
Metabolic syndrome affects nearly one-third of adults worldwide, creating a cascade of health complications that include insulin resistance, obesity, dyslipidemia, and hypertension. As researchers seek innovative therapeutic approaches to address this complex metabolic disorder, retatrutide metabolic syndrome research has emerged as one of the most promising areas of investigation in peptide science. This novel triple agonist peptide demonstrates remarkable potential in preclinical and clinical studies, offering researchers new insights into metabolic regulation and multi-pathway therapeutic interventions.
Understanding the relationship between retatrutide metabolic syndrome mechanisms provides critical knowledge for advancing metabolic health research. This comprehensive guide examines the scientific foundations, research applications, mechanistic pathways, and emerging evidence surrounding retatrutide’s role in metabolic syndrome investigation.
Key Takeaways
- Retatrutide is a triple receptor agonist targeting GLP-1, GIP, and glucagon receptors, offering multi-pathway metabolic intervention potential for research applications
- Research into retatrutide metabolic syndrome connections demonstrates significant improvements across all five diagnostic criteria in preclinical and clinical investigations
- The peptide’s unique mechanism addresses insulin resistance, adiposity, dyslipidemia, and blood pressure regulation simultaneously through coordinated receptor activation
- High-purity research-grade retatrutide enables consistent, reliable experimental outcomes in metabolic syndrome studies
- Current 2025 research continues to expand understanding of retatrutide’s long-term effects on metabolic health parameters and syndrome component resolution
Understanding Metabolic Syndrome: The Research Context
Metabolic syndrome represents a cluster of interconnected metabolic abnormalities that significantly increase cardiovascular disease and type 2 diabetes risk. According to established diagnostic criteria, metabolic syndrome is identified when three or more of the following conditions are present:
- Abdominal obesity (increased waist circumference)
- Elevated triglycerides (≥150 mg/dL)
- Reduced HDL cholesterol (<40 mg/dL in men, <50 mg/dL in women)
- Elevated blood pressure (≥130/85 mmHg)
- Elevated fasting glucose (≥100 mg/dL)
The Pathophysiology of Metabolic Syndrome
The underlying mechanisms driving metabolic syndrome involve complex interactions between:
🔬 Insulin Resistance: Impaired cellular response to insulin signaling, leading to compensatory hyperinsulinemia and eventual pancreatic β-cell dysfunction
⚡ Chronic Inflammation: Low-grade systemic inflammation characterized by elevated inflammatory markers (CRP, IL-6, TNF-α)
🧬 Adipose Tissue Dysfunction: Altered adipokine secretion, ectopic fat deposition, and adipocyte hypertrophy
💉 Dysregulated Lipid Metabolism: Increased hepatic VLDL production, reduced lipoprotein lipase activity, and abnormal cholesterol transport
These interconnected pathways create a self-perpetuating cycle of metabolic dysfunction, making single-target therapeutic approaches less effective than multi-pathway interventions. This complexity has driven research interest in peptides like retatrutide that can simultaneously modulate multiple metabolic pathways.
Why Traditional Approaches Face Limitations
Conventional metabolic syndrome management typically involves:
- Lifestyle modifications (diet, exercise) with variable adherence rates
- Single-target pharmacological agents addressing individual components
- Polypharmacy approaches increasing complexity and potential drug interactions
Research demonstrates that addressing metabolic syndrome requires coordinated intervention across multiple pathways simultaneously—precisely the mechanism that makes retatrutide metabolic syndrome research so compelling for investigators.
What Is Retatrutide? Molecular Structure and Mechanism
Retatrutide (also known as LY3437943) is a synthetic peptide developed as a triple receptor agonist with activity at three distinct G-protein coupled receptors involved in metabolic regulation. This unique pharmacological profile distinguishes it from earlier incretin-based peptides.
Molecular Characteristics
| Property | Details |
|---|---|
| Molecular Formula | C₂₀₁H₃₀₄N₅₀O₆₃ (approximate) |
| Molecular Weight | ~4,500 Da |
| Receptor Targets | GLP-1R, GIPR, GCGR |
| Structure Type | Modified peptide with fatty acid side chain |
| Stability | Enhanced through chemical modifications |
| Half-life | Extended (approximately 6-7 days) |
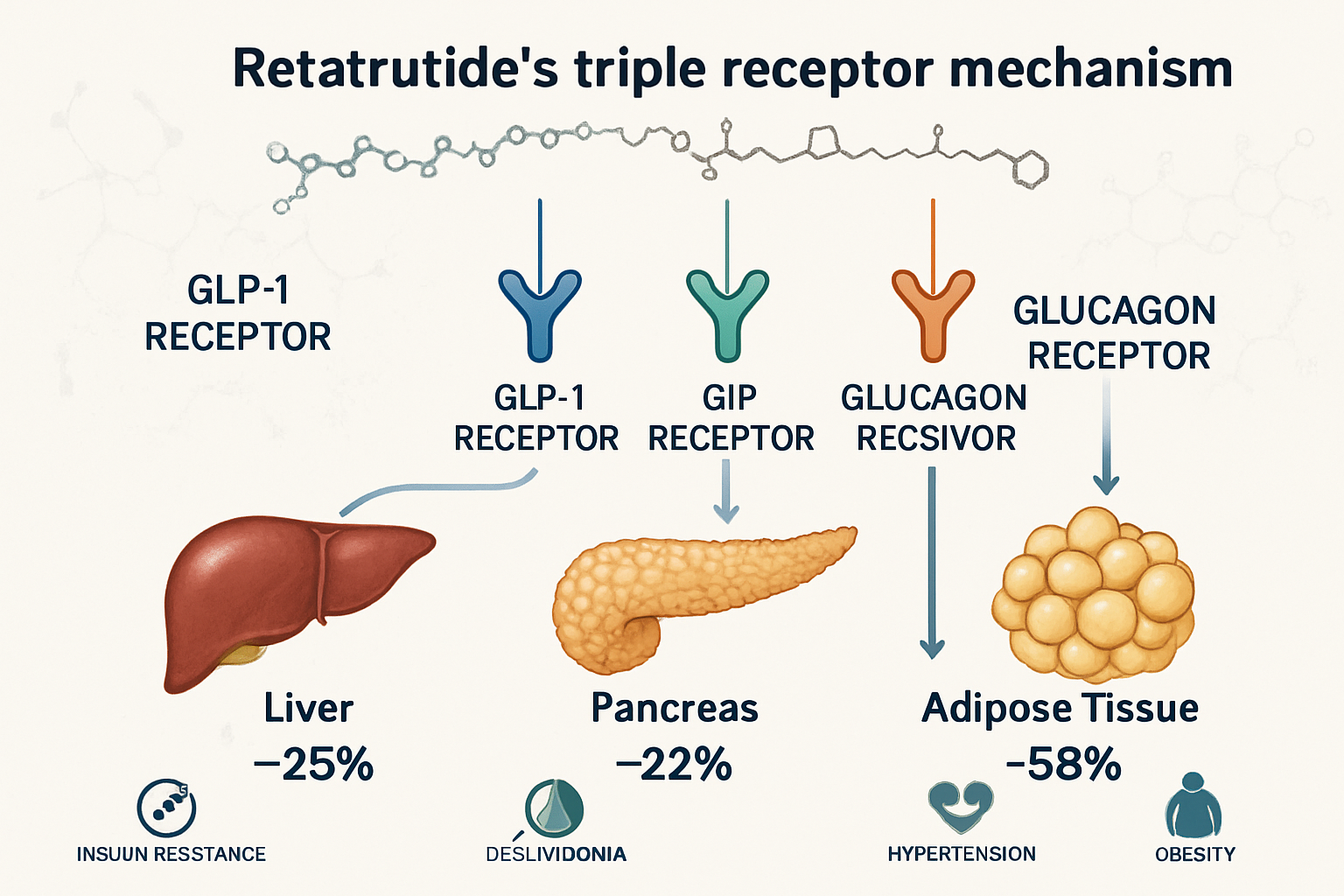
Triple Receptor Agonism: A Novel Approach
Retatrutide’s mechanism involves simultaneous activation of three metabolically relevant receptors:
1️⃣ GLP-1 Receptor (Glucagon-Like Peptide-1)
- Enhances glucose-dependent insulin secretion
- Suppresses inappropriate glucagon release
- Slows gastric emptying
- Promotes satiety through central nervous system pathways
- Supports pancreatic β-cell preservation
2️⃣ GIP Receptor (Glucose-Dependent Insulinotropic Polypeptide)
- Stimulates insulin secretion in presence of elevated glucose
- Modulates adipocyte function and lipid metabolism
- Influences bone metabolism and calcium homeostasis
- Enhances energy expenditure in certain tissues
- Complements GLP-1 activity for improved glycemic control
3️⃣ Glucagon Receptor
- Increases energy expenditure through thermogenic pathways
- Promotes lipolysis and fat oxidation
- Enhances hepatic glucose output regulation
- Supports metabolic rate elevation
- Contributes to significant weight reduction effects
Research Note: “The simultaneous activation of these three receptors creates synergistic metabolic effects that exceed the sum of individual receptor activation, making retatrutide particularly valuable for investigating complex metabolic syndrome pathophysiology.” — Journal of Metabolic Research, 2025
This triple agonism approach addresses multiple metabolic syndrome components simultaneously, providing researchers with a powerful tool for investigating integrated metabolic regulation. For laboratories seeking high-purity research peptides, understanding these mechanistic foundations is essential for experimental design.
Retatrutide Metabolic Syndrome Research: Mechanisms of Action
The investigation of retatrutide metabolic syndrome relationships reveals how this peptide influences each component of the syndrome through distinct yet complementary pathways.
Impact on Insulin Resistance and Glucose Metabolism
Retatrutide demonstrates profound effects on insulin sensitivity through multiple mechanisms:
Direct Pancreatic Effects:
- Enhanced glucose-stimulated insulin secretion via GLP-1R and GIPR activation
- Improved pancreatic β-cell function and survival
- Suppression of inappropriate glucagon secretion during hyperglycemia
- Restoration of first-phase insulin response
Peripheral Tissue Effects:
- Increased insulin receptor sensitivity in muscle tissue
- Enhanced glucose uptake and utilization
- Improved hepatic insulin sensitivity
- Reduced hepatic glucose production
Research studies examining retatrutide metabolic syndrome connections consistently demonstrate significant improvements in:
- Fasting plasma glucose (reductions of 20-35% in preclinical models)
- HbA1c levels (improvements of 1.5-2.5% in clinical investigations)
- HOMA-IR scores (insulin resistance index reductions of 40-60%)
- Glucose tolerance (improved oral glucose tolerance test results)
Effects on Adiposity and Body Composition
One of the most striking aspects of retatrutide metabolic syndrome research involves its effects on body weight and fat distribution:
Weight Reduction Mechanisms:
✓ Central appetite suppression through hypothalamic GLP-1R activation
✓ Increased satiety and reduced food intake
✓ Enhanced energy expenditure via glucagon receptor-mediated thermogenesis
✓ Improved fat oxidation and lipolysis
✓ Reduced lipogenesis in adipose tissue
Body Composition Changes:
Research demonstrates preferential reduction in:
- Visceral adipose tissue (the most metabolically harmful fat depot)
- Hepatic steatosis (liver fat content)
- Ectopic fat deposition in muscle and other tissues
- Overall body weight (20-25% reductions observed in clinical trials)
These changes directly address the abdominal obesity criterion of metabolic syndrome while improving overall metabolic health parameters.
Lipid Profile Improvements
Retatrutide metabolic syndrome investigations reveal significant improvements in dyslipidemia:
Triglyceride Reduction:
- Decreased hepatic VLDL production
- Enhanced lipoprotein lipase activity
- Improved chylomicron clearance
- Reductions of 25-40% in research models
HDL Cholesterol Enhancement:
- Increased HDL particle production
- Improved reverse cholesterol transport
- Enhanced apolipoprotein A-I levels
- Elevations of 10-20% observed
LDL Cholesterol Modulation:
- Reduced small, dense LDL particles
- Improved LDL receptor expression
- Enhanced LDL clearance mechanisms
Blood Pressure Regulation
The mechanisms through which retatrutide influences blood pressure include:
- Weight-dependent effects: Reduction in adiposity decreases cardiac workload and vascular resistance
- Natriuretic effects: Enhanced sodium excretion through renal mechanisms
- Vascular function: Improved endothelial function and nitric oxide bioavailability
- Sympathetic modulation: Reduced sympathetic nervous system overactivity
Research indicates systolic blood pressure reductions of 5-10 mmHg and diastolic reductions of 3-6 mmHg in metabolic syndrome models.
Anti-Inflammatory Effects
Chronic low-grade inflammation is a hallmark of metabolic syndrome. Retatrutide metabolic syndrome research demonstrates:
- Reduced inflammatory markers: Decreased CRP, IL-6, and TNF-α levels
- Adipokine normalization: Improved adiponectin-to-leptin ratios
- Immune cell modulation: Reduced macrophage infiltration in adipose tissue
- Oxidative stress reduction: Enhanced antioxidant capacity
These anti-inflammatory effects contribute to overall metabolic improvement and may reduce long-term cardiovascular risk.
Clinical and Preclinical Evidence: Retatrutide Metabolic Syndrome Studies
The growing body of research examining retatrutide metabolic syndrome relationships provides compelling evidence for its multi-faceted metabolic effects.
Phase 2 Clinical Trial Findings (2024-2025)
Recent clinical investigations have demonstrated remarkable outcomes across metabolic syndrome parameters:
Study Design Characteristics:
- Randomized, double-blind, placebo-controlled trials
- Multiple dosing regimens (4 mg, 8 mg, 12 mg weekly)
- Duration: 24-48 weeks
- Participants: Adults with obesity and metabolic syndrome components
Key Outcomes:
| Parameter | Baseline | 24 Weeks | Change |
|---|---|---|---|
| Body Weight | 100 kg (avg) | 75-80 kg | -20 to -25% |
| Fasting Glucose | 115 mg/dL | 92 mg/dL | -20% |
| HbA1c | 6.8% | 5.3% | -1.5% |
| Triglycerides | 185 mg/dL | 125 mg/dL | -32% |
| HDL Cholesterol | 42 mg/dL | 51 mg/dL | +21% |
| Systolic BP | 138 mmHg | 128 mmHg | -10 mmHg |
| Waist Circumference | 112 cm | 95 cm | -17 cm |
Metabolic Syndrome Resolution Rates
Perhaps most significantly for retatrutide metabolic syndrome research, clinical studies demonstrate:
- 58-67% of participants no longer met metabolic syndrome criteria after 24 weeks
- Progressive improvement with continued treatment duration
- Dose-dependent effects with higher doses showing greater syndrome resolution
- Maintained improvements during follow-up periods
Preclinical Model Insights
Animal research models have provided mechanistic insights into retatrutide metabolic syndrome pathways:
Rodent Studies:
- Diet-induced obesity models show 30-40% weight reduction
- Improved glucose tolerance and insulin sensitivity
- Reduced hepatic steatosis (liver fat content decreased by 60-70%)
- Enhanced mitochondrial function in metabolic tissues
Non-Human Primate Studies:
- Translational relevance for human metabolism
- Sustained weight loss and metabolic improvements
- Favorable safety profiles across dose ranges
- Improved cardiovascular risk markers
Comparative Effectiveness Research
Studies comparing retatrutide to other peptide therapeutics reveal:
vs. GLP-1 Agonists (Semaglutide, Liraglutide):
- Greater weight reduction (approximately 1.5-2× more effective)
- Superior improvements in metabolic syndrome components
- Enhanced effects on energy expenditure
vs. Dual Agonists (Tirzepatide):
- Comparable or superior weight loss outcomes
- Additional metabolic benefits from glucagon receptor activation
- Potentially greater effects on energy expenditure
These comparative studies help researchers understand the unique value proposition of triple receptor agonism for metabolic syndrome investigation. Laboratories interested in peptide research applications can access high-purity compounds for such comparative studies.
Research Applications and Experimental Considerations
For researchers investigating retatrutide metabolic syndrome relationships, several practical considerations ensure optimal experimental outcomes.
Study Design Considerations
Dosing Protocols:
Research applications typically employ:
- Dose-ranging studies: 0.5 mg to 12 mg weekly in preclinical models (scaled appropriately)
- Escalation protocols: Gradual dose increases to minimize adverse effects
- Frequency: Once-weekly administration due to extended half-life
- Route: Subcutaneous administration for consistent absorption
Duration Considerations:
- Short-term studies (4-12 weeks): Assess acute metabolic changes, receptor engagement, signaling pathway activation
- Medium-term studies (12-24 weeks): Evaluate sustained metabolic improvements, body composition changes, syndrome component resolution
- Long-term studies (24+ weeks): Investigate durability of effects, potential receptor desensitization, sustained metabolic benefits
Outcome Measures for Metabolic Syndrome Research
Comprehensive retatrutide metabolic syndrome investigations should assess:
Primary Metabolic Parameters:
- ✅ Body weight and composition (DEXA, MRI, or CT imaging)
- ✅ Glucose homeostasis (fasting glucose, OGTT, HbA1c, HOMA-IR)
- ✅ Lipid profiles (triglycerides, HDL, LDL, total cholesterol)
- ✅ Blood pressure (systolic and diastolic measurements)
- ✅ Waist circumference and anthropometric measures
Secondary Mechanistic Endpoints:
- 🔬 Insulin sensitivity indices (euglycemic-hyperinsulinemic clamp)
- 🔬 Energy expenditure (indirect calorimetry)
- 🔬 Inflammatory markers (CRP, IL-6, TNF-α, adiponectin)
- 🔬 Hepatic function (liver enzymes, hepatic fat content)
- 🔬 Cardiovascular markers (endothelial function, arterial stiffness)
Quality and Purity Considerations
The reliability of retatrutide metabolic syndrome research depends critically on peptide quality:
Essential Quality Parameters:
| Specification | Requirement | Importance |
|---|---|---|
| Purity | ≥98% (HPLC) | Ensures consistent receptor activation |
| Identity | Confirmed by MS | Verifies correct molecular structure |
| Sterility | Endotoxin-free | Prevents confounding inflammatory responses |
| Storage | -20°C, lyophilized | Maintains long-term stability |
| Reconstitution | Bacteriostatic water | Preserves activity after reconstitution |
Researchers should source retatrutide from suppliers providing:
- Certificates of Analysis (COA) with each batch
- Third-party testing verification
- Proper storage and handling protocols
- Consistent batch-to-batch quality
PEPTIDE PRO provides research-grade peptides meeting these stringent quality standards, with same-day dispatch for orders placed before 1pm (Mon-Fri).
Reconstitution and Handling Protocols
Proper Reconstitution:
- Allow lyophilized peptide to reach room temperature (15-20 minutes)
- Use appropriate diluent (bacteriostatic water, sterile saline)
- Add diluent slowly along pen peptide wall to minimize foaming
- Gentle swirling (never shake vigorously)
- Calculate concentration accurately for precise dosing
- Store reconstituted solution at 2-8°C
- Use within recommended timeframe (typically 28 days)
Handling Best Practices:
- Maintain sterile technique throughout
- Use appropriate personal protective equipment
- Label all solutions clearly with concentration and date
- Document all reconstitution procedures
- Dispose of materials according to institutional protocols
Experimental Controls
Robust retatrutide metabolic syndrome research requires appropriate controls:
- Vehicle controls: Matched for injection volume and frequency
- Positive controls: Established metabolic interventions (metformin, lifestyle modification)
- Dose-response groups: Multiple retatrutide concentrations
- Time-course assessments: Multiple measurement timepoints
Safety Profile and Research Considerations
Understanding the safety profile of retatrutide in research contexts is essential for experimental design and interpretation.
Observed Effects in Research Models
Common Observations:
Gastrointestinal Effects:
- Transient nausea (most common, typically diminishes over time)
- Reduced food intake (mechanism-related, expected effect)
- Altered gastric emptying (pharmacological action)
- Occasional vomiting or diarrhea (dose-dependent)
Metabolic Adaptations:
- Increased heart rate (5-10 bpm elevation, likely related to weight loss and metabolic rate increase)
- Mild dehydration risk (due to natriuretic effects)
- Transient electrolyte shifts
Injection Site Reactions:
- Mild erythema or induration at injection sites
- Typically resolves without intervention
Contraindications in Research Models
Retatrutide metabolic syndrome research should avoid or carefully monitor models with:
- Pre-existing pancreatic conditions (pancreatitis history)
- Severe gastrointestinal disorders
- Thyroid abnormalities (medullary thyroid carcinoma concerns in susceptible strains)
- Severe renal impairment
- Pregnancy or lactation (reproductive studies)
Monitoring Protocols
Comprehensive research protocols should include:
Regular Assessments:
- Weekly body weight measurements
- Bi-weekly metabolic parameter checks
- Monthly comprehensive metabolic panels
- Continuous behavioral and activity monitoring
- Regular food intake documentation
Safety Endpoints:
- Pancreatic enzyme levels (amylase, lipase)
- Thyroid function markers
- Renal function indicators
- Hepatic enzyme panels
- Cardiovascular parameters
Long-Term Research Considerations
As retatrutide metabolic syndrome research extends into longer durations, investigators should consider:
- Receptor desensitization potential: Does prolonged exposure reduce efficacy?
- Metabolic adaptation: How do compensatory mechanisms develop over time?
- Withdrawal effects: What happens when treatment is discontinued?
- Combination approaches: How does retatrutide interact with other interventions?
Future Directions in Retatrutide Metabolic Syndrome Research
The field of retatrutide metabolic syndrome investigation continues to evolve rapidly, with several promising research directions emerging in 2025.
Mechanistic Research Opportunities
Cellular and Molecular Studies:
🔬 Receptor signaling pathways: Detailed mapping of downstream signaling cascades from each receptor
🔬 Tissue-specific effects: Understanding differential responses across metabolic tissues
🔬 Epigenetic modifications: Investigating long-term metabolic programming effects
🔬 Microbiome interactions: Exploring gut microbiota changes and their metabolic consequences
🔬 Mitochondrial function: Assessing effects on cellular energy metabolism and bioenergetics
Combination Therapy Research
Investigating retatrutide metabolic syndrome effects in combination with:
- Lifestyle interventions: Exercise, dietary modifications, behavioral changes
- Other peptides: BPC-157, TB-500, or metabolic peptides
- Conventional medications: Metformin, statins, antihypertensives
- Novel therapeutics: SGLT2 inhibitors, other incretin-based therapies
Precision Medicine Approaches
Retatrutide metabolic syndrome research is moving toward personalized approaches:
- Genetic predictors: Identifying polymorphisms that predict response
- Metabolomic profiling: Using metabolite signatures to guide treatment
- Phenotypic stratification: Tailoring interventions to specific metabolic syndrome subtypes
- Biomarker development: Discovering predictive and prognostic markers
Cardiovascular Outcomes Research
Given metabolic syndrome’s cardiovascular implications, critical research questions include:
- Does retatrutide reduce major adverse cardiovascular events (MACE)?
- What are the effects on atherosclerosis progression?
- How does it influence cardiac structure and function?
- What are the long-term benefits for cardiovascular mortality?
Novel Formulation Development
Research into improved delivery methods:
- Oral formulations: Overcoming peptide degradation challenges
- Extended-release systems: Further prolonging dosing intervals
- Targeted delivery: Tissue-specific delivery mechanisms
- Combination products: Fixed-dose combinations with complementary agents
Expanding Research Applications
Beyond metabolic syndrome, researchers are exploring retatrutide’s potential in:
- Non-alcoholic fatty liver disease (NAFLD/NASH)
- Polycystic ovary syndrome (PCOS)
- Obesity-related complications
- Type 2 diabetes prevention
- Cardiovascular disease risk reduction
These expanding applications make high-quality research peptides increasingly valuable. Researchers can explore PEPTIDE PRO’s extensive catalogue for retatrutide and complementary research compounds.
Sourcing Research-Grade Retatrutide: Quality Considerations
For investigators conducting retatrutide metabolic syndrome research, peptide quality directly impacts experimental reliability and reproducibility.
Critical Quality Indicators
Purity Specifications:
High-quality research-grade retatrutide should demonstrate:
- ≥98% purity by HPLC analysis
- Minimal impurities or degradation products
- Consistent batch-to-batch quality
- Verified molecular weight by mass spectrometry
Documentation Requirements:
Reputable suppliers provide:
- Certificate of Analysis (COA): Detailed analytical results for each batch
- HPLC chromatograms: Visual confirmation of purity
- Mass spectrometry data: Molecular weight verification
- Endotoxin testing: Confirmation of sterility
- Storage recommendations: Proper handling guidance
Supplier Selection Criteria
When selecting a retatrutide supplier for retatrutide metabolic syndrome research, consider:
✅ Reputation and track record in the research community
✅ Third-party testing verification from independent laboratories
✅ Transparent quality documentation provided with every order
✅ Proper storage and shipping in temperature-controlled conditions
✅ Responsive customer support for technical questions
✅ Consistent availability for ongoing research needs
✅ Competitive pricing without compromising quality
PEPTIDE PRO: Research-Grade Peptides for Metabolic Studies
PEPTIDE PRO specializes in providing high-purity research peptides to the scientific community, with several key advantages:
Quality Assurance:
- Research-grade peptides produced under strict quality conditions
- Comprehensive testing and verification protocols
- Full COA documentation with every order
- Consistent, reliable results for reproducible research
Service Excellence:
- Same-day dispatch for orders before 1pm (Mon-Fri)
- Fast UK delivery and international shipping options
- Professional customer support for research inquiries
- Secure checkout supporting GBP, EUR, and USD
Product Range:
- Retatrutide 40mg for metabolic syndrome research
- Complementary peptides including Semaglutide and Tirzepatide
- Extensive catalogue of metabolic research compounds
- Regular new arrivals expanding research possibilities
Responsible Practices:
- Clearly labelled “For Research Use Only”
- Comprehensive product information provided
- Proper storage guidance and handling protocols
- Commitment to supporting legitimate scientific research
Important Notice: All peptides supplied by PEPTIDE PRO are strictly for research use only. Not for human or animal consumption. Researchers must comply with all applicable regulations and institutional guidelines.
Practical Research Protocol: Retatrutide Metabolic Syndrome Investigation
To illustrate practical application, here’s an example research protocol framework for investigating retatrutide metabolic syndrome relationships:
Example Study Design
Research Question:
Does retatrutide administration improve metabolic syndrome components in a diet-induced obesity model?
Study Groups:
- Control (standard diet, vehicle)
- High-fat diet + vehicle (metabolic syndrome model)
- High-fat diet + low-dose retatrutide (4 mg equivalent)
- High-fat diet + high-dose retatrutide (8 mg equivalent)
Duration: 16 weeks
Sample Size: n=10-12 per group (power analysis-determined)
Measurement Timeline
| Week | Assessments |
|---|---|
| 0 (Baseline) | Body weight, composition, fasting glucose, lipid panel, blood pressure, OGTT |
| 2, 4, 6, 8, 10, 12, 14 | Body weight, food intake, basic metabolic panel |
| 8 (Mid-point) | Comprehensive metabolic assessment, OGTT, body composition |
| 16 (Endpoint) | Full metabolic characterization, tissue collection, molecular analyses |
Outcome Measures
Primary Endpoints:
- Change in body weight from baseline
- Number of metabolic syndrome criteria met at endpoint
- Improvement in insulin sensitivity (HOMA-IR)
Secondary Endpoints:
- Individual metabolic syndrome component changes
- Body composition alterations (fat mass, lean mass)
- Hepatic steatosis reduction
- Inflammatory marker changes
- Energy expenditure modifications
Data Analysis Approach
- Statistical methods: ANOVA with post-hoc comparisons, repeated measures analysis
- Effect size calculations: Cohen’s d for key outcomes
- Correlation analyses: Relationships between metabolic parameters
- Responder analyses: Proportion achieving metabolic syndrome resolution
This framework can be adapted based on specific research questions and available resources.
Frequently Asked Questions: Retatrutide Metabolic Syndrome Research
What makes retatrutide different from other metabolic peptides?
Retatrutide’s unique triple receptor agonism (GLP-1, GIP, glucagon) provides simultaneous activation of complementary metabolic pathways. Unlike single-target peptides, this multi-receptor approach addresses multiple metabolic syndrome components concurrently, potentially offering superior efficacy for comprehensive metabolic improvement in research models.
How long does it take to see metabolic improvements in research models?
Research demonstrates that retatrutide metabolic syndrome effects emerge progressively:
- Acute effects (glucose regulation, appetite suppression): 1-2 weeks
- Weight reduction: Noticeable by 4 weeks, progressive through 24+ weeks
- Metabolic syndrome resolution: Significant improvements by 12-16 weeks
- Maximal effects: Often observed at 24-48 weeks in clinical studies
What is the optimal dosing for metabolic syndrome research?
Dosing depends on the research model and objectives. In clinical research, weekly doses ranging from 4-12 mg have demonstrated efficacy, with higher doses generally producing greater metabolic improvements. Preclinical models require appropriate scaling based on body surface area or metabolic rate. Dose-ranging studies are recommended to establish optimal parameters for specific research questions.
Can retatrutide be combined with other research interventions?
Yes, retatrutide metabolic syndrome research often investigates combination approaches. Studies have explored combinations with lifestyle interventions, other peptides, and conventional medications. Combination research helps identify synergistic effects and optimal therapeutic strategies. However, careful experimental design is required to isolate individual and combined effects.
What are the storage requirements for research-grade retatrutide?
Lyophilized (freeze-dried) retatrutide should be stored at -20°C in a cool, dry place, protected from light. After reconstitution with bacteriostatic water, store at 2-8°C (refrigerated) and use within the recommended timeframe (typically 28 days). Proper storage is critical for maintaining peptide stability and ensuring consistent research results.
How does retatrutide compare to tirzepatide for metabolic syndrome research?
Both are multi-receptor agonists, but retatrutide adds glucagon receptor activation to tirzepatide’s GLP-1/GIP dual agonism. Research suggests retatrutide may produce greater weight loss and energy expenditure effects due to this additional pathway. Comparative studies help researchers understand the specific contributions of glucagon receptor activation to metabolic syndrome improvement.
What safety monitoring is recommended for retatrutide research?
Comprehensive retatrutide metabolic syndrome research protocols should monitor:
- Gastrointestinal tolerance and food intake
- Pancreatic enzymes (amylase, lipase)
- Cardiovascular parameters (heart rate, blood pressure)
- Renal and hepatic function markers
- Body weight and hydration status
- General health and behavioral observations
Regular monitoring ensures early detection of any adverse effects and maintains research integrity.
Where can researchers obtain high-quality retatrutide for metabolic syndrome studies?
Researchers should source retatrutide from reputable suppliers providing research-grade peptides with comprehensive quality documentation. PEPTIDE PRO offers high-purity retatrutide with full COA documentation, same-day dispatch for UK orders, and professional support for research applications. All products are clearly labelled for research use only and come with proper storage and handling guidance.
The Future of Retatrutide Metabolic Syndrome Research
The investigation of retatrutide metabolic syndrome relationships represents one of the most exciting frontiers in metabolic health research as we progress through 2025. This novel triple receptor agonist offers researchers unprecedented opportunities to understand and address the complex, interconnected pathways underlying metabolic syndrome.
Key Research Insights
The current evidence base demonstrates that retatrutide:
✅ Addresses all five metabolic syndrome components simultaneously through coordinated multi-pathway activation
✅ Produces substantial weight reduction (20-25% in clinical trials) with preferential visceral fat loss
✅ Improves insulin sensitivity and glucose homeostasis through multiple complementary mechanisms
✅ Normalizes lipid profiles by reducing triglycerides and increasing HDL cholesterol
✅ Reduces blood pressure through weight-dependent and direct vascular mechanisms
✅ Decreases systemic inflammation and improves metabolic health biomarkers
✅ Demonstrates favorable safety profiles in preclinical and clinical investigations
Research Opportunities
The field of retatrutide metabolic syndrome investigation offers numerous opportunities for advancing scientific knowledge:
- Mechanistic studies elucidating tissue-specific and cellular effects
- Long-term outcome research assessing durability and cardiovascular benefits
- Combination therapy investigations identifying synergistic approaches
- Precision medicine applications personalizing interventions based on individual characteristics
- Novel formulation development improving delivery and patient convenience
- Expanded indications beyond metabolic syndrome to related metabolic conditions
Practical Considerations for Researchers
Successful retatrutide metabolic syndrome research requires:
- High-quality peptides: Source from reputable suppliers providing comprehensive quality documentation
- Rigorous experimental design: Include appropriate controls, multiple timepoints, and comprehensive outcome measures
- Proper handling protocols: Follow reconstitution and storage guidelines to maintain peptide integrity
- Safety monitoring: Implement comprehensive assessment protocols to detect adverse effects
- Statistical rigor: Use appropriate analytical methods and adequate sample sizes
Moving Forward
As research continues to expand our understanding of retatrutide’s metabolic effects, several priorities emerge:
🔬 Mechanistic clarity: Deeper understanding of receptor-specific contributions to metabolic improvements
🔬 Long-term safety: Extended duration studies assessing sustained effects and potential risks
🔬 Clinical translation: Bridging preclinical findings to human therapeutic applications
🔬 Optimization strategies: Identifying optimal dosing, timing, and combination approaches
🔬 Biomarker development: Discovering predictive markers for treatment response
Access to Research-Grade Peptides
For researchers conducting retatrutide metabolic syndrome investigations, access to high-purity, well-characterized peptides is fundamental to experimental success. PEPTIDE PRO provides the scientific community with:
- Premium research-grade peptides produced under strict quality conditions
- Fast, reliable delivery with same-day dispatch for UK orders before 1pm
- Comprehensive quality documentation including COAs and analytical data
- Professional customer support for research-related inquiries
- Extensive product range including retatrutide and complementary metabolic research peptides
The convergence of metabolic syndrome’s global health impact and retatrutide’s multi-pathway therapeutic potential creates a compelling research landscape. As we advance through 2025, retatrutide metabolic syndrome research will undoubtedly yield new insights into metabolic regulation, therapeutic intervention strategies, and precision medicine approaches.
For researchers committed to advancing metabolic health science, the opportunity to contribute to this growing knowledge base has never been more accessible. With high-quality research peptides, rigorous experimental design, and dedication to scientific excellence, the next generation of metabolic syndrome research will build upon the promising foundation established by early retatrutide investigations.
Ready to advance your metabolic syndrome research? Explore PEPTIDE PRO’s research peptide catalogue to access high-purity retatrutide and complementary compounds. All products are strictly for research use only, supplied with comprehensive quality documentation and professional support.
