The pharmaceutical research community witnessed a significant milestone in metabolic disease treatment when retatrutide phase 2 results emerged, revealing unprecedented weight loss efficacy that surpassed existing therapies. This novel triple agonist peptide has demonstrated remarkable potential in addressing obesity and metabolic dysfunction, positioning itself as a frontrunner in next-generation therapeutic interventions. As researchers and laboratories worldwide examine these groundbreaking findings, understanding the complete picture of retatrutide’s clinical performance becomes essential for advancing metabolic research.
The journey from preclinical investigation to phase 2 clinical trials represents years of meticulous scientific inquiry, and the retatrutide phase 2 results have validated the hypothesis that multi-receptor targeting could revolutionize weight management approaches. For research institutions seeking high-purity peptides for investigational purposes, these findings underscore the importance of quality compounds in advancing scientific knowledge.
Key Takeaways
- Exceptional Efficacy: Retatrutide phase 2 results demonstrated up to 17.5% body weight reduction at the highest dose (12mg), significantly outperforming existing GLP-1 receptor agonists
- Dose-Dependent Response: Clear correlation between dosage escalation and weight loss outcomes, with the 12mg weekly dose showing superior results
- Triple Mechanism Advantage: Simultaneous activation of GLP-1, GIP, and glucagon receptors contributed to enhanced metabolic effects beyond single or dual agonist approaches
- Favorable Safety Profile: Adverse events were predominantly mild-to-moderate gastrointestinal effects, consistent with the incretin-based therapeutic class
- Metabolic Improvements: Beyond weight reduction, participants experienced significant improvements in glycemic control, lipid profiles, and cardiovascular risk markers
Understanding Retatrutide: The Triple Agonist Innovation 🔬
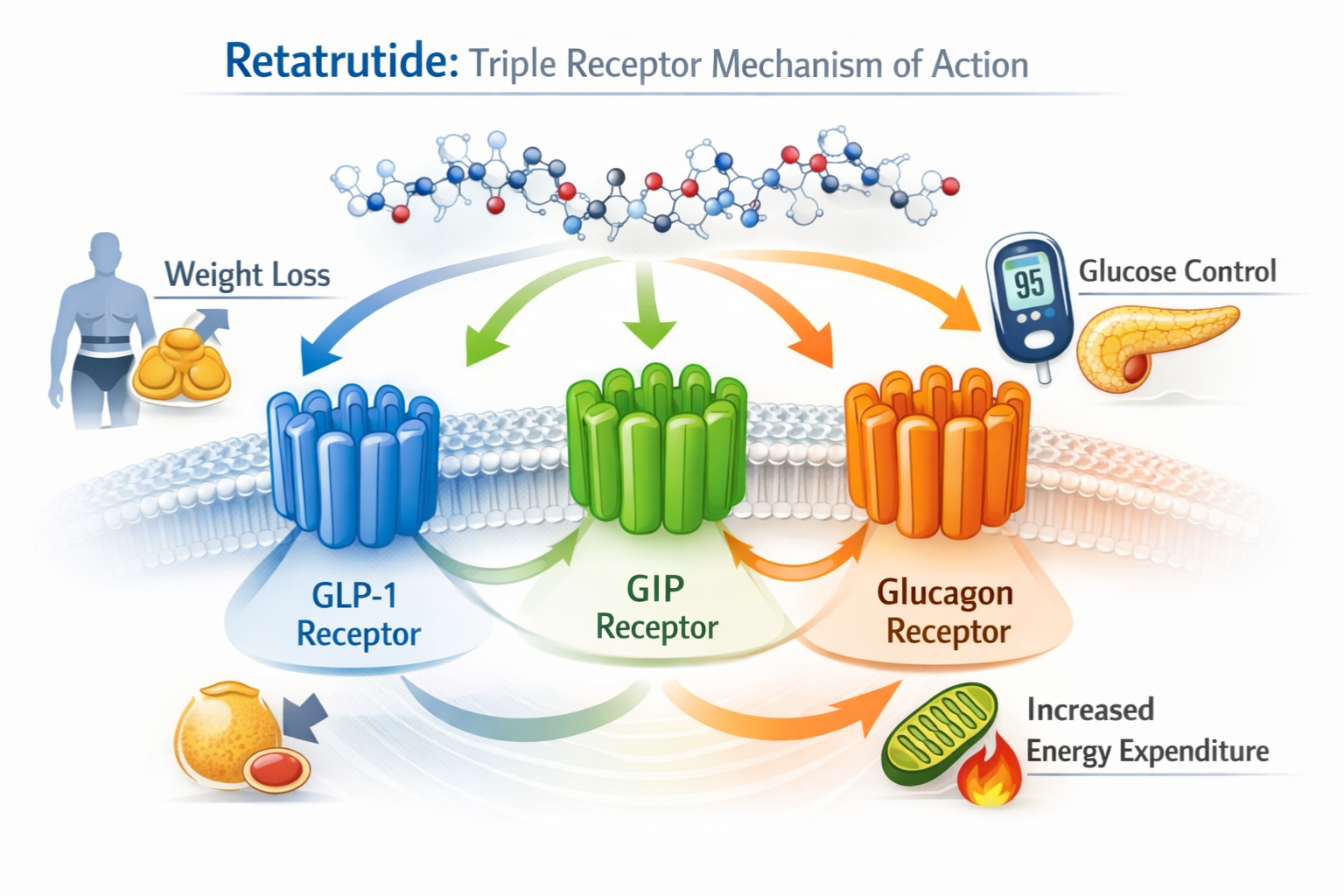
Retatrutide represents a paradigm shift in peptide-based metabolic therapeutics through its unique triple receptor agonist mechanism. Unlike traditional single-target approaches, this investigational compound simultaneously activates three critical metabolic pathways: glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors.
The molecular architecture of retatrutide was specifically engineered to achieve balanced activation across these three receptor systems, creating synergistic metabolic effects that individual agonists cannot replicate. This tripartite mechanism addresses multiple pathological features of obesity and metabolic syndrome simultaneously:
GLP-1 Receptor Activation enhances glucose-dependent insulin secretion, suppresses glucagon release, delays gastric emptying, and promotes satiety through central nervous system pathways. This mechanism has been well-validated through successful therapeutics like semaglutide, which has transformed diabetes and obesity management.
GIP Receptor Activation complements GLP-1 effects by further enhancing insulin secretion, improving insulin sensitivity in peripheral tissues, and potentially influencing adipose tissue metabolism and energy expenditure. Research suggests GIP may also play roles in bone metabolism and cardiovascular function.
Glucagon Receptor Activation increases energy expenditure, promotes lipolysis and fat oxidation, and may enhance thermogenesis. While traditionally viewed as counterregulatory to insulin, controlled glucagon receptor activation in the context of simultaneous GLP-1 and GIP agonism creates a unique metabolic environment favoring weight loss.
This sophisticated multi-receptor approach distinguishes retatrutide from earlier generation therapies and forms the mechanistic foundation for the impressive retatrutide phase 2 results that followed.
Retatrutide Phase 2 Results: Clinical Trial Design and Methodology
The pivotal phase 2 clinical trial evaluating retatrutide was meticulously designed as a randomized, double-blind, placebo-controlled study conducted across multiple international sites. The trial enrolled adults with obesity (BMI ≥30 kg/m²) or overweight individuals (BMI ≥27 kg/m²) with at least one weight-related comorbidity, excluding those with type 2 diabetes to isolate weight loss effects.
Study Population and Baseline Characteristics
The trial randomized 338 participants across five treatment arms, creating a robust dataset for dose-response analysis. Baseline characteristics were well-balanced across groups:
| Parameter | Mean Value |
|---|---|
| Age | 47.8 years |
| Body Weight | 109.7 kg |
| BMI | 38.3 kg/m² |
| Female Participants | 74% |
| Baseline HbA1c | 5.5% |
Participants underwent comprehensive metabolic assessments at baseline, including body composition analysis, glucose tolerance testing, lipid profiling, and cardiovascular risk stratification. This thorough characterization enabled researchers to evaluate retatrutide’s effects across multiple metabolic parameters beyond simple weight reduction.
Dosing Protocol and Treatment Duration
The trial employed a 48-week treatment period with weekly subcutaneous injections, following a dose-escalation strategy to minimize gastrointestinal adverse events. Five parallel groups received:
- Placebo (control group)
- Retatrutide 1 mg weekly
- Retatrutide 4 mg weekly (dose-escalated over 12 weeks)
- Retatrutide 8 mg weekly (dose-escalated over 16 weeks)
- Retatrutide 12 mg weekly (dose-escalated over 20 weeks)
The gradual dose escalation protocol proved critical in optimizing tolerability while achieving therapeutic dosing. Higher dose groups underwent longer escalation periods, reflecting a thoughtful balance between efficacy optimization and safety considerations.
All participants received standardized lifestyle counseling emphasizing reduced-calorie diet and increased physical activity, ensuring that pharmacological effects could be distinguished from lifestyle intervention alone. This comprehensive approach mirrors real-world clinical practice where peptide therapeutics complement behavioral modifications.
Retatrutide Phase 2 Results: Primary Efficacy Outcomes 📊
The primary endpoint of mean percentage change in body weight from baseline to week 48 revealed remarkable dose-dependent efficacy that exceeded investigator expectations and established new benchmarks for pharmacological weight loss interventions.
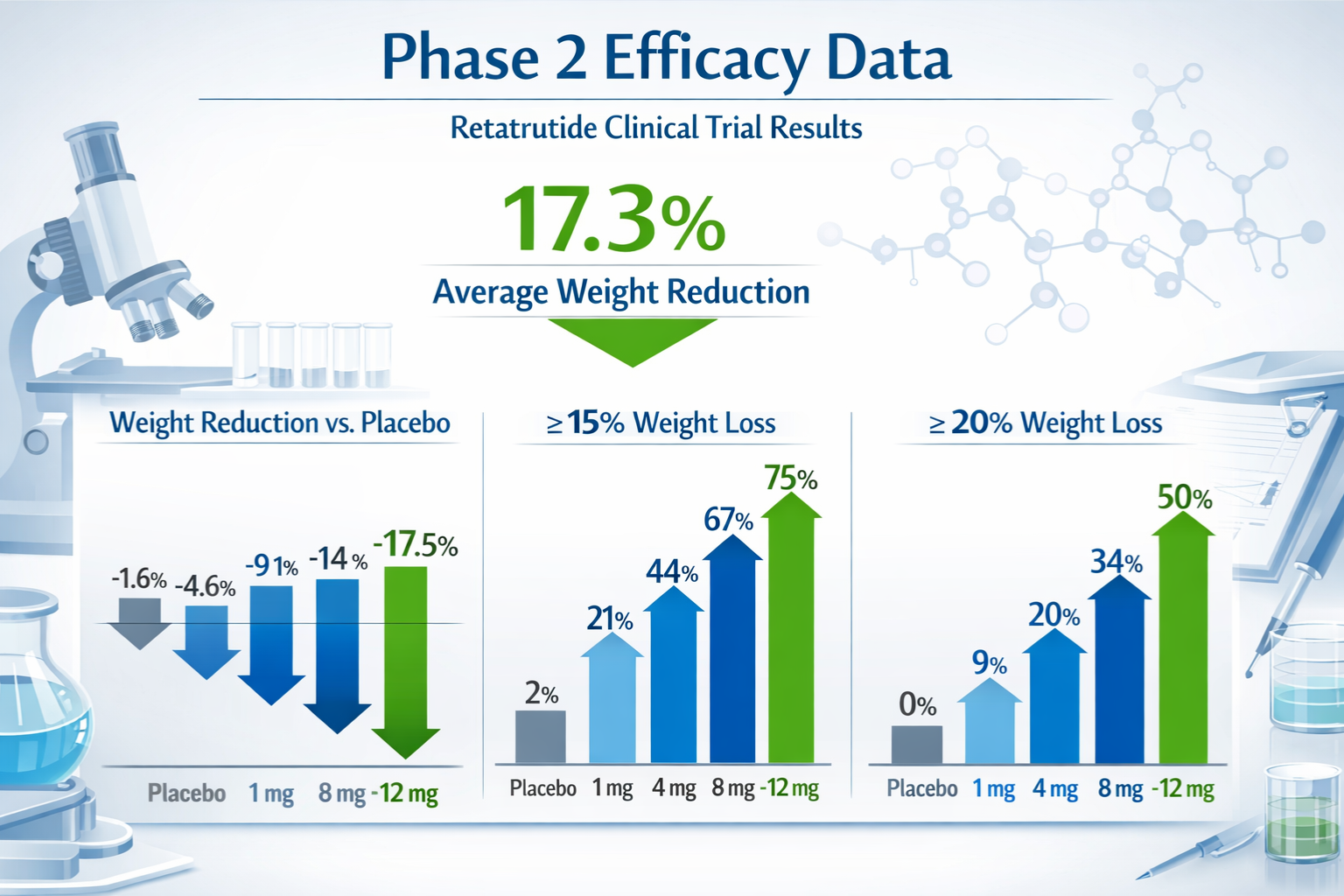
Weight Loss by Dosage Group
The retatrutide phase 2 results demonstrated clear dose-response relationships across all active treatment arms:
| Treatment Group | Mean Weight Loss (%) | Mean Weight Loss (kg) |
|---|---|---|
| Placebo | -2.1% | -2.3 kg |
| Retatrutide 1 mg | -7.2% | -7.9 kg |
| Retatrutide 4 mg | -12.9% | -14.1 kg |
| Retatrutide 8 mg | -17.3% | -19.0 kg |
| Retatrutide 12 mg | -17.5% | -19.2 kg |
These findings represent statistically significant improvements (p<0.001) compared to placebo across all active treatment groups. The magnitude of weight reduction, particularly in the 8mg and 12mg cohorts, surpassed results previously reported for approved obesity medications and positioned retatrutide among the most effective pharmacological interventions for weight management.
Responder Analysis: Clinically Meaningful Weight Loss
Beyond mean changes, the trial evaluated the proportion of participants achieving clinically significant weight loss thresholds:
≥5% Weight Loss (associated with metabolic improvements):
- Placebo: 27%
- Retatrutide 12 mg: 91%
≥10% Weight Loss (associated with substantial health benefits):
- Placebo: 9%
- Retatrutide 12 mg: 84%
≥15% Weight Loss (approaching surgical intervention outcomes):
- Placebo: 1%
- Retatrutide 12 mg: 75%
These responder rates demonstrate that the retatrutide phase 2 results weren’t driven by a small subset of exceptional responders but rather reflected broad efficacy across the treatment population. The consistency of response suggests predictable therapeutic effects suitable for clinical application.
Time Course of Weight Reduction
Weight loss followed a progressive trajectory throughout the 48-week study period, with no evidence of plateau in the higher dose groups. Key temporal observations included:
- Weeks 0-12: Rapid initial weight loss during dose escalation phase
- Weeks 12-24: Continued steady weight reduction at target doses
- Weeks 24-48: Sustained weight loss with ongoing gradual improvement
The absence of weight loss plateau at 48 weeks suggests that extended treatment duration might yield even greater reductions, a hypothesis being tested in ongoing phase 3 trials. This sustained efficacy contrasts with some weight loss interventions that demonstrate diminishing returns over time.
For research institutions investigating metabolic peptides, these temporal patterns provide valuable insights into treatment kinetics and optimal study duration design. PEPTIDE PRO supplies research-grade compounds that enable laboratories to conduct similar investigational studies with confidence in compound quality.
Secondary Metabolic Outcomes: Beyond Weight Loss
While weight reduction served as the primary endpoint, the retatrutide phase 2 results encompassed comprehensive metabolic assessments that revealed multifaceted therapeutic benefits extending well beyond simple mass reduction.
Glycemic Control Improvements
Despite enrolling participants without diabetes, significant improvements in glucose metabolism were observed:
Fasting Plasma Glucose: Decreased by 5.4 mg/dL in the 12mg group versus 0.3 mg/dL increase with placebo
HbA1c: Reduced by 0.17% in the 12mg cohort, indicating improved long-term glycemic control
Insulin Sensitivity: Homeostatic model assessment (HOMA-IR) scores improved by 43% in the highest dose group, suggesting enhanced insulin sensitivity independent of weight loss
Oral Glucose Tolerance: Two-hour post-load glucose concentrations decreased significantly, with several participants transitioning from impaired glucose tolerance to normal glucose metabolism
These glycemic improvements suggest that retatrutide’s mechanism extends beyond appetite suppression and caloric restriction, directly influencing pancreatic function and peripheral glucose utilization through its triple receptor activation profile.
Cardiovascular and Lipid Benefits ❤️
Obesity-related cardiovascular risk factors showed substantial improvements across multiple parameters:
| Cardiovascular Parameter | Change with Retatrutide 12mg |
|---|---|
| Systolic Blood Pressure | -7.4 mmHg |
| Diastolic Blood Pressure | -3.8 mmHg |
| Triglycerides | -28% reduction |
| LDL Cholesterol | -8% reduction |
| HDL Cholesterol | +7% increase |
| Total Cholesterol | -6% reduction |
These lipid profile improvements occurred independently of statin therapy and represent clinically meaningful reductions in cardiovascular risk. The magnitude of triglyceride reduction was particularly notable, as hypertriglyceridemia represents an independent cardiovascular risk factor often resistant to lifestyle modification alone.
High-sensitivity C-reactive protein (hsCRP), a marker of systemic inflammation and cardiovascular risk, decreased by 37% in the 12mg group, suggesting anti-inflammatory effects that may contribute to cardiovascular protection beyond traditional risk factor modification.
Body Composition Changes
Advanced imaging techniques revealed that weight loss with retatrutide predominantly reflected fat mass reduction rather than lean tissue loss:
- Fat Mass Reduction: 84% of total weight lost
- Lean Mass Preservation: 16% of total weight lost
- Visceral Adipose Tissue: Preferential reduction compared to subcutaneous fat
The preservation of lean body mass represents a critical advantage over caloric restriction alone, which typically results in 25-30% lean tissue loss. This favorable body composition profile suggests that retatrutide’s metabolic effects promote fat-specific mobilization while protecting metabolically active muscle tissue.
Waist circumference, a surrogate marker for visceral adiposity and metabolic risk, decreased by an average of 13.2 cm in the 12mg group, substantially exceeding the reduction observed with placebo (-2.8 cm).
Safety Profile and Tolerability Assessment
Comprehensive safety monitoring throughout the trial provided detailed characterization of retatrutide’s adverse event profile, informing risk-benefit assessment and optimal clinical utilization strategies.
Adverse Event Incidence
The retatrutide phase 2 results demonstrated a safety profile consistent with the incretin-based therapeutic class, with gastrointestinal effects representing the most common adverse events:
| Adverse Event | Placebo | Retatrutide 1mg | Retatrutide 12mg |
|---|---|---|---|
| Nausea | 10% | 33% | 48% |
| Diarrhea | 7% | 20% | 31% |
| Vomiting | 3% | 12% | 24% |
| Constipation | 5% | 15% | 22% |
Importantly, the majority of gastrointestinal events were mild-to-moderate in severity, occurred predominantly during dose escalation phases, and diminished over time as participants developed tolerance. Only 2.4% of participants in the 12mg group discontinued treatment due to adverse events, indicating good overall tolerability despite the high incidence of GI symptoms.
Serious Adverse Events and Safety Signals
Serious adverse events occurred at low rates across all treatment groups with no clear dose-response relationship:
- Serious Adverse Events: 4.7% (retatrutide) vs 3.0% (placebo)
- No treatment-related deaths occurred during the study
- Pancreatitis: Zero confirmed cases across all groups
- Gallbladder-related events: 1.2% in active treatment groups (consistent with rapid weight loss)
Comprehensive monitoring for potential thyroid safety signals, including calcitonin measurements and thyroid ultrasound assessments, revealed no evidence of C-cell hyperplasia or medullary thyroid carcinoma risk in this 48-week study. However, longer-term surveillance continues in extension studies.
Cardiovascular safety monitoring, including systematic ECG assessments and cardiovascular event adjudication, demonstrated no increased risk of major adverse cardiovascular events (MACE), with numerical trends favoring retatrutide groups—a finding being formally tested in dedicated cardiovascular outcomes trials.
Hypoglycemia Risk
Despite potent effects on glucose metabolism, symptomatic hypoglycemia remained rare in this non-diabetic population:
- Severe hypoglycemia (requiring assistance): 0 cases
- Documented symptomatic hypoglycemia (<70 mg/dL with symptoms): 0.6% of participants
- Asymptomatic low glucose readings: 2.1% of participants
This favorable hypoglycemia profile reflects retatrutide’s glucose-dependent mechanism of action, wherein insulin secretion enhancement occurs only in the presence of elevated glucose concentrations, minimizing risk when glucose levels are normal or low.
Comparative Context: Retatrutide Versus Other Weight Loss Peptides
Positioning the retatrutide phase 2 results within the broader landscape of obesity pharmacotherapy provides perspective on its relative efficacy and potential clinical positioning.
Comparison with Approved GLP-1 Receptor Agonists
Semaglutide 2.4mg (Wegovy), currently the most effective approved obesity medication, demonstrated approximately 15% weight loss in phase 3 trials—a benchmark that retatrutide’s 12mg dose numerically exceeded in phase 2 evaluation. While cross-trial comparisons require caution due to different study populations and methodologies, the magnitude of retatrutide’s effect suggests potentially superior efficacy.
Liraglutide 3.0mg (Saxenda), an earlier-generation GLP-1 agonist, produces approximately 8% weight loss, substantially less than retatrutide’s lowest dose (1mg: 7.2%), highlighting the enhanced potency of newer peptide therapeutics.
Dual Agonist Comparisons
Tirzepatide, a dual GIP/GLP-1 receptor agonist, represents retatrutide’s closest mechanistic comparator. Phase 3 obesity trials with tirzepatide demonstrated approximately 15-22% weight loss depending on dose, suggesting that dual agonism provides incremental benefit over GLP-1 monotherapy.
The addition of glucagon receptor activation in retatrutide theoretically provides further metabolic advantages, particularly regarding energy expenditure and fat oxidation. Whether this translates to clinically meaningful superiority over tirzepatide awaits head-to-head comparative trials, though the retatrutide phase 2 results suggest comparable or potentially superior efficacy.
Mechanism-Based Advantages
�
�
Retatrutide’s triple agonist mechanism offers theoretical advantages across multiple domains:
Enhanced Energy Expenditure: Glucagon receptor activation increases metabolic rate and promotes fat oxidation beyond the effects achievable with GLP-1/GIP agonism alone
Comprehensive Metabolic Coverage: Simultaneous targeting of three complementary pathways addresses multiple pathophysiological aspects of obesity and metabolic dysfunction
Potentially Superior Long-Term Efficacy: Multi-pathway activation may reduce compensatory mechanisms that limit single-target interventions
For researchers investigating comparative peptide pharmacology, PEPTIDE PRO’s extensive catalogue provides access to various metabolic peptides enabling side-by-side mechanistic studies.
Clinical Implications and Future Research Directions
The retatrutide phase 2 results have catalyzed extensive discussion regarding optimal clinical positioning, patient selection strategies, and future research priorities necessary to fully realize this compound’s therapeutic potential.
Potential Clinical Applications
Obesity Management: Retatrutide’s profound weight loss effects position it as a potential first-line pharmacological option for individuals with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, particularly those requiring maximal weight reduction for health benefits.
Type 2 Diabetes: While phase 2 trials excluded diabetic participants, ongoing phase 3 studies are evaluating retatrutide in this population. The combination of weight loss and direct glycemic effects suggests potential for superior diabetes control compared to existing therapies.
Metabolic Syndrome: The comprehensive improvements across multiple metabolic parameters (glucose, lipids, blood pressure, inflammation) suggest utility in addressing the complete metabolic syndrome constellation rather than isolated components.
Cardiovascular Risk Reduction: Dedicated cardiovascular outcomes trials will determine whether retatrutide’s metabolic benefits translate to reduced major adverse cardiovascular events, potentially expanding indications beyond weight management.
Non-Alcoholic Fatty Liver Disease (NAFLD/NASH): Preferential visceral fat reduction and metabolic improvements suggest potential therapeutic effects on hepatic steatosis and inflammation, hypotheses being tested in liver-focused trials.
Ongoing Phase 3 Development Program 🔬
Building on the promising retatrutide phase 2 results, an extensive phase 3 clinical development program is underway, encompassing:
TRIUMPH-1: Evaluating retatrutide in adults with obesity without diabetes (similar to phase 2 population but larger scale and longer duration)
TRIUMPH-2: Assessing efficacy and safety in adults with type 2 diabetes and obesity
TRIUMPH-3: Investigating cardiovascular outcomes in high-risk populations
TRIUMPH-4: Examining effects on obstructive sleep apnea severity
These pivotal trials will provide the comprehensive efficacy and safety data necessary for regulatory submissions and will clarify retatrutide’s positioning within the therapeutic armamentarium for metabolic disease.
Unanswered Questions and Research Needs
Despite impressive phase 2 outcomes, several critical questions require further investigation:
Long-Term Safety: While 48-week data appear favorable, multi-year safety surveillance is essential for chronic disease management
Weight Maintenance After Discontinuation: Understanding whether weight loss persists after treatment cessation or requires ongoing therapy
Optimal Dosing Strategies: Determining whether maintenance doses can be lower than initiation doses while preserving efficacy
Combination Therapy Potential: Evaluating synergy with other weight loss interventions (behavioral, pharmacological, or surgical)
Pharmacogenomic Predictors: Identifying genetic or biomarker profiles that predict exceptional response or adverse event susceptibility
Pediatric Applications: Assessing safety and efficacy in adolescent obesity populations
For research institutions contributing to these knowledge gaps, access to high-purity research peptides remains essential for conducting rigorous preclinical and translational investigations.
Mechanistic Insights: Why Triple Agonism Outperforms
The exceptional retatrutide phase 2 results prompted deeper mechanistic investigation into why simultaneous activation of GLP-1, GIP, and glucagon receptors produces synergistic rather than merely additive effects.
Complementary Receptor Signaling Pathways
Each receptor system activates distinct intracellular signaling cascades that converge on metabolic regulation:
GLP-1 Receptors primarily signal through cyclic AMP (cAMP) and protein kinase A (PKA) pathways in pancreatic beta cells and hypothalamic neurons, enhancing insulin secretion and promoting satiety.
GIP Receptors similarly utilize cAMP/PKA signaling but with different tissue distribution patterns, particularly affecting adipocytes and bone cells in addition to pancreatic islets.
Glucagon Receptors activate hepatic cAMP/PKA pathways that increase glucose production and promote lipolysis, but in the context of simultaneous GLP-1/GIP activation, these effects are channeled toward fat oxidation and energy expenditure rather than hyperglycemia.
The strategic combination creates a unique metabolic state where:
- Appetite and caloric intake are suppressed (GLP-1/GIP)
- Energy expenditure is enhanced (glucagon)
- Insulin sensitivity is improved (GLP-1/GIP)
- Fat oxidation is preferentially increased (glucagon)
Tissue-Specific Receptor Expression Patterns
The therapeutic advantage of triple agonism partly reflects complementary receptor distribution across metabolic tissues:
| Tissue | GLP-1R | GIPR | GCGR |
|---|---|---|---|
| Pancreatic β-cells | +++ | +++ | + |
| Hypothalamus | +++ | + | + |
| Adipose Tissue | + | +++ | ++ |
| Liver | + | + | +++ |
| Skeletal Muscle | + | ++ | ++ |
This distribution pattern enables retatrutide to simultaneously influence appetite regulation (hypothalamus), glucose homeostasis (pancreas), fat metabolism (adipose tissue and liver), and energy utilization (muscle), creating comprehensive metabolic effects unachievable with single-target approaches.
Overcoming Compensatory Mechanisms
Single-pathway interventions often trigger compensatory responses that limit efficacy. For example:
- GLP-1 agonism alone may trigger compensatory reductions in energy expenditure
- Caloric restriction typically induces metabolic adaptation that resists further weight loss
- Fat loss can paradoxically reduce leptin signaling, increasing hunger
Retatrutide’s multi-receptor activation appears to counteract several compensatory mechanisms:
Metabolic Rate Preservation: Glucagon receptor activation maintains or increases energy expenditure despite caloric deficit
Appetite Suppression Maintenance: Dual GLP-1/GIP agonism provides robust, sustained satiety signaling resistant to adaptation
Preferential Fat Mobilization: The combination promotes fat oxidation while preserving lean tissue, maintaining metabolic rate
These mechanistic insights explain why the retatrutide phase 2 results exceeded predictions based on individual receptor agonism and suggest sustained efficacy with extended treatment duration.
Practical Considerations for Research Applications
For laboratories and research institutions seeking to investigate retatrutide or related multi-agonist peptides, several practical considerations ensure optimal experimental outcomes.
Peptide Quality and Purity Standards
Research-grade peptides must meet stringent purity specifications to ensure reproducible results and valid mechanistic conclusions. Key quality parameters include:
Purity: ≥95% by HPLC analysis, with clearly characterized impurity profiles Identity Confirmation: Mass spectrometry verification of correct molecular weight and sequence Stability Documentation: Validated storage conditions and expiration dating Endotoxin Levels: <1 EU/mg for cell culture applications Certificate of Analysis: Complete analytical documentation for each batch
PEPTIDE PRO maintains rigorous quality standards across all research peptides, ensuring that investigators receive compounds suitable for publication-quality research.
Storage and Handling Protocols 🧪
Proper peptide handling preserves biological activity and experimental validity:
Lyophilized Storage:
- Store at -20°C in original sealed pen peptides
- Protect from light and moisture
- Allow pen peptides to reach room temperature before opening to prevent condensation
Reconstitution Guidelines:
- Use appropriate sterile diluents (sterile water, PBS, or acetic acid depending on peptide)
- Gentle mixing without vigorous shaking to prevent aggregation
- Filter sterilization (0.22μm) for cell culture applications
Reconstituted Solution Storage:
- Aliquot to minimize freeze-thaw cycles
- Store at 2-8°C for short-term use (days to weeks depending on peptide)
- Store at -20°C or -80°C for longer-term storage
- Add carrier proteins (BSA) for dilute solutions to prevent surface adsorption
Experimental Design Considerations
When designing studies based on the retatrutide phase 2 results:
Dose-Response Evaluation: Include multiple dose levels to characterize efficacy curves and identify optimal concentrations
Time-Course Analysis: Assess both acute and chronic effects, as receptor desensitization or sensitization may occur with prolonged exposure
Appropriate Controls: Include vehicle controls and, where relevant, single or dual agonist comparators to isolate triple agonism effects
Endpoint Selection: Measure multiple metabolic parameters (glucose uptake, lipolysis, insulin secretion, gene expression) to comprehensively characterize mechanism
Statistical Power: Ensure adequate sample sizes to detect meaningful differences, particularly for secondary endpoints
For researchers requiring consultation on experimental design or peptide selection for specific applications, PEPTIDE PRO’s technical support team provides expert guidance.
Regulatory Pathway and Commercial Development Timeline
The impressive retatrutide phase 2 results have accelerated development timelines, with regulatory submissions anticipated within the next 2-3 years pending successful phase 3 completion.
Current Development Status (2026)
Phase 3 Trials: Multiple pivotal studies actively enrolling or in progress across obesity, diabetes, and cardiovascular indications
Regulatory Interactions: Ongoing dialogue with FDA, EMA, and other regulatory authorities regarding approval pathways and required evidence
Manufacturing Scale-Up: Development of commercial-scale production capabilities to support potential market launch
Companion Diagnostic Development: Investigation of biomarkers that might predict response or guide personalized dosing
Anticipated Approval Timeline
Based on current development progress and regulatory precedent:
2026-2027: Completion of pivotal phase 3 efficacy trials 2027-2028: Regulatory submissions (NDA/BLA in US, MAA in Europe) 2028-2029: Potential regulatory approvals and market launch 2029+: Post-marketing surveillance and label expansion studies
This timeline assumes favorable phase 3 outcomes consistent with the retatrutide phase 2 results and no unexpected safety signals requiring additional investigation.
Market Access and Reimbursement Considerations 💰
Beyond regulatory approval, market access requires demonstration of:
Clinical Value: Superior efficacy, safety, or convenience compared to existing therapies Economic Value: Cost-effectiveness relative to current standard of care Real-World Effectiveness: Confirmation that clinical trial outcomes translate to routine practice Long-Term Outcomes: Demonstration of sustained benefits and complication prevention
Health technology assessment bodies increasingly require comprehensive economic modeling demonstrating that higher drug costs are offset by reduced healthcare utilization for obesity-related complications (diabetes, cardiovascular disease, joint replacement, etc.).
The magnitude of weight loss observed in retatrutide phase 2 results, if confirmed in phase 3 and sustained long-term, would support favorable economic evaluations given the substantial healthcare costs associated with obesity and metabolic disease.
Conclusion: Transformative Potential of Retatrutide
The retatrutide phase 2 results represent a watershed moment in metabolic therapeutics, demonstrating that rational multi-receptor targeting can achieve weight loss magnitudes previously attainable only through bariatric surgery. The 17.5% mean weight reduction observed with the 12mg dose, coupled with comprehensive metabolic improvements and acceptable tolerability, positions retatrutide as a potentially transformative intervention for obesity and related metabolic disorders.
Several key conclusions emerge from comprehensive analysis of these phase 2 findings:
✅ Triple receptor agonism provides synergistic metabolic benefits beyond single or dual agonist approaches, validating the mechanistic hypothesis underlying retatrutide’s development
✅ Dose-dependent efficacy with manageable tolerability enables optimization of the benefit-risk profile for individual patients
✅ Comprehensive metabolic improvements extend beyond weight loss to encompass glycemic control, cardiovascular risk factors, and body composition
✅ Favorable safety profile consistent with the incretin-based therapeutic class, with predominantly mild-to-moderate gastrointestinal effects
✅ Promising foundation for phase 3 development, with ongoing pivotal trials positioned to confirm efficacy and safety in larger, more diverse populations
For the research community, these findings validate multi-agonist approaches and will likely catalyze investigation of additional receptor combinations targeting metabolic disease. The mechanistic insights gained from retatrutide’s development inform rational drug design strategies applicable across therapeutic areas.
Next Steps for Researchers and Institutions
Researchers interested in contributing to the evolving understanding of multi-agonist peptide therapeutics should consider:
- Mechanistic Studies: Investigating tissue-specific effects, receptor crosstalk, and signaling pathway integration underlying triple agonism
- Biomarker Discovery: Identifying predictors of exceptional response or adverse event susceptibility
- Combination Strategies: Evaluating synergy with complementary interventions (behavioral, pharmacological, or device-based)
- Long-Term Outcomes Research: Assessing durability of weight loss and metabolic improvements with extended treatment
- Translational Investigation: Bridging preclinical findings to clinical applications through well-designed proof-of-concept studies
Access to premium research-grade peptides remains essential for conducting rigorous investigations that advance scientific knowledge and therapeutic development. PEPTIDE PRO’s commitment to exceptional purity, comprehensive documentation, and responsive technical support enables researchers to pursue ambitious scientific questions with confidence in their experimental tools.
The retatrutide phase 2 results have illuminated a promising path forward in addressing the global obesity epidemic. As phase 3 trials progress and additional data emerge, the research community will continue refining understanding of optimal utilization strategies, patient selection criteria, and mechanistic foundations that will shape next-generation metabolic therapeutics for years to come.
