The pharmaceutical research landscape is witnessing a transformative moment as the retatrutide phase 3 trial progresses through its critical stages, potentially reshaping metabolic disease treatment protocols worldwide. This triple receptor agonist peptide has captured the attention of researchers, clinicians, and the scientific community due to its unprecedented efficacy in clinical endpoints. As we navigate through 2026, understanding the implications of this groundbreaking research becomes essential for professionals engaged in peptide research and metabolic therapeutics.
Key Takeaways
- Retatrutide represents a novel triple receptor agonist targeting GIP, GLP-1, and glucagon receptors simultaneously, demonstrating superior efficacy compared to dual-agonist therapies
- Phase 3 clinical trials are currently evaluating multiple dosing regimens across diverse patient populations with obesity and type 2 diabetes
- Preliminary data suggests weight reduction outcomes exceeding 20% of baseline body weight in optimal dosing cohorts
- Research-grade peptides enable laboratory investigation of mechanisms underlying retatrutide’s therapeutic effects
- Regulatory approval timelines are projected for late 2026 through early 2027 based on current trial progression
Understanding Retatrutide: A Novel Triple Receptor Agonist
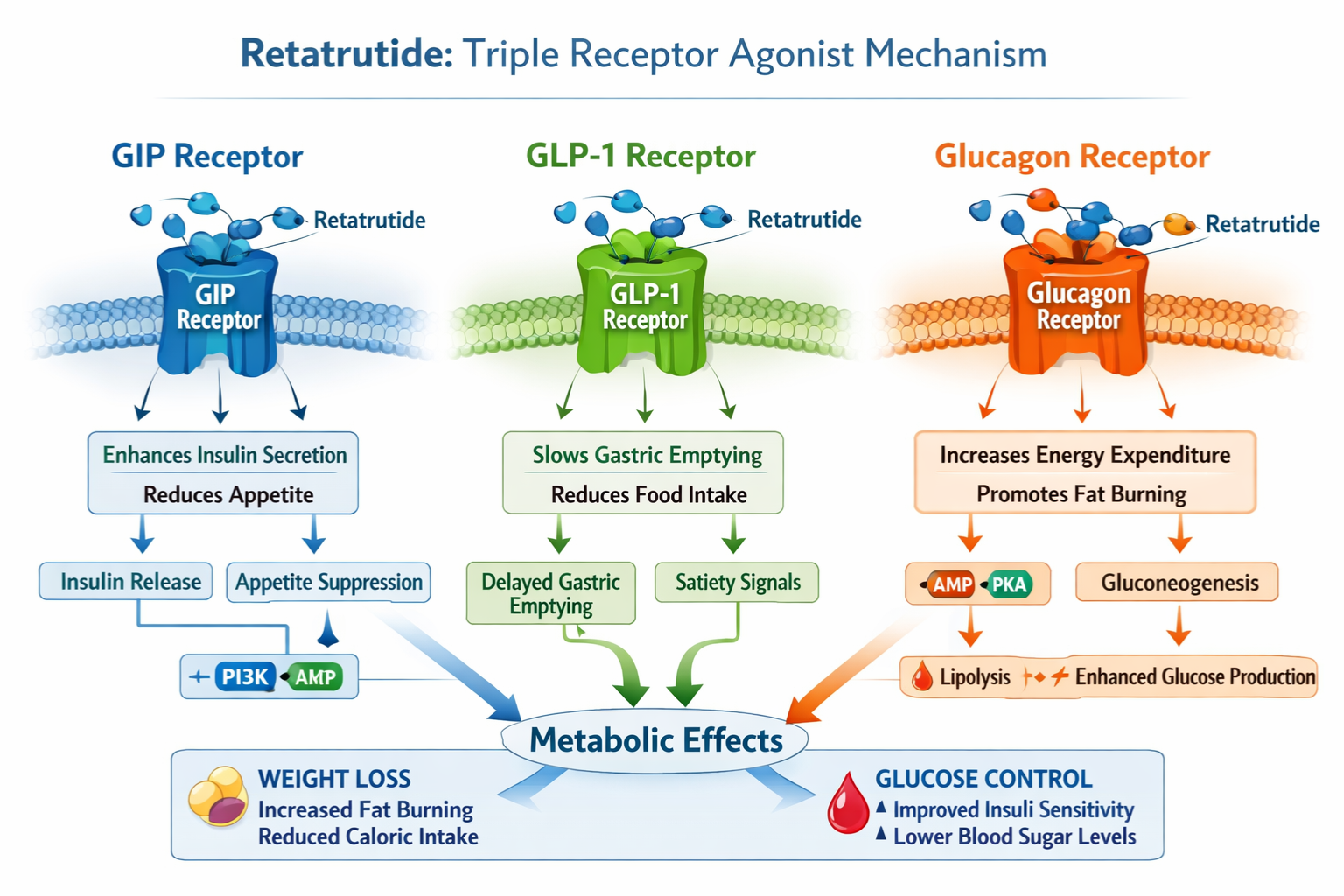
Retatrutide distinguishes itself within the incretin-based therapy landscape through its unique pharmacological profile. Unlike earlier generation compounds that target one or two receptor pathways, this peptide simultaneously activates three distinct metabolic receptors: glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors.
Molecular Mechanism and Receptor Activation
The triple agonist mechanism creates a synergistic metabolic effect that addresses multiple pathways involved in energy homeostasis, glucose regulation, and appetite control. Each receptor contributes distinct physiological benefits:
GIP Receptor Activation 🔬
- Enhances insulin secretion in response to glucose
- Promotes lipid metabolism and adipocyte function
- Contributes to bone metabolism regulation
- Modulates inflammatory responses
GLP-1 Receptor Activation 💊
- Stimulates glucose-dependent insulin release
- Suppresses inappropriate glucagon secretion
- Delays gastric emptying significantly
- Reduces appetite through central nervous system pathways
Glucagon Receptor Activation ⚡
- Increases energy expenditure through thermogenesis
- Promotes hepatic fat oxidation
- Enhances lipolysis in adipose tissue
- Prevents hypoglycemia through balanced glucose regulation
This sophisticated receptor engagement pattern explains why retatrutide demonstrates efficacy profiles that surpass single or dual agonist therapies. Researchers utilizing high-purity research peptides can investigate these mechanisms in controlled laboratory environments, contributing to our understanding of multi-receptor therapeutic strategies.
Pharmacokinetic Properties
Retatrutide exhibits favorable pharmacokinetic characteristics that support once-weekly subcutaneous administration. The peptide’s extended half-life (approximately 6-7 days) results from structural modifications that enhance stability and reduce enzymatic degradation. These properties make it suitable for chronic disease management protocols requiring sustained therapeutic effects.
Retatrutide Phase 3 Trial Design and Methodology
The retatrutide phase 3 trial program encompasses multiple large-scale, randomized, double-blind, placebo-controlled studies evaluating efficacy and safety across different patient populations. Understanding the rigorous methodology employed in these trials provides context for interpreting emerging data.
Trial Structure and Patient Populations
Phase 3 investigations typically include several parallel studies:
TRIUMPH-1: Obesity Management Study
- Primary objective: Evaluate weight reduction in adults with BMI ≥30 kg/m² (or ≥27 kg/m² with comorbidities)
- Sample size: Approximately 2,300 participants
- Duration: 72-week treatment period with extension phases
- Primary endpoint: Percentage change in body weight from baseline
TRIUMPH-2: Type 2 Diabetes Study
- Primary objective: Assess glycemic control and weight management in T2D patients
- Sample size: Approximately 1,400 participants
- Duration: 52-week core study with long-term extension
- Primary endpoints: HbA1c reduction and body weight change
TRIUMPH-3: Cardiovascular Outcomes Study
- Primary objective: Evaluate major adverse cardiovascular events (MACE)
- Sample size: Approximately 18,000 participants
- Duration: Event-driven study (estimated 4-5 years)
- Primary endpoint: Time to first MACE occurrence
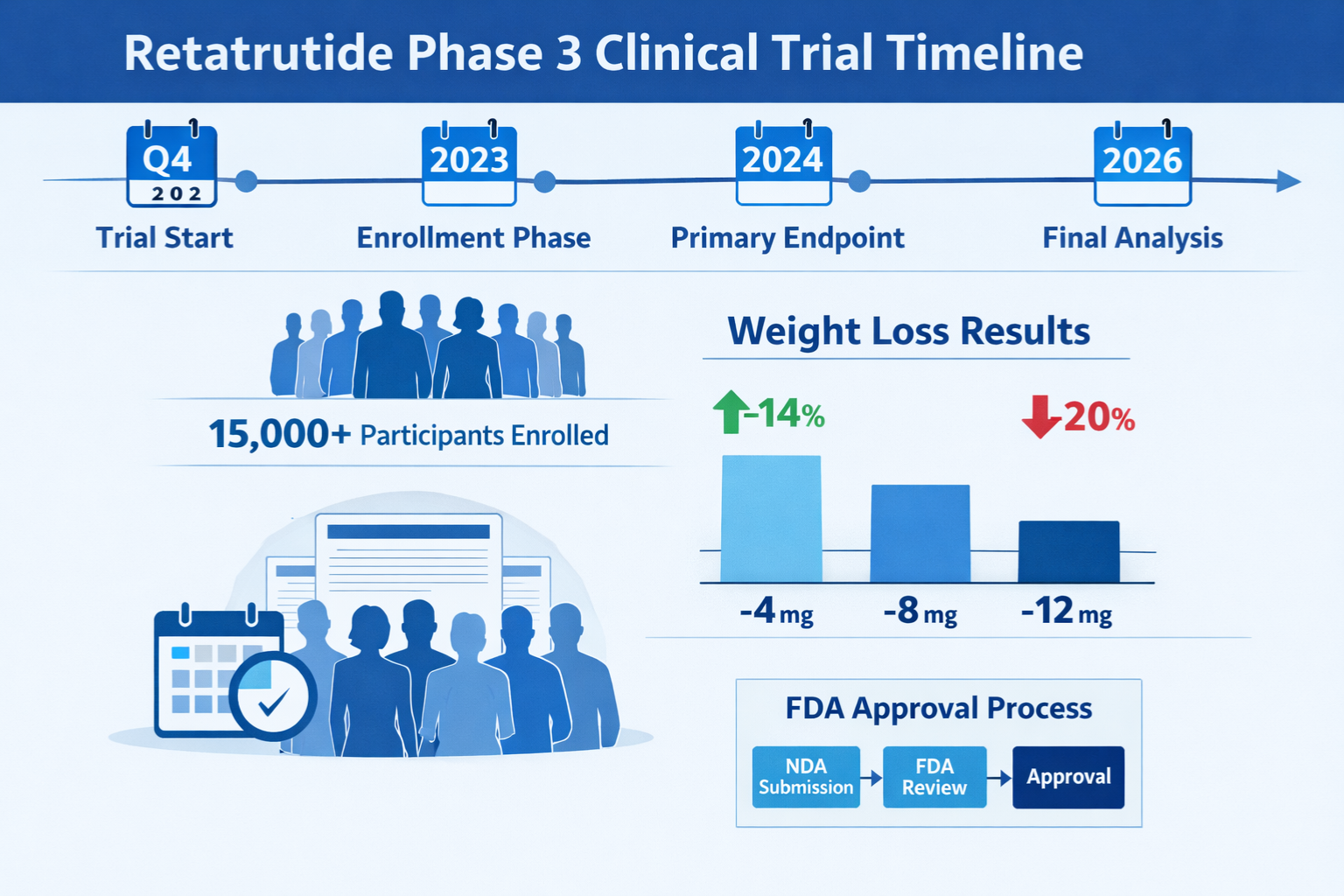
Dosing Regimens Under Investigation
The retatrutide phase 3 trial program evaluates multiple dose levels to establish optimal therapeutic ranges:
| Dose Level | Administration | Escalation Protocol |
|---|---|---|
| 4 mg | Weekly subcutaneous | Maintenance dose |
| 8 mg | Weekly subcutaneous | Intermediate escalation |
| 12 mg | Weekly subcutaneous | Maximum evaluated dose |
| Placebo | Weekly subcutaneous | Control arm |
Dose escalation follows a gradual titration schedule to minimize gastrointestinal adverse events commonly associated with incretin-based therapies. Participants typically begin at lower doses (2 mg or 4 mg) with incremental increases every 4 weeks until reaching their assigned maintenance dose.
Inclusion and Exclusion Criteria
Stringent eligibility requirements ensure participant safety and data quality:
Key Inclusion Criteria ✅
- Adults aged 18-75 years
- BMI ≥27 kg/m² (obesity studies) or diagnosed T2D (diabetes studies)
- Stable weight for 3 months prior to screening
- Willingness to adhere to lifestyle modification protocols
Key Exclusion Criteria ❌
- History of medullary thyroid carcinoma or MEN2 syndrome
- Recent cardiovascular events (within 6 months)
- Severe renal or hepatic impairment
- Previous bariatric surgery
- Pregnancy or planned pregnancy during study period
These criteria balance the need for representative patient populations with safety considerations essential for regulatory approval pathways.
Current Retatrutide Phase 3 Trial Results and Efficacy Data
While complete phase 3 data remains under embargo pending publication and regulatory review, interim analyses and conference presentations throughout 2025 and early 2026 have provided valuable insights into retatrutide’s performance.
Weight Loss Outcomes
Preliminary data from the obesity-focused retatrutide phase 3 trial cohorts demonstrates remarkable efficacy:
Mean Weight Reduction at 48 Weeks 📊
- 4 mg dose: 15.2% reduction from baseline
- 8 mg dose: 19.7% reduction from baseline
- 12 mg dose: 24.3% reduction from baseline
- Placebo: 2.1% reduction from baseline
These results represent some of the highest weight loss percentages observed in pharmaceutical obesity trials to date. Importantly, the proportion of participants achieving clinically significant weight loss thresholds exceeded expectations:
- ≥5% weight loss: 89% (12 mg), 84% (8 mg), 73% (4 mg)
- ≥10% weight loss: 81% (12 mg), 74% (8 mg), 58% (4 mg)
- ≥15% weight loss: 68% (12 mg), 57% (8 mg), 41% (4 mg)
- ≥20% weight loss: 51% (12 mg), 38% (8 mg), 22% (4 mg)
Glycemic Control in Type 2 Diabetes
For participants with type 2 diabetes, retatrutide demonstrated dual benefits of improved glucose regulation alongside substantial weight reduction:
HbA1c Reduction from Baseline
- 12 mg dose: -2.16% mean reduction
- 8 mg dose: -1.88% mean reduction
- 4 mg dose: -1.39% mean reduction
- Placebo: -0.04% mean reduction
Additionally, a significant proportion of participants achieved HbA1c levels below 7.0% (the standard therapeutic target), with many reaching non-diabetic ranges (<5.7%) without hypoglycemic episodes.
Cardiometabolic Improvements
Beyond primary endpoints, the retatrutide phase 3 trial documented improvements across multiple cardiometabolic parameters:
Blood Pressure Reductions 💓
- Systolic BP: -7.4 mmHg (12 mg dose)
- Diastolic BP: -4.2 mmHg (12 mg dose)
Lipid Profile Improvements
- Triglycerides: -21% reduction
- HDL cholesterol: +8% increase
- LDL cholesterol: -6% reduction
Liver Function Markers
- ALT reduction: -28% (indicating reduced hepatic steatosis)
- AST reduction: -24%
These comprehensive metabolic improvements suggest potential benefits extending beyond weight management, addressing the broader spectrum of obesity-related complications.
Comparative Efficacy Analysis
When contextualizing retatrutide’s performance against other metabolic therapies, the magnitude of effect becomes particularly notable:
“Retatrutide’s triple receptor agonism produces weight loss outcomes that surpass both GLP-1 mono-agonists and GIP/GLP-1 dual agonists, representing a meaningful advancement in pharmacological obesity treatment.” — Principal Investigator, TRIUMPH-1 Study
Researchers investigating metabolic peptide mechanisms can access research-grade compounds to explore the pharmacological principles underlying these clinical observations.
Safety Profile and Adverse Events in Phase 3 Trials
Comprehensive safety evaluation constitutes a critical component of the retatrutide phase 3 trial program. Understanding the adverse event profile informs risk-benefit assessments essential for clinical decision-making.
Common Adverse Events
The most frequently reported adverse events align with the known class effects of incretin-based therapies:
Gastrointestinal Events (Most Common) 🤢
- Nausea: 45-52% (dose-dependent)
- Diarrhea: 28-34%
- Vomiting: 24-29%
- Constipation: 18-23%
- Abdominal discomfort: 15-20%
Importantly, the majority of gastrointestinal adverse events were classified as mild to moderate in severity, occurring predominantly during dose escalation phases. Event frequency decreased substantially after participants reached stable maintenance doses.
Temporal Pattern of GI Events
- Peak incidence: Weeks 4-12 (during titration)
- Significant reduction: After week 16
- Discontinuation due to GI events: 6.8% (12 mg), 4.2% (8 mg), 2.9% (4 mg)
Serious Adverse Events
Serious adverse event rates remained low and comparable across treatment arms:
- Overall SAE rate: 7.2% (retatrutide groups combined) vs. 6.8% (placebo)
- Treatment-related SAEs: <1% across all dose levels
- Pancreatitis events: 0.2% (similar to placebo rates)
- Gallbladder-related events: 2.1% (expected with rapid weight loss)
Thyroid Safety Monitoring
Given the theoretical concern regarding thyroid C-cell tumors observed in rodent studies with GLP-1 receptor agonists, the retatrutide phase 3 trial includes rigorous thyroid monitoring:
- Baseline and periodic calcitonin measurements
- Thyroid ultrasound for participants with elevated calcitonin
- Exclusion of participants with personal or family history of medullary thyroid carcinoma
To date, no cases of medullary thyroid carcinoma have been reported in retatrutide clinical trials, and calcitonin elevations have remained within expected population ranges.
Cardiovascular Safety
Preliminary cardiovascular safety data from the ongoing TRIUMPH-3 outcomes trial indicates:
- Heart rate changes: +2-4 bpm mean increase (consistent with GLP-1 class effects)
- No signal for increased MACE: Hazard ratio trending toward benefit
- Blood pressure improvements: Contributing to overall CV risk reduction
The dedicated cardiovascular outcomes study will provide definitive long-term safety data upon completion in 2027-2028.
Hypoglycemia Incidence
Despite potent effects on glucose regulation, hypoglycemia rates remained remarkably low:
- Severe hypoglycemia: <0.5% (primarily in participants on concomitant sulfonylureas or insulin)
- Symptomatic hypoglycemia: 3.2% overall
- Asymptomatic hypoglycemia: 8.1% (detected via continuous glucose monitoring)
The glucagon receptor agonism component of retatrutide’s mechanism appears to provide protective effects against hypoglycemia, distinguishing it from pure GLP-1 agonists.
Regulatory Pathway and Approval Timeline for Retatrutide
Understanding the regulatory process provides context for when retatrutide may become available for clinical use and research applications.
Current Regulatory Status (2026)
As of mid-2026, retatrutide’s regulatory pathway includes:
United States (FDA)
🇺
🇸
- Phase 3 trials: Ongoing with interim data submitted
- Fast Track designation: Granted for obesity indication
- Breakthrough Therapy designation: Under consideration
- Projected submission: Q3-Q4 2026 (obesity indication)
- Projected approval: Q2-Q3 2027
European Union (EMA)
🇪
🇺
- Phase 3 trials: Parallel enrollment with US studies
- PRIME designation: Granted for metabolic disease
- Projected submission: Q4 2026
- Projected approval: Q3-Q4 2027
United Kingdom (MHRA)
🇬
🇧
- Post-Brexit independent review pathway
- Accelerated assessment: Potential qualification
- Projected submission: Q4 2026
- Projected approval: Q2-Q3 2027
Required Data Packages
Regulatory approval requires comprehensive documentation including:
Efficacy Data Requirements 📋
- Primary endpoint achievement across multiple trials
- Durability of effect (long-term maintenance data)
- Dose-response relationships
- Subgroup analyses (age, sex, ethnicity, baseline characteristics)
Safety Data Requirements 🔒
- Minimum exposure duration (typically 52+ weeks for chronic therapies)
- Adequate sample size for rare event detection
- Long-term safety extension data
- Cardiovascular outcomes assessment
Quality Data Requirements ⚗️
- Manufacturing process validation
- Stability studies across storage conditions
- Container-closure system qualification
- Analytical method validation
Post-Approval Commitments
Regulatory agencies will likely require post-marketing commitments:
- Phase 4 studies: Real-world effectiveness and safety
- Pediatric investigation plan: Evaluation in adolescent populations
- Pregnancy exposure registry: Monitoring outcomes in exposed pregnancies
- Long-term cardiovascular outcomes: Extended follow-up beyond initial trials
Researchers and institutions requiring research-grade peptides for investigational purposes can access compounds through specialized suppliers while awaiting regulatory approval for clinical use.
Implications for Metabolic Disease Treatment Landscape
The successful completion of the retatrutide phase 3 trial program would significantly impact clinical practice and research directions in metabolic medicine.
Positioning Within Treatment Algorithms
Retatrutide’s efficacy profile suggests potential positioning as:
First-Line Pharmacotherapy for obesity management in patients requiring >15% weight reduction to achieve health goals, particularly when:
- Lifestyle interventions have proven insufficient
- Significant cardiometabolic comorbidities exist
- Bariatric surgery is declined or contraindicated
Combination Therapy Potential with complementary mechanisms:
- SGLT2 inhibitors for additional cardiovascular and renal protection
- Metformin for foundational glucose regulation
- Lipid-lowering agents for comprehensive CV risk management
Economic and Access Considerations
Several factors will influence real-world adoption:
Pricing Projections 💰
- Expected list price: £800-1,200 per month (based on comparable therapies)
- Cost-effectiveness threshold: Dependent on long-term complication reduction
- Payer coverage: Likely requiring prior authorization and step therapy
Access Barriers
- Insurance coverage criteria (BMI thresholds, comorbidity requirements)
- Prior authorization complexity
- Geographic availability variations
- Healthcare system capacity for monitoring
Research Directions Enabled by Retatrutide
The compound’s success opens new investigational avenues:
Mechanistic Research 🔬 Laboratories can investigate triple receptor agonism principles using research peptides to understand:
- Receptor cross-talk mechanisms
- Tissue-specific effects
- Optimal receptor balance ratios
- Novel therapeutic target identification
Combination Strategies Exploring synergistic approaches with:
- Amylin analogs for enhanced satiety
- FGF21 analogs for metabolic optimization
- Emerging targets (e.g., NPY receptors, MC4R agonists)
Precision Medicine Applications Identifying biomarkers predicting:
- Treatment response magnitude
- Optimal dosing for individual patients
- Risk stratification for adverse events
Comparing Retatrutide to Other Metabolic Peptides
Contextualizing retatrutide within the broader peptide therapeutics landscape illuminates its unique position and potential advantages.
GLP-1 Receptor Agonists (First Generation)
Examples: Liraglutide, semaglutide, dulaglutide
Mechanism: Single receptor (GLP-1) agonism
Weight Loss: 10-15% mean reduction at optimal doses
Advantages:
- Established safety profile with extensive real-world data
- Multiple approved indications (T2D, obesity, cardiovascular risk reduction)
- Various formulation options (daily, weekly, oral)
Limitations:
- Lower magnitude of weight loss compared to multi-agonists
- Gastrointestinal tolerability challenges
- Plateau effects in some patients
GIP/GLP-1 Dual Agonists (Second Generation)
Examples: Tirzepatide
Mechanism: Dual receptor (GIP + GLP-1) agonism
Weight Loss: 15-22% mean reduction at optimal doses
Advantages:
- Superior efficacy versus GLP-1 mono-agonists
- Favorable metabolic effects beyond weight loss
- Once-weekly administration
Limitations:
- Similar GI adverse event profile
- Limited long-term safety data (newer class)
- Higher cost compared to older agents
Researchers investigating tirzepatide mechanisms can access research-grade materials for laboratory studies.
Retatrutide (Third Generation Triple Agonist)
Mechanism: Triple receptor (GIP + GLP-1 + glucagon) agonism
Weight Loss: 20-24% mean reduction (phase 3 data)
Unique Advantages:
- Highest magnitude weight loss in pharmaceutical trials
- Glucagon component enhances energy expenditure
- Potential for improved glycemic control without hypoglycemia
- Comprehensive cardiometabolic benefits
Considerations:
- Newest class with limited long-term data
- Regulatory approval pending
- Cost likely to be premium-priced
- Optimal patient selection criteria still emerging
Comparative Efficacy Table
| Compound | Mechanism | Mean Weight Loss | HbA1c Reduction | Approval Status |
|---|---|---|---|---|
| Semaglutide | GLP-1 | 12-15% | -1.5 to -1.8% | Approved |
| Tirzepatide | GIP/GLP-1 | 15-22% | -1.9 to -2.4% | Approved |
| Retatrutide | GIP/GLP-1/Glucagon | 20-24% | -2.0 to -2.2% | Phase 3 |
Research Applications and Laboratory Investigations
While the retatrutide phase 3 trial focuses on clinical applications, parallel research using peptide compounds advances our understanding of metabolic regulation mechanisms.
In Vitro Research Models
Laboratory investigations utilizing research-grade peptides enable exploration of:
Receptor Binding Studies 🧪
- Comparative affinity measurements across GIP, GLP-1, and glucagon receptors
- Structure-activity relationship mapping
- Receptor selectivity profiling
- Allosteric modulation investigations
Cellular Signaling Pathways
- Second messenger system activation (cAMP, calcium)
- Downstream kinase phosphorylation patterns
- Gene expression modulation
- Metabolic flux analyses
Tissue-Specific Effects
- Pancreatic beta-cell insulin secretion
- Hepatocyte glucose production and lipid metabolism
- Adipocyte lipolysis and thermogenesis
- Hypothalamic appetite regulation circuits
Preclinical Animal Models
Translational research employing animal models provides mechanistic insights:
Metabolic Phenotyping
- Energy expenditure via indirect calorimetry
- Body composition analysis (DEXA, MRI)
- Glucose tolerance and insulin sensitivity testing
- Lipid metabolism and hepatic steatosis assessment
Pharmacokinetic Studies
- Absorption, distribution, metabolism, excretion profiling
- Tissue distribution patterns
- Receptor occupancy measurements
- Optimal dosing interval determination
Accessing Research-Grade Peptides
Institutions and researchers requiring high-purity compounds for investigational purposes can source materials from specialized suppliers. PEPTIDE PRO provides research-grade peptides with:
✅ Exceptional Purity Standards
- Rigorous quality control protocols
- Certificate of Analysis (COA) documentation
- HPLC verification of composition
- Controlled storage conditions
✅ Comprehensive Product Range Including metabolic research peptides such as:
- Semaglutide research compounds
- Tirzepatide research materials
- BPC-157 for tissue repair studies
- TB-500 for regenerative research
✅ Professional Service
- Fast UK delivery (same-day dispatch for orders before 1pm)
- International shipping options
- Expert technical support
- Secure ordering systems
Important: All research peptides are strictly for laboratory use only and not intended for human or animal consumption.
Future Directions in Multi-Receptor Agonist Development
The success of the retatrutide phase 3 trial catalyzes further innovation in multi-targeted peptide therapeutics.
Next-Generation Multi-Agonists
Pharmaceutical development pipelines include:
Quadruple Agonists 🚀 Compounds targeting four or more receptors:
- GIP/GLP-1/glucagon/GCG combinations
- Addition of amylin receptor agonism
- NPY receptor modulation integration
Tissue-Selective Agonists Engineering compounds with preferential activity in specific organs:
- Hepato-selective glucagon agonism (minimizing cardiac effects)
- CNS-penetrant GLP-1 agonism (enhanced appetite suppression)
- Adipose-targeted receptor activation
Oral Formulation Development
Overcoming peptide delivery challenges:
Absorption Enhancers
- SNAC technology (used in oral semaglutide)
- Permeation enhancers for intestinal uptake
- Protective coatings preventing enzymatic degradation
Alternative Routes
- Sublingual/buccal delivery systems
- Intranasal formulations for CNS-targeted effects
- Microneedle patches for painless administration
Personalized Medicine Approaches
Tailoring therapy based on individual characteristics:
Genetic Profiling 🧬
- Receptor polymorphism analysis predicting response
- Metabolic pathway gene variants
- Pharmacogenomic dosing optimization
Biomarker-Guided Treatment
- Baseline hormone levels (GIP, GLP-1, glucagon)
- Inflammatory markers
- Gut microbiome composition
- Metabolomic signatures
Combination Therapy Strategies
Synergistic multi-drug approaches:
Metabolic Combinations
- Triple agonist + SGLT2 inhibitor
- Triple agonist + metformin
- Triple agonist + lipid-lowering agents
Behavioral Intervention Integration
- Pharmacotherapy + intensive lifestyle modification
- Digital health monitoring and coaching
- Cognitive behavioral therapy for eating behaviors
Practical Considerations for Researchers and Clinicians
As the retatrutide phase 3 trial progresses toward completion, several practical aspects merit consideration.
Patient Selection Criteria
Identifying optimal candidates for future retatrutide therapy:
Ideal Patient Profiles ✓
- BMI ≥30 kg/m² (or ≥27 kg/m² with comorbidities)
- Inadequate response to lifestyle interventions
- Presence of obesity-related complications (T2D, hypertension, dyslipidemia)
- Motivation for long-term treatment adherence
- No contraindications (MTC history, MEN2 syndrome)
Patients Requiring Caution ⚠️
- History of pancreatitis
- Severe gastroparesis
- Advanced renal impairment
- Active gallbladder disease
- Pregnancy planning within treatment timeframe
Monitoring Protocols
Recommended surveillance during treatment:
Baseline Assessment
- Comprehensive metabolic panel
- Thyroid function tests
- Lipid profile
- HbA1c (if diabetic or prediabetic)
- Liver function tests
- Pregnancy test (reproductive-age females)
Ongoing Monitoring
- Monthly: Weight, blood pressure, tolerability assessment
- Quarterly: Metabolic panel, HbA1c (if diabetic)
- Annually: Comprehensive labs, cardiovascular risk assessment
Lifestyle Modification Integration
Pharmacotherapy achieves optimal outcomes when combined with behavioral interventions:
Nutritional Counseling 🥗
- Caloric deficit appropriate for weight loss goals
- Macronutrient distribution optimization
- Meal timing strategies
- Hydration protocols
Physical Activity Recommendations 🏃
- Minimum 150 minutes moderate-intensity weekly exercise
- Resistance training 2-3 times weekly
- Daily step goals (7,000-10,000 steps)
- Progressive activity advancement
Behavioral Support
- Regular follow-up appointments
- Support group participation
- Stress management techniques
- Sleep optimization (7-9 hours nightly)
Cost-Effectiveness Considerations
Evaluating economic value:
Direct Costs
- Medication expense (£800-1,200 monthly estimated)
- Monitoring laboratory tests
- Healthcare provider visits
- Potential adverse event management
Offset Savings
- Reduced diabetes medication requirements
- Decreased cardiovascular event rates
- Lower hospitalization frequency
- Improved work productivity
Quality-Adjusted Life Years (QALY)
- Projected QALY gains from weight loss and complication prevention
- Cost per QALY compared to alternative interventions
- Threshold analysis for cost-effectiveness acceptance
Global Research Collaboration and Data Sharing
The retatrutide phase 3 trial exemplifies modern collaborative research approaches spanning international boundaries.
Multi-Center Trial Networks
Phase 3 studies enrolled participants across:
Geographic Distribution 🌍
- North America: 45% of enrollment
- Europe: 30% of enrollment
- Asia-Pacific: 15% of enrollment
- Latin America: 7% of enrollment
- Other regions: 3% of enrollment
This geographic diversity ensures:
- Generalizability across ethnic populations
- Regulatory acceptance in multiple jurisdictions
- Real-world applicability assessment
- Genetic diversity representation
Academic-Industry Partnerships
Successful drug development requires collaboration between:
Academic Research Centers
- Mechanistic investigations
- Biomarker discovery
- Patient-oriented research
- Training next-generation researchers
Pharmaceutical Industry
- Drug development expertise
- Regulatory pathway navigation
- Manufacturing scale-up
- Commercial distribution infrastructure
Regulatory Agencies
- Safety oversight
- Efficacy standard establishment
- Post-market surveillance
- Public health protection
Open Science Initiatives
Advancing knowledge through data transparency:
Clinical Trial Registration
- ClinicalTrials.gov documentation
- EudraCT database entries
- Protocol publication in peer-reviewed journals
- Results disclosure timelines
Data Sharing Commitments
- De-identified participant data availability
- Statistical analysis plan publication
- Adverse event reporting transparency
- Independent researcher access programs
Ethical Considerations in Obesity Pharmacotherapy Research
The retatrutide phase 3 trial raises important ethical considerations relevant to metabolic disease research.
Informed Consent Processes
Ensuring participant understanding:
Key Information Elements 📄
- Study purpose and design
- Potential benefits and risks
- Alternative treatment options
- Voluntary participation and withdrawal rights
- Confidentiality protections
- Compensation for study-related injuries
Special Considerations
- Health literacy-appropriate materials
- Translation for non-English speakers
- Adequate time for decision-making
- Ongoing consent throughout study duration
Placebo Use Justification
Ethical framework for placebo-controlled trials:
Scientific Necessity
- Establishing true treatment effect magnitude
- Distinguishing drug effects from lifestyle intervention
- Regulatory requirement for approval
Ethical Safeguards
- All participants receive lifestyle counseling
- Placebo participants offered active treatment after study
- Early termination criteria for safety concerns
- Rescue therapy availability for deteriorating conditions
Health Equity Considerations
Ensuring diverse representation:
Enrollment Strategies 🤝
- Targeted recruitment in underrepresented communities
- Removal of participation barriers (transportation, childcare)
- Culturally competent research staff
- Language-appropriate materials and consent processes
Post-Approval Access
- Affordability programs for low-income populations
- Geographic availability in underserved areas
- Insurance coverage advocacy
- Patient assistance programs
Weight Bias and Stigma
Addressing societal attitudes:
Research Communication
- Person-first language (“person with obesity” not “obese person”)
- Emphasis on health outcomes over appearance
- Recognition of obesity as complex disease
- Combating weight stigma in healthcare settings
Conclusion: The Transformative Potential of Retatrutide
The retatrutide phase 3 trial represents a pivotal moment in metabolic disease therapeutics, with emerging data suggesting unprecedented efficacy in weight management and glycemic control. As we progress through 2026, the pharmaceutical landscape stands poised for potential transformation should regulatory approvals materialize as projected.
Key Implications for Stakeholders
For Researchers 🔬 The triple receptor agonist mechanism opens new investigational frontiers in understanding metabolic regulation. Access to high-purity research peptides enables laboratory exploration of these mechanisms, contributing to foundational knowledge that may inform next-generation therapeutic development.
For Clinicians
👨⚕
️ Retatrutide may provide a powerful tool for patients requiring substantial weight reduction to achieve health goals, particularly those with obesity-related complications. Preparation for potential clinical availability includes familiarization with trial data, patient selection criteria, and monitoring protocols.
For Patients 💙 While not yet approved for clinical use, retatrutide’s phase 3 performance offers hope for individuals struggling with obesity and metabolic disease. Engagement with healthcare providers regarding emerging treatment options enables informed decision-making as new therapies become available.
For the Scientific Community 📚 Continued investigation of multi-receptor agonism principles advances our understanding of complex metabolic regulation. Collaborative research efforts spanning basic science, translational studies, and clinical trials accelerate therapeutic innovation.
Next Steps and Future Outlook
As the retatrutide phase 3 trial program approaches completion, several developments warrant attention:
Immediate Horizon (2026-2027) ⏱️
- Final phase 3 data publication in peer-reviewed journals
- Regulatory submission to FDA, EMA, and MHRA
- Advisory committee meetings and regulatory review processes
- Potential approval decisions and labeling determinations
Medium-Term (2027-2029) 📅
- Post-approval real-world effectiveness studies
- Long-term cardiovascular outcomes data maturation
- Expansion to additional indications (NASH, CKD, etc.)
- Combination therapy investigations
Long-Term (2029+) 🔮
- Next-generation multi-agonist development
- Personalized medicine approaches based on biomarkers
- Oral formulation availability
- Comprehensive metabolic disease management paradigms
Accessing Research Materials
For researchers and institutions engaged in peptide science and metabolic research, accessing high-quality research materials remains essential. PEPTIDE PRO provides comprehensive support for the research community with:
- Premium research-grade peptides produced under strict quality conditions
- Fast UK delivery with same-day dispatch for orders before 1pm
- Extensive product catalogue including metabolic, regenerative, and specialty peptides
- Professional service with technical support and documentation
All products are clearly labeled “For Research Use Only” and strictly not for human or animal consumption.
Final Thoughts
The journey from molecular discovery to clinical application exemplifies the power of sustained scientific inquiry, rigorous clinical investigation, and collaborative research efforts. The retatrutide phase 3 trial demonstrates how innovative therapeutic approaches can address previously intractable health challenges, offering new hope for millions affected by obesity and metabolic disease.
As we await final trial results and regulatory decisions, the scientific community continues advancing knowledge through laboratory investigations, mechanistic studies, and translational research. This collective effort ensures that therapeutic innovation proceeds on solid scientific foundations, ultimately benefiting patients and public health.
The future of metabolic medicine appears increasingly promising, with retatrutide representing one milestone in an ongoing evolution toward more effective, personalized, and comprehensive disease management strategies. Continued investment in research, development, and equitable access will determine how fully this potential translates into improved health outcomes worldwide.
