Benefit-risk assessment: Ongoing evaluation of research value versus participant risk
- Vulnerable populations: Enhanced protections for high-risk groups
- Data transparency: Commitment to reporting both positive and negative safety findings
Researchers can access detailed product information and certificates of analysis to ensure compound purity and quality, critical factors in safety-conscious research design.
Future Directions in Retatrutide Safety Research
The evolving understanding of retatrutide long term safety will continue to develop through multiple research avenues.
Emerging Research Questions 🔬
Priority Safety Research Areas:
- Multi-year safety outcomes: Effects beyond 2-3 years of continuous exposure
- Rare adverse events: Detection of low-frequency safety signals requiring large populations
- Mechanistic safety studies: Understanding biological basis of observed adverse events
- Biomarker development: Identifying predictive markers for adverse event susceptibility
- Combination safety: Interactions with other therapeutic agents
- Withdrawal effects: Safety of discontinuation after long-term exposure
Real-World Safety Data Collection
Post-marketing surveillance and real-world evidence will complement controlled trial data:
Real-World Safety Monitoring:
- Pharmacovigilance systems: Systematic collection of adverse event reports
- Registry studies: Long-term follow-up of diverse patient populations
- Electronic health records: Mining large datasets for safety signals
- Patient-reported outcomes: Capturing tolerability from user perspective
Personalized Safety Assessment
Future research may enable individualized retatrutide long term safety prediction:
Precision Safety Approaches:
- Genetic screening: Identifying polymorphisms affecting drug metabolism or receptor sensitivity
- Baseline biomarkers: Predicting GI tolerability or metabolic response
- Machine learning models: Integrating multiple factors for personalized risk assessment
- Pharmacokinetic profiling: Optimizing dosing based on individual clearance patterns
Regulatory Perspectives on Retatrutide Long Term Safety
Understanding regulatory frameworks provides context for retatrutide long term safety assessment standards.
Regulatory Requirements for Long-Term Safety Data
Regulatory agencies require comprehensive safety demonstration before approval:
Key Regulatory Expectations:
- Duration: Minimum 12-24 months of controlled exposure data
- Population size: Thousands of subjects to detect rare events
- Special populations: Safety data in relevant subgroups
- Cardiovascular outcomes: Rigorous CV safety assessment for metabolic drugs
- Post-marketing commitments: Ongoing safety monitoring after approval
The TRIUMPH trial program is specifically designed to meet these regulatory standards, providing the definitive retatrutide long term safety evidence required for potential therapeutic applications.
Quality Standards for Research-Grade Compounds
For researchers utilizing retatrutide in laboratory settings, compound quality directly influences safety assessment:
Critical Quality Parameters:
- Purity levels: ≥98% purity for reliable safety evaluation
- Endotoxin content: Low endotoxin levels to prevent confounding inflammatory effects
- Storage conditions: Proper handling to maintain compound integrity
- Certificates of analysis: Documentation of batch-specific quality metrics
PEPTIDE PRO’s commitment to exceptional purity and comprehensive quality documentation supports rigorous safety-conscious research applications.
Current State of Retatrutide Long Term Safety Knowledge
The accumulated evidence regarding retatrutide long term safety presents an encouraging profile for this novel triple agonist peptide. Clinical trial data encompassing 878 patients demonstrates no significant increase in overall adverse events compared to placebo, with a predictable pattern of primarily gastrointestinal effects that align with the established incretin-based therapeutic class.
Key Conclusions:
✅ Favorable overall safety profile with relative risk of adverse events (1.11) not significantly different from placebo
✅ Manageable adverse events predominantly gastrointestinal in nature and responsive to proper dose titration
✅ No hepatotoxicity or renal impairment signals observed in Phase 2 trials
✅ Ongoing Phase III TRIUMPH trials will provide definitive long-term safety data by 2026
✅ Safety characteristics align with established GLP-1 agonists, suggesting predictable risk profiles
However, researchers must acknowledge important limitations in current knowledge. Observation periods remain relatively short, sample sizes are modest for detecting rare events, and multi-year safety data is not yet available. The retatrutide long term safety profile will continue to evolve as Phase III trials progress and real-world evidence accumulates.
Actionable Next Steps for Researchers
For researchers considering retatrutide in experimental protocols:
- Stay informed: Monitor publication of TRIUMPH trial results, particularly TRIUMPH-2 (May 2026)
- Design robust protocols: Incorporate gradual titration, comprehensive monitoring, and clear safety endpoints
- Source quality compounds: Utilize research-grade peptides from established suppliers with rigorous quality standards
- Document systematically: Maintain detailed adverse event records to contribute to safety knowledge
- Engage ethically: Ensure appropriate informed consent and risk communication in research involving human subjects
The current evidence supports cautious optimism regarding retatrutide long term safety, with the compound demonstrating a manageable safety profile suitable for continued research investigation. As Phase III trials progress and our understanding deepens, retatrutide may represent a valuable addition to the research toolkit for investigating multi-receptor metabolic pathways.
For researchers seeking high-purity retatrutide and other research peptides, PEPTIDE PRO offers exceptional quality, comprehensive documentation, and professional service supporting rigorous scientific investigation.
Disclaimer: This article discusses retatrutide strictly in the context of research applications. All peptides supplied by PEPTIDE PRO are clearly labelled “For Research Use Only” and are not intended for human consumption or therapeutic use. Researchers must comply with all applicable regulations and ethical standards when conducting peptide research.
