
Missing a scheduled dose in any research protocol can create uncertainty and compromise experimental consistency. When working with advanced peptides like retatrutide in laboratory settings, understanding retatrutide missed dose what to do becomes essential for maintaining research integrity and achieving reliable outcomes. This comprehensive guide addresses the critical protocols, timing considerations, and best practices that researchers must follow when dose administration schedules are interrupted.
Retatrutide represents a triple agonist peptide targeting GIP, GLP-1, and glucagon receptors, making it a compound of significant interest in metabolic research. The precision required in research protocols means that even a single missed administration can raise important questions about experimental continuity and data validity. Whether you’re conducting long-term studies or short-term investigations, knowing the proper response to dosing interruptions ensures your research maintains scientific rigor.
Key Takeaways
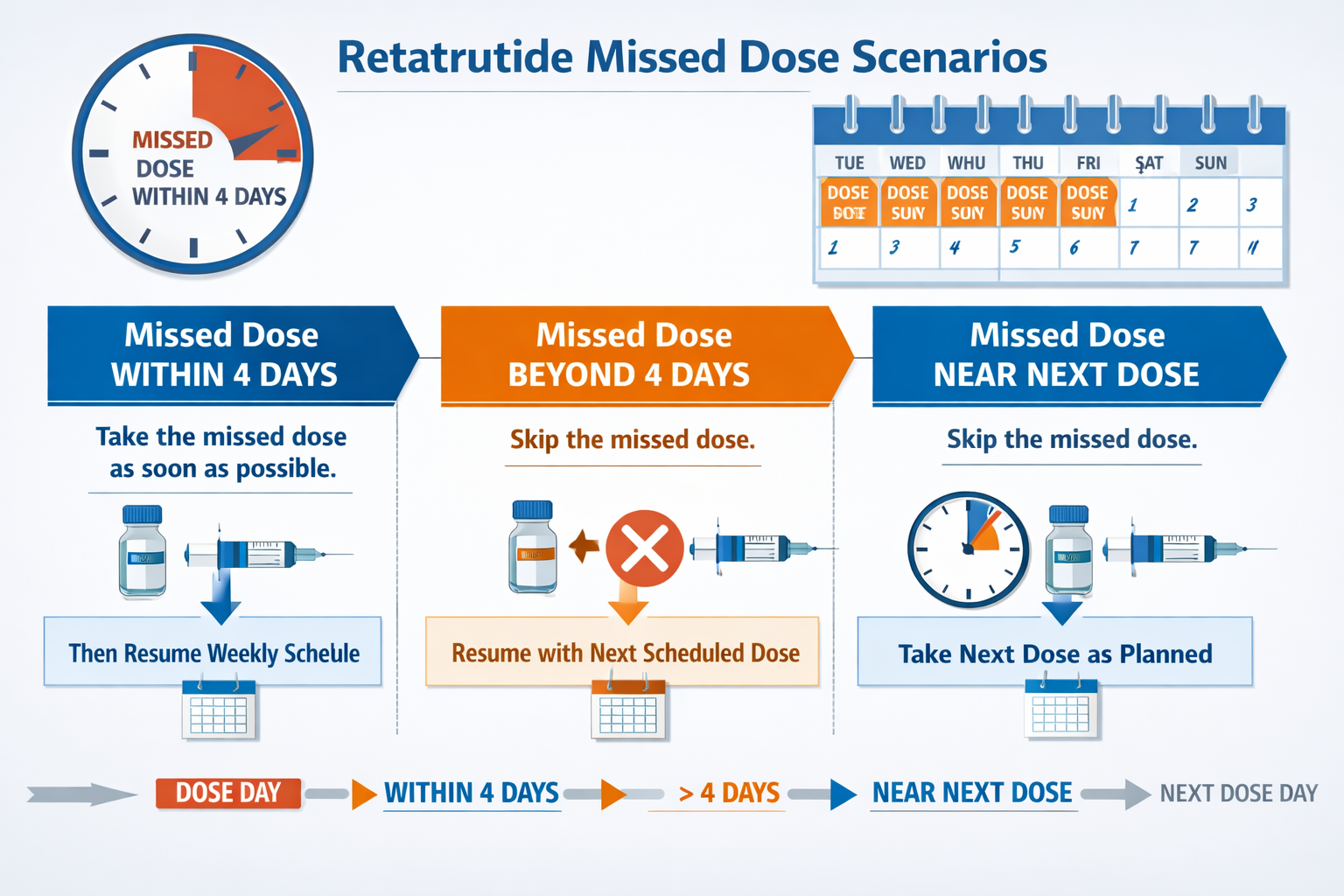
- Timing is critical: If a dose is missed, administration should occur within 4 days of the scheduled time to maintain research protocol consistency
- Skip if too late: When more than 4 days have passed, researchers should skip the missed dose and resume the regular schedule rather than doubling up
- Never double dose: Administering two doses simultaneously compromises research integrity and invalidates experimental parameters
- Document all deviations: Proper research practice requires thorough documentation of any protocol variations, including missed doses
- Storage matters: Proper peptide storage between doses is essential—reconstituted retatrutide must be refrigerated at 2-8°C to maintain stability
Understanding Retatrutide Dosing Schedules in Research
Retatrutide research protocols typically follow weekly administration schedules, though specific timing may vary based on experimental design and research objectives. The peptide’s pharmacokinetic profile, with an extended half-life, allows for this once-weekly dosing pattern, making it convenient for long-term studies while maintaining consistent compound exposure.
Standard Research Dosing Protocols
Research-grade retatrutide is typically administered according to predetermined schedules that align with experimental endpoints and measurement intervals. Most laboratory protocols establish:
- Weekly administration cycles: Fixed day-of-week scheduling to maintain consistency
- Dose escalation phases: Gradual increases following specific timelines
- Maintenance phases: Stable dosing periods for data collection
- Washout considerations: Appropriate intervals between experimental phases
The high-purity peptides used in research settings require meticulous handling and administration timing to ensure data reliability. When working with compounds like Retatrutide 40mg, researchers must establish clear protocols before initiating studies.
Why Dosing Consistency Matters
Maintaining consistent administration schedules serves several critical research functions:
- Steady-state achievement: Regular dosing allows the compound to reach predictable plasma concentrations
- Data comparability: Consistent timing enables valid comparisons across measurement points
- Protocol integrity: Adherence to established schedules maintains experimental validity
- Reproducibility: Standardized timing allows other researchers to replicate findings
Deviations from established schedules can introduce variables that complicate data interpretation and potentially compromise research conclusions.
Retatrutide Missed Dose: What to Do – Immediate Steps
When a scheduled retatrutide administration is missed in a research protocol, researchers must take systematic steps to minimize impact on experimental integrity while ensuring proper documentation and protocol adherence.
The 4-Day Rule for Research Protocols
The primary guideline for addressing retatrutide missed dose what to do centers on a critical 4-day window:
Within 4 Days of Scheduled Dose:
- ✅ Administer the missed dose as soon as the oversight is discovered
- ✅ Resume the regular weekly schedule from the new administration date
- ✅ Document the deviation with exact timing details
- ✅ Note any observable changes in experimental parameters
Beyond 4 Days Past Scheduled Dose:
- ❌ Do NOT administer the missed dose
- ✅ Skip the missed dose entirely
- ✅ Wait for the next regularly scheduled administration
- ✅ Continue with the established protocol timeline
- ✅ Document the gap and monitor for protocol impact
This timing framework reflects the peptide’s pharmacological properties and helps maintain research consistency while preventing compound accumulation that could skew results.
Decision-Making Flowchart
| Time Since Missed Dose | Action Required | Next Steps |
|---|---|---|
| Less than 24 hours | Administer immediately | Continue regular schedule |
| 1-4 days | Administer promptly | Adjust schedule from new date |
| 4-7 days | Skip missed dose | Wait for next scheduled dose |
| More than 7 days | Skip missed dose | Resume regular schedule; consider protocol impact |
Documentation Requirements
Proper research practice demands comprehensive documentation of any protocol deviations:
📋 Essential Documentation Elements:
- Date and time of scheduled dose
- Date and time deviation was discovered
- Actual administration time (if within window)
- Reason for the missed dose
- Any observable effects or changes
- Adjustments made to subsequent scheduling
- Impact assessment on data collection points
This documentation becomes part of the research record and may be critical for data interpretation and publication.
Factors Affecting Retatrutide Missed Dose Decisions
Several variables influence the appropriate response when addressing retatrutide missed dose what to do in research settings. Understanding these factors helps researchers make informed decisions that preserve experimental integrity.
Research Phase Considerations
The stage of your research protocol significantly impacts how missed doses should be handled:
Dose Escalation Phase: Missing a dose during escalation requires careful consideration. Researchers may need to:
- Restart the escalation sequence
- Extend the escalation timeline
- Document the interruption’s impact on tolerance assessment
- Consider whether the gap affects the escalation rationale
Maintenance Phase: During stable dosing periods, a single missed dose may have less impact, but researchers should still:
- Assess whether steady-state concentrations are affected
- Evaluate if the gap creates a mini-washout period
- Consider implications for upcoming measurement timepoints
- Document any changes in experimental parameters
Washout or Transition Phases: Missed doses during these periods may actually align with protocol objectives, but still require documentation.
Compound Stability and Storage
The stability of reconstituted retatrutide directly affects decision-making around missed doses. Research-grade peptides from PEPTIDE PRO maintain optimal stability when stored properly:
🌡️ Storage Requirements:
- Lyophilized (pre-reconstitution): Cool, dry environment; protected from light
- Reconstituted: Refrigerated at 2-8°C in sterile conditions
- Maximum stability: Follow manufacturer guidelines for reconstituted peptide shelf life
- Contamination prevention: Use sterile technique for all handling
If a dose was missed because reconstituted peptide exceeded stability windows, researchers must:
- Discard the compromised solution
- Reconstitute fresh peptide from lyophilized stock
- Document the preparation date
- Adjust protocols to prevent future stability issues
Experimental Timeline Impact
Consider how the missed dose affects critical experimental milestones:
- Measurement timepoints: Does the gap affect scheduled data collection?
- Comparative studies: Will the deviation create inconsistencies with control groups?
- Publication timelines: Could the interruption affect research completion schedules?
- Resource allocation: Does rescheduling impact laboratory resource availability?
These practical considerations help determine whether to continue the current protocol or restart the experimental sequence.
Best Practices for Preventing Missed Doses in Research
Prevention remains the most effective strategy for maintaining research protocol integrity. Implementing systematic approaches minimizes the likelihood of dosing interruptions.
Laboratory Scheduling Systems
Establishing robust scheduling infrastructure creates accountability and reduces oversight:
Digital Calendar Systems:
- Set recurring reminders with 24-hour advance notices
- Create backup alerts on multiple devices
- Share calendars with research team members
- Link dosing schedules to data collection calendars
Physical Documentation:
- Maintain laboratory logbooks with dosing schedules
- Post visible calendars in preparation areas
- Use checklist systems for weekly protocols
- Implement sign-off procedures for completed administrations
Team Communication:
- Designate primary and backup personnel for each dosing day
- Establish clear communication protocols for schedule changes
- Create emergency contact procedures for unexpected absences
- Conduct regular protocol review meetings
Proper Peptide Preparation and Storage
Many missed doses occur due to preparation or storage issues rather than simple oversight. Implementing proper handling protocols prevents these problems:
✨ Preparation Best Practices:
- Reconstitute peptides only when needed for immediate use or short-term protocols
- Use appropriate bacteriostatic water for reconstitution
- Label all pen peptides with reconstitution date and concentration
- Calculate doses in advance to ensure adequate supply
- Maintain backup stock of lyophilized peptide
When working with premium compounds like those available through PEPTIDE PRO’s research peptide catalogue, proper preparation ensures optimal compound integrity throughout your research timeline.
Protocol Design Considerations
Thoughtful experimental design can build flexibility into protocols while maintaining scientific rigor:
Scheduling Flexibility:
- Choose dosing days that align with regular laboratory schedules
- Avoid scheduling during known holiday periods or conference seasons
- Build buffer periods around critical measurement timepoints
- Design protocols with defined windows rather than exact timepoints where scientifically appropriate
Resource Planning:
- Ensure adequate peptide supply before initiating long-term studies
- Verify refrigeration and storage capacity
- Confirm availability of reconstitution supplies
- Establish relationships with reliable suppliers for rapid restocking
Contingency Protocols:
- Develop written procedures for common disruptions
- Establish criteria for protocol continuation vs. restart
- Define acceptable deviation parameters
- Create decision trees for various scenarios
What NOT to Do When You Miss a Retatrutide Dose
Understanding incorrect responses to missed doses is equally important for maintaining research integrity and compound safety.
❌ Never Double Dose
The most critical rule when addressing retatrutide missed dose what to do is never administering a double dose to “make up” for the missed administration. This practice:
- Compromises research validity: Creates unpredictable plasma concentrations
- Introduces uncontrolled variables: Makes data interpretation impossible
- Violates protocol parameters: Deviates from established experimental design
- Risks compound waste: May necessitate protocol restart
Research protocols are designed around specific dosing amounts and intervals. Doubling doses fundamentally alters the experimental conditions and invalidates comparisons with previous data points.
❌ Don’t Ignore Documentation Requirements
Failing to document missed doses and protocol deviations creates serious research integrity issues:
Problems Created by Poor Documentation:
- Data interpretation becomes unreliable
- Reproducibility is compromised
- Publication requirements may not be met
- Research audits may reveal compliance gaps
- Future researchers cannot accurately replicate protocols
Every deviation, regardless of perceived significance, must be thoroughly documented in research records.
❌ Avoid Frequent Schedule Changes
While occasional missed doses may be unavoidable, frequent disruptions suggest systemic protocol issues:
Warning Signs of Protocol Problems:
- Multiple missed doses within a single experimental phase
- Recurring storage or preparation issues
- Inconsistent personnel availability
- Inadequate planning or resource allocation
Frequent disruptions warrant protocol reassessment rather than continued deviation management. Consider whether:
- The dosing schedule is realistic for your laboratory operations
- Adequate resources are allocated to the research
- Team training is sufficient for protocol requirements
- The experimental design needs modification
❌ Don’t Use Compromised Peptides
Never attempt to use peptides that have been improperly stored or have exceeded stability windows:
Peptide Integrity Indicators:
- Visible precipitation or cloudiness in reconstituted solutions
- Discoloration of lyophilized or reconstituted peptide
- Storage outside recommended temperature ranges
- Exceeded manufacturer stability timeframes
- Broken sterility (contamination suspected)
Using compromised peptides introduces uncontrolled variables and potentially invalidates entire experimental sequences. The cost of fresh peptide is minimal compared to the waste of time and resources from invalid data.
Retatrutide Storage and Handling to Minimize Dose Issues

Proper storage and handling practices form the foundation of reliable research protocols and significantly reduce the likelihood of missed doses due to peptide degradation or preparation problems.
Optimal Storage Conditions
Research-grade peptides require specific environmental conditions to maintain stability and activity:
Lyophilized (Unreconstituted) Peptide:
🌡️ Temperature: Store at -20°C to -80°C for long-term storage, or 2-8°C for short-term (up to 3 months)
- 💧 Humidity: Keep in dry environment; desiccants recommended
- ☀️ Light exposure: Protect from direct light; use amber pen peptides or foil wrapping
- 📦 Packaging: Maintain in original sealed pen peptides until ready for use
Reconstituted Peptide:
🌡️ Temperature: Refrigerate at 2-8°C consistently
- ⏱️ Duration: Use within manufacturer-specified timeframe (typically 7-14 days)
- 🧪 Sterility: Maintain aseptic conditions; use sterile technique for all withdrawals
- 🏷️ Labeling: Clearly mark reconstitution date and concentration
The high-purity research peptides supplied by reputable providers include detailed storage specifications that must be followed precisely.
Reconstitution Protocols
Proper reconstitution technique ensures peptide stability and accurate dosing:
Step-by-Step Reconstitution:
- Preparation
- Allow lyophilized peptide to reach room temperature
- Gather sterile bacteriostatic water
- Prepare sterile workspace
- Calculate required concentration
- Reconstitution Process
- Add bacteriostatic water slowly down the pen peptide side
- Avoid direct stream onto lyophilized peptide
- Gently swirl (never shake vigorously)
- Allow complete dissolution before use
- Inspect for clarity and absence of particulates
- Post-Reconstitution
- Label pen peptide with date, time, and concentration
- Store immediately at 2-8°C
- Document in laboratory records
- Plan usage schedule to ensure use within stability window
Quality Assurance Measures
Implementing quality control procedures ensures peptide integrity throughout research protocols:
Regular Checks:
- ✅ Daily temperature monitoring of storage equipment
- ✅ Weekly inventory of peptide supplies
- ✅ Monthly equipment calibration verification
- ✅ Quarterly protocol compliance audits
Documentation Systems:
- Maintain chain of custody records for all peptides
- Log all reconstitution activities
- Track usage patterns to optimize ordering
- Record any deviations or unusual observations
Supplier Relationships: Establishing relationships with reliable suppliers like PEPTIDE PRO ensures:
- Consistent product quality with Certificates of Analysis
- Rapid restocking when supplies run low
- Technical support for handling questions
- Reliable delivery schedules for protocol planning
Long-Term Research Protocol Considerations
Extended research protocols involving retatrutide require additional planning and flexibility to accommodate the realities of long-term experimental work.
Managing Extended Research Timelines
Multi-month or multi-year research projects face unique challenges regarding dosing consistency:
Planning Strategies:
📅 Timeline Mapping
- Identify potential conflict periods (holidays, conferences, facility closures)
- Build flexibility into non-critical phases
- Establish clear criteria for acceptable deviations
- Plan measurement timepoints with buffer windows
🔄 Protocol Adaptability
- Design protocols with defined acceptable deviation ranges
- Establish decision criteria for continuation vs. restart
- Create tiered response plans for various disruption scenarios
- Document all adaptations with scientific justification
👥 Team Continuity
- Cross-train multiple team members on all protocols
- Develop comprehensive standard operating procedures
- Establish clear succession plans for personnel changes
- Maintain institutional knowledge through documentation
Data Integrity with Protocol Deviations
When missed doses occur in long-term studies, researchers must carefully consider data integrity implications:
Impact Assessment Framework:
- Immediate Impact
- How does the gap affect current experimental phase?
- Are upcoming measurement timepoints compromised?
- Does the deviation affect steady-state assumptions?
- Cumulative Impact
- Is this an isolated incident or part of a pattern?
- How do multiple deviations affect overall data quality?
- Can statistical methods account for the irregularity?
- Publication Implications
- Will reviewers question data validity?
- Can the deviation be adequately explained?
- Does the gap affect primary outcome measures?
Statistical Considerations:
- Consult with biostatisticians about deviation impact
- Consider sensitivity analyses excluding affected data points
- Document all deviations in methods sections
- Be transparent about limitations in discussion sections
Coordinating with Other Research Activities
Retatrutide protocols often run concurrently with other research activities, requiring careful coordination:
Integration Strategies:
- Synchronize dosing schedules with measurement protocols
- Coordinate with other laboratory activities using shared resources
- Plan around facility maintenance and shutdown periods
- Align with funding cycles and reporting requirements
Frequently Asked Questions About Retatrutide Missed Doses
How long can retatrutide stay at room temperature?
Lyophilized retatrutide can briefly tolerate room temperature during handling and preparation, but should be returned to proper storage quickly. Reconstituted peptide should not remain at room temperature for more than 30 minutes during preparation and administration. Extended room temperature exposure degrades peptide stability and compromises research results.
Can I restart my research protocol after multiple missed doses?
Multiple missed doses typically warrant protocol reassessment rather than simple continuation. Researchers should evaluate whether the experimental design remains valid, consider whether steady-state conditions have been lost, and determine if restarting with fresh baseline measurements would provide more reliable data. Document the decision-making process thoroughly.
What if I discover a missed dose several weeks later?
If a missed dose is discovered weeks after the scheduled administration, simply continue with the current protocol schedule. Do not attempt to “catch up” or adjust dosing. Document the discovery and the missed dose in research records, assess potential impact on data interpretation, and consider whether the gap affects experimental conclusions.
Does retatrutide require special disposal procedures?
Yes, research peptides require proper disposal following institutional biosafety and chemical waste protocols. Never dispose of peptides in regular trash or down drains. Unused or expired peptides should be treated as chemical waste, following your institution’s hazardous waste disposal procedures and local regulations.
How do I know if my peptide has degraded?
Signs of peptide degradation include visible changes (discoloration, precipitation, cloudiness), exceeded storage timeframes, improper storage temperature exposure, or broken sterility. When in doubt, discard the peptide and reconstitute fresh material. The cost of replacement is minimal compared to the risk of invalid data from degraded compounds.
Should I adjust my research timeline after a missed dose?
Timeline adjustments depend on the experimental design and the timing of the missed dose. If the gap occurs during critical measurement periods or affects steady-state assumptions, timeline adjustment may be necessary. For isolated missed doses during maintenance phases with flexible measurement windows, continuation without adjustment may be appropriate. Document the decision rationale thoroughly.
Maintaining Research Excellence with Retatrutide
Understanding retatrutide missed dose what to do represents a crucial component of research protocol management. While missed doses can occur despite best planning efforts, systematic approaches to prevention, response, and documentation ensure that research integrity remains intact and experimental data maintains validity.
The key principles for managing missed retatrutide doses in research settings include:
Immediate Response: Apply the 4-day rule consistently—administer within the window or skip and continue with regular scheduling. Never double dose or make unauthorized protocol modifications.
📊 Documentation Excellence: Maintain comprehensive records of all protocol deviations, including timing, circumstances, and impact assessments. This documentation protects research integrity and enables transparent reporting.
🔬 Prevention Focus: Implement robust scheduling systems, proper storage protocols, and team coordination strategies to minimize missed doses before they occur. Prevention is always preferable to remediation.
⚖️ Quality Assurance: Establish relationships with reputable suppliers, maintain proper storage conditions, and implement regular quality checks to ensure peptide integrity throughout research timelines.
Next Steps for Researchers
For researchers working with retatrutide in laboratory settings:
- Review and update your current dosing protocols to incorporate the guidelines outlined in this article
- Implement systematic scheduling and documentation procedures if not already in place
- Ensure adequate supply of high-purity research peptides through reliable sources
- Train all team members on proper handling, storage, and protocol deviation procedures
- Establish clear criteria for acceptable deviations and protocol continuation decisions
Access Premium Research Peptides
Maintaining research excellence begins with access to high-quality, research-grade compounds. PEPTIDE PRO provides researchers with premium peptides backed by rigorous quality control, comprehensive documentation, and professional support services.
Why Researchers Choose PEPTIDE PRO:
- ✅ Exceptional purity standards with full Certificates of Analysis
- ✅ Fast UK delivery with same-day dispatch for orders before 1pm
- ✅ Extensive peptide catalogue for diverse research applications
- ✅ Professional customer support for technical questions
- ✅ Proper labeling and storage guidance for all products
Whether you’re initiating new research protocols or maintaining ongoing studies, establishing relationships with reliable peptide suppliers ensures uninterrupted access to the compounds you need, when you need them.
Ready to ensure your research maintains the highest standards? Explore PEPTIDE PRO’s research peptide collection to find the high-purity compounds your laboratory requires, backed by the quality assurance and professional service that research excellence demands.
Disclaimer: This article provides information for research purposes only. All peptides discussed are intended strictly for laboratory research use and are not for human or animal consumption. Researchers must comply with all applicable regulations, institutional guidelines, and ethical standards when conducting peptide research. Always consult with qualified research supervisors and follow established laboratory protocols.
