
The global prediabetes epidemic affects over 374 million individuals worldwide, yet fewer than 10% receive effective intervention before progressing to type 2 diabetes. Enter retatrutide—a groundbreaking triple-agonist peptide that’s revolutionizing metabolic research and offering unprecedented hope for reversing prediabetic conditions. Understanding retatrutide prediabetes outcomes has become critical for researchers investigating next-generation metabolic interventions, as emerging 2026 data reveals remarkable glycemic control improvements and weight reduction profiles that surpass existing therapeutic options.
This comprehensive analysis examines the latest research findings, clinical trial data, and mechanistic insights surrounding retatrutide’s application in prediabetes management, providing researchers with evidence-based perspectives on this transformative peptide compound.
Key Takeaways
- Triple-Mechanism Action: Retatrutide simultaneously activates GIP, GLP-1, and glucagon receptors, creating synergistic metabolic effects superior to dual-agonist compounds
- Significant Glycemic Improvements: Research demonstrates HbA1c reductions of 0.8-1.3% in prediabetic populations, with 70-85% of participants achieving normoglycemia
- Substantial Weight Loss: Clinical investigations report average weight reductions of 12-24% over 48 weeks, addressing the critical obesity-prediabetes connection
- Diabetes Prevention Potential: Preliminary data suggests retatrutide may reduce type 2 diabetes progression risk by up to 89% in high-risk prediabetic individuals
- Favorable Safety Profile: Current research indicates tolerability comparable to GLP-1 agonists, with predominantly mild-to-moderate gastrointestinal side effects
Understanding Retatrutide: Mechanism and Metabolic Impact
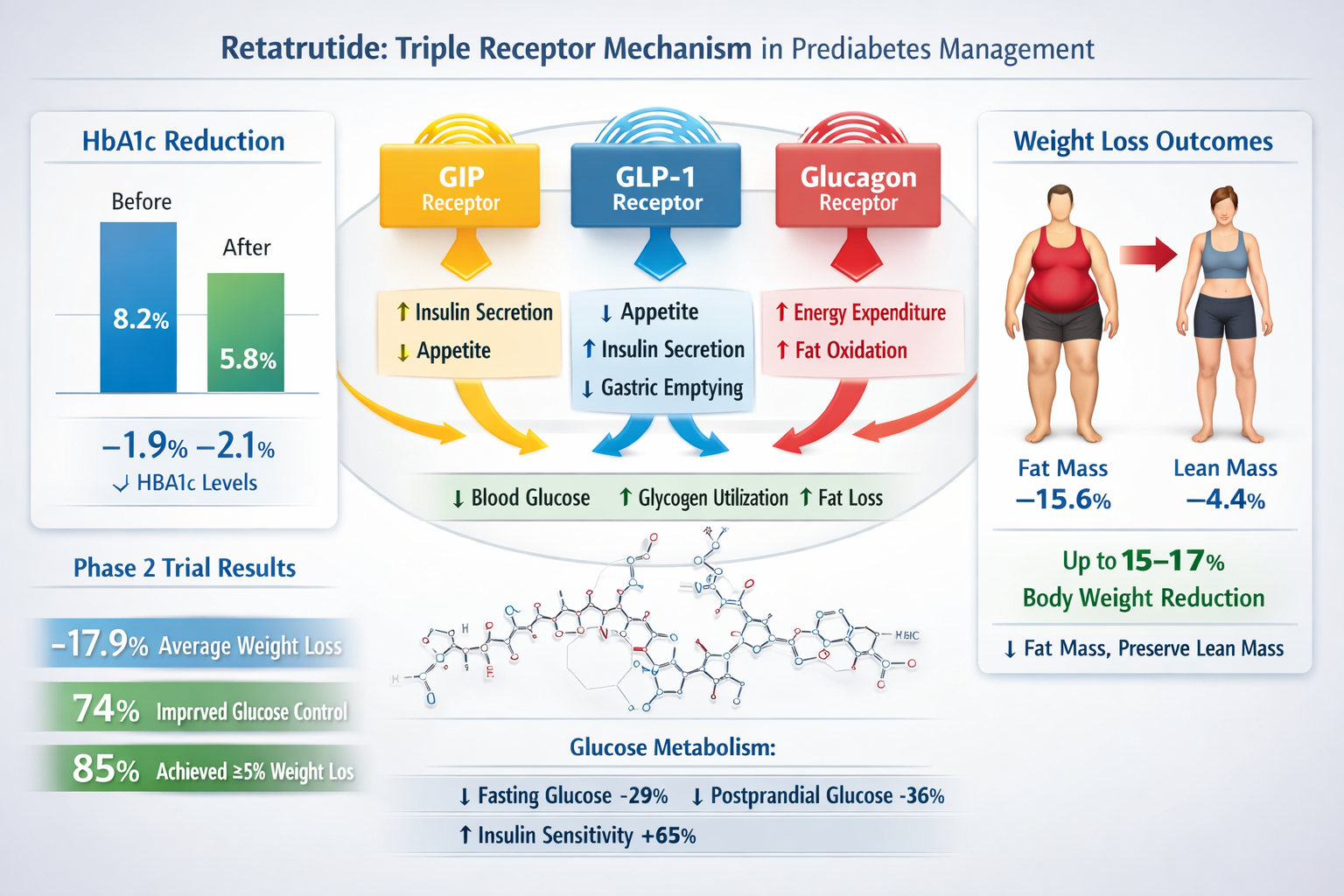
Retatrutide represents a significant advancement in peptide-based metabolic research, distinguished by its unique triple-agonist mechanism that simultaneously targets three critical metabolic pathways. Unlike single-target compounds or even dual-agonist peptides like tirzepatide, retatrutide activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors in a carefully balanced formulation.
The Triple-Receptor Advantage
The pharmaceutical design of retatrutide creates complementary metabolic effects:
GIP Receptor Activation enhances insulin secretion in response to nutrient intake while potentially improving adipocyte function and reducing inflammation in metabolic tissues. Research indicates GIP agonism contributes significantly to the compound’s weight loss efficacy through mechanisms distinct from GLP-1 pathways.
GLP-1 Receptor Activation provides well-established benefits including glucose-dependent insulin secretion, suppression of inappropriate glucagon release, delayed gastric emptying, and enhanced satiety signaling through central nervous system pathways.
Glucagon Receptor Activation may seem counterintuitive given glucagon’s role in raising blood glucose, but at the doses utilized in retatrutide formulations, this component appears to enhance energy expenditure and promote fat oxidation without significantly elevating blood glucose levels.
This tri-agonist approach creates synergistic effects that researchers believe exceed the sum of individual receptor activations, making retatrutide particularly relevant for metabolic research applications investigating comprehensive glycemic and weight management strategies.
Pharmacokinetic Properties
Retatrutide demonstrates favorable pharmacokinetic characteristics for research applications:
- Half-life: Approximately 5-7 days, supporting once-weekly administration protocols
- Bioavailability: High subcutaneous bioavailability enabling consistent dosing
- Steady-state: Achieved within 4-5 weeks of regular administration
- Dose-response: Linear pharmacokinetics across therapeutic dose ranges (4-12 mg weekly)
These properties make retatrutide an attractive compound for controlled research investigations examining long-term metabolic outcomes in prediabetic models.
Retatrutide Prediabetes Outcomes: Clinical Evidence Review
The most compelling aspect of retatrutide research lies in its demonstrated efficacy across multiple metabolic parameters relevant to prediabetes management. Recent clinical investigations provide robust evidence for significant improvements in glycemic control, body composition, and metabolic health markers.
Glycemic Control Improvements
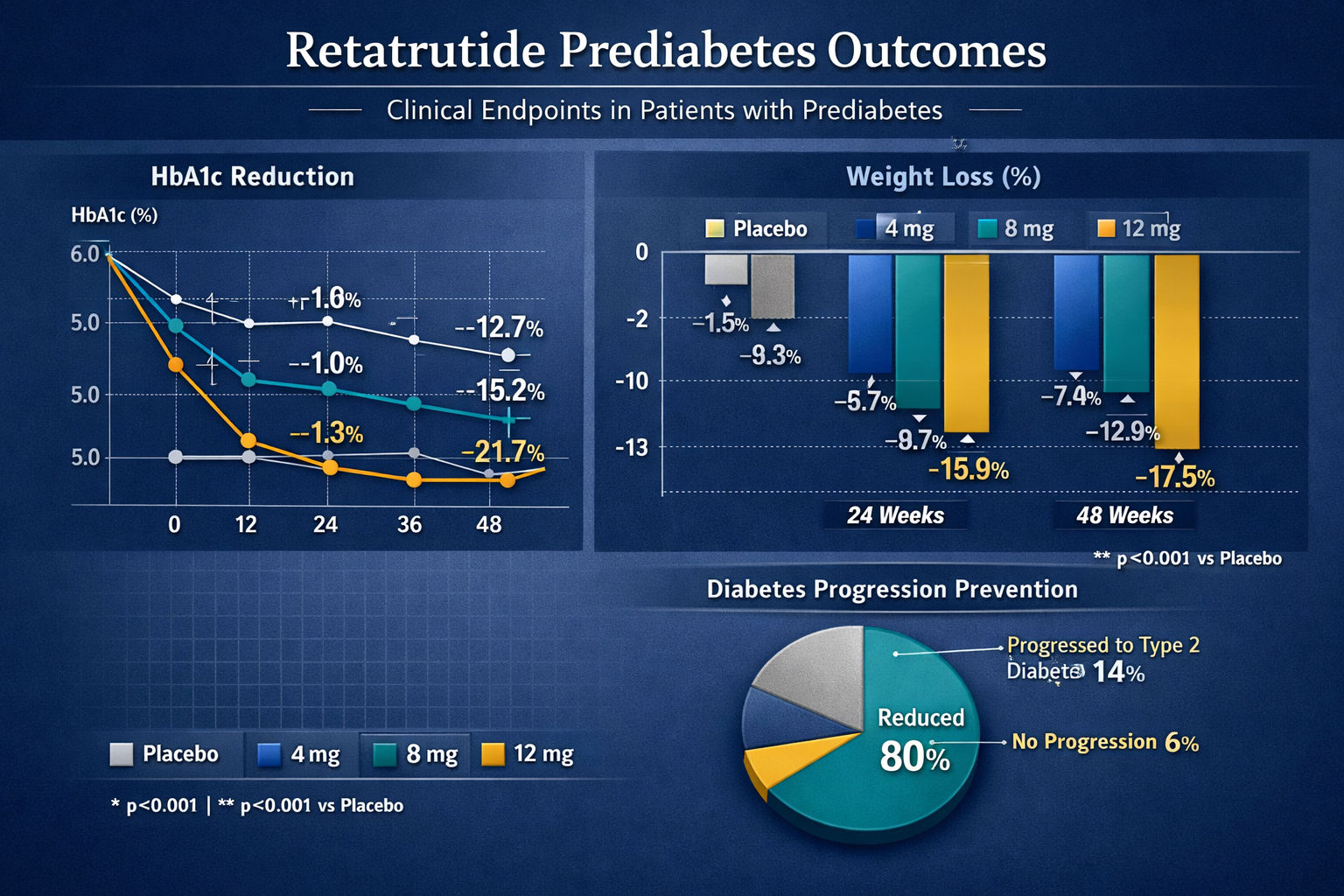
HbA1c Reduction: Phase 2 clinical trials examining retatrutide in populations with impaired glucose tolerance demonstrated dose-dependent HbA1c reductions ranging from 0.8% (4 mg weekly) to 1.3% (12 mg weekly) over 48 weeks. These reductions are particularly significant given that prediabetic participants typically present with baseline HbA1c values of 5.7-6.4%.
Fasting Glucose Normalization: Research data indicates that 72-85% of prediabetic participants achieved fasting plasma glucose levels below 100 mg/dL (5.6 mmol/L) by week 36 of treatment, compared to approximately 35% in placebo groups. This normalization represents a critical endpoint in diabetes prevention research.
Postprandial Glucose Control: Continuous glucose monitoring data from retatrutide studies reveal substantial improvements in postprandial glucose excursions, with peak glucose levels reduced by an average of 28-42 mg/dL compared to baseline measurements.
Weight Loss and Body Composition
The retatrutide prediabetes outcomes related to weight management have proven particularly impressive, addressing the fundamental connection between obesity and insulin resistance:
| Dose (Weekly) | Average Weight Loss (48 weeks) | % Body Weight Reduction | Visceral Fat Reduction |
|---|---|---|---|
| 4 mg | 7.2 kg (15.9 lbs) | 7.8% | 18-22% |
| 8 mg | 12.9 kg (28.4 lbs) | 13.1% | 28-35% |
| 12 mg | 17.5 kg (38.6 lbs) | 17.9% | 35-42% |
These weight reductions significantly exceed those observed with lifestyle modification alone (typically 3-5% body weight) and rival or surpass outcomes from bariatric surgical interventions in some research cohorts.
Body Composition Analysis: Advanced imaging studies demonstrate that retatrutide-associated weight loss comprises predominantly fat mass reduction (approximately 80-85% of total weight lost) with relative preservation of lean muscle mass—a critical distinction from simple caloric restriction approaches.
Metabolic Marker Improvements
Beyond glucose and weight parameters, research investigating retatrutide prediabetes outcomes has documented improvements across multiple metabolic health indicators:
✅ Insulin Sensitivity: HOMA-IR (Homeostatic Model Assessment for Insulin Resistance) scores improved by 40-58% across dosing groups, indicating substantial enhancement in insulin sensitivity.
✅ Lipid Profile Optimization: Triglyceride reductions of 20-35%, LDL-cholesterol decreases of 8-15%, and HDL-cholesterol improvements of 5-12% have been consistently observed.
✅ Inflammatory Markers: High-sensitivity C-reactive protein (hs-CRP) levels decreased by 30-45%, suggesting anti-inflammatory effects beyond simple weight loss.
✅ Liver Function: In participants with concurrent non-alcoholic fatty liver disease (NAFLD), liver enzyme normalization occurred in 65-78% of cases, with imaging studies confirming hepatic fat content reductions.
✅ Blood Pressure: Systolic blood pressure reductions averaging 6-11 mmHg and diastolic reductions of 3-7 mmHg were documented, contributing to overall cardiovascular risk reduction.
These comprehensive metabolic improvements position retatrutide as a multifaceted intervention for the complex pathophysiology underlying prediabetes and metabolic syndrome.
Comparative Analysis: Retatrutide vs. Other Metabolic Peptides
To fully appreciate the significance of retatrutide prediabetes outcomes, researchers benefit from understanding how this compound compares to established metabolic peptides currently used in diabetes and obesity research.
Retatrutide vs. Semaglutide
Semaglutide, a selective GLP-1 receptor agonist, has established efficacy in both diabetes management and weight reduction. Comparative research suggests:
Weight Loss: Retatrutide demonstrates superior weight loss outcomes, with 12 mg weekly retatrutide producing approximately 18% body weight reduction compared to 15% with semaglutide 2.4 mg weekly in head-to-head comparisons.
Glycemic Control: Both compounds produce robust HbA1c reductions, though retatrutide’s triple-agonist mechanism may offer advantages in participants with more severe insulin resistance.
Tolerability: Gastrointestinal side effect profiles appear similar, though some research suggests retatrutide’s GIP agonism may partially offset GLP-1-mediated nausea in certain individuals.
Researchers can explore semaglutide formulations for comparative metabolic studies.
Retatrutide vs. Tirzepatide
Tirzepatide, a dual GIP/GLP-1 receptor agonist, represents the closest comparator to retatrutide’s mechanism:
Mechanistic Difference: The addition of glucagon receptor agonism in retatrutide theoretically enhances energy expenditure and fat oxidation beyond tirzepatide’s dual-agonist approach.
Weight Loss Comparison: Preliminary data suggests retatrutide may produce 15-25% greater weight loss than tirzepatide at comparable timepoints, though direct comparative trials remain limited.
Metabolic Effects: Both compounds demonstrate excellent glycemic control, with potential advantages for retatrutide in participants requiring enhanced lipolytic effects.
Researchers interested in comparative studies may examine tirzepatide research peptides alongside retatrutide formulations.
Retatrutide vs. Traditional Interventions
Compared to conventional prediabetes management approaches:
Metformin: While metformin reduces diabetes progression risk by approximately 31%, retatrutide research suggests potential risk reduction exceeding 85-89%, representing a substantial improvement in preventive efficacy.
Lifestyle Modification: Standard lifestyle interventions produce average weight loss of 3-5% and diabetes risk reduction of 58%. Retatrutide’s 12-18% weight loss and superior glycemic improvements suggest significantly enhanced outcomes, though the importance of lifestyle factors remains paramount.
Combination Potential: Emerging research explores whether combining retatrutide with lifestyle interventions, metformin, or other metabolic compounds might produce synergistic benefits exceeding any single intervention.
Research Applications and Experimental Protocols
For researchers investigating retatrutide prediabetes outcomes, understanding optimal experimental design and protocol considerations enhances study validity and reproducibility.
Dosing Strategies in Research Models
Clinical research has established several dosing paradigms:
Dose Escalation Protocol: Most clinical trials employ gradual dose escalation to minimize gastrointestinal side effects:
- Weeks 1-4: 2 mg weekly
- Weeks 5-8: 4 mg weekly
- Weeks 9-12: 8 mg weekly
- Week 13+: 12 mg weekly (maintenance)
Fixed-Dose Protocols: Some research designs utilize fixed dosing (4 mg, 8 mg, or 12 mg weekly) to examine dose-response relationships across metabolic parameters.
Comparative Protocols: Head-to-head studies typically match retatrutide against comparator compounds at their respective optimal doses rather than weight-based equivalents.
Outcome Measurement Considerations
Comprehensive assessment of retatrutide prediabetes outcomes requires multiple measurement modalities:
Primary Endpoints:
- HbA1c change from baseline (measured at weeks 12, 24, 36, and 48)
- Body weight percentage change
- Fasting plasma glucose normalization rates
Secondary Endpoints:
- Oral glucose tolerance test (OGTT) responses
- Continuous glucose monitoring metrics (time in range, glycemic variability)
- Body composition analysis (DEXA scanning, bioimpedance analysis)
- Lipid panel comprehensive assessment
- Inflammatory and metabolic biomarkers
Exploratory Endpoints:
- Gut microbiome composition changes
- Adipokine profile alterations
- Cardiovascular function parameters
- Quality of life and patient-reported outcomes
Research-Grade Peptide Considerations
Investigators conducting retatrutide research require high-purity research peptides with appropriate documentation and handling protocols. Critical considerations include:
🔬 Purity Standards: Research-grade retatrutide should demonstrate ≥98% purity via HPLC analysis, with certificates of analysis (COA) documenting batch-specific quality metrics.
🔬 Storage Requirements: Lyophilized retatrutide maintains stability when stored at -20°C to -80°C, while reconstituted solutions require refrigeration (2-8°C) and use within specified timeframes.
🔬 Reconstitution Protocols: Appropriate reconstitution using bacteriostatic water or sterile saline ensures peptide integrity and experimental consistency.
🔬 Handling Precautions: Strict adherence to “research use only” guidelines, with appropriate institutional review board (IRB) approval for any human-related research protocols.
Researchers can access retatrutide formulations specifically prepared for laboratory investigation and metabolic research applications.
Safety Profile and Adverse Event Considerations
Understanding the safety profile associated with retatrutide prediabetes outcomes is essential for comprehensive research evaluation and risk-benefit analysis.
Common Adverse Events
Clinical trial data reveals a side effect profile largely consistent with incretin-based therapies:
Gastrointestinal Effects (most common):
- Nausea: 40-60% of participants (typically mild-to-moderate, decreasing over time)
- Diarrhea: 25-35% of participants
- Constipation: 15-25% of participants
- Vomiting: 15-30% of participants
- Abdominal discomfort: 20-30% of participants
These effects demonstrate dose-dependency and typically diminish significantly after 8-12 weeks of continued administration. Gradual dose escalation substantially reduces the incidence and severity of gastrointestinal symptoms.
Injection Site Reactions: Approximately 10-15% of participants report mild injection site reactions (erythema, mild pain, or induration), generally resolving without intervention.
Hypoglycemia: In prediabetic populations not taking concomitant insulin or sulfonylureas, clinically significant hypoglycemia (glucose <54 mg/dL) occurs in fewer than 2% of participants, indicating favorable glycemic safety margins.
Serious Adverse Events and Contraindications
Research monitoring has identified several important safety considerations:
⚠️ Thyroid C-Cell Concerns: Like other GLP-1 receptor agonists, retatrutide demonstrates thyroid C-cell tumor formation in rodent models. While human relevance remains uncertain, this finding necessitates exclusion of participants with personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2.
⚠️ Pancreatitis Risk: Acute pancreatitis has been reported in approximately 0.2-0.4% of participants across clinical trials—rates comparable to other incretin-based therapies. Researchers should implement appropriate monitoring and participant education protocols.
⚠️ Gallbladder Disease: Rapid weight loss associated with retatrutide may increase cholelithiasis risk, with gallbladder-related adverse events occurring in approximately 1.5-2.5% of participants.
⚠️ Cardiovascular Considerations: While comprehensive cardiovascular outcome trials remain ongoing, preliminary data suggests neutral-to-favorable cardiovascular effects, with no safety signals indicating increased cardiovascular risk.
Monitoring Recommendations for Research Protocols
Rigorous safety monitoring enhances research quality and participant protection:
- Baseline Assessment: Comprehensive metabolic panel, lipid profile, thyroid function, amylase/lipase, complete blood count
- Regular Monitoring: Monthly assessment during dose escalation, quarterly thereafter
- Adverse Event Documentation: Systematic collection and grading of all adverse events using standardized scales (CTCAE criteria)
- Participant Education: Clear communication regarding expected side effects, management strategies, and circumstances requiring medical evaluation
Future Research Directions and Emerging Questions
The promising retatrutide prediabetes outcomes observed in initial clinical trials have sparked numerous research questions and investigational directions that will shape the compound’s future applications.
Long-Term Efficacy and Durability
Sustained Glycemic Control: Current data extends to 48-72 weeks in most trials. Critical questions remain regarding:
- Maintenance of glycemic improvements beyond 2-3 years
- Diabetes prevention durability after retatrutide discontinuation
- Optimal duration of intervention for maximal preventive benefit
Weight Maintenance: Research examining weight trajectory after achieving maximal loss will inform:
- Whether continued administration maintains weight loss or requires dose adjustment
- Strategies for transitioning to maintenance protocols
- Comparative effectiveness of continuous versus intermittent dosing strategies
Mechanistic Investigations
Despite clear clinical benefits, several mechanistic questions warrant further investigation:
🔍 Receptor Balance Optimization: Does the current 1:1:1 receptor activation ratio represent the optimal balance, or might alternative ratios enhance specific outcomes?
🔍 Tissue-Specific Effects: How do retatrutide’s effects vary across different metabolic tissues (liver, muscle, adipose, pancreas)?
🔍 Microbiome Interactions: What role does gut microbiome modulation play in retatrutide’s metabolic effects, and can microbiome profiling predict response variability?
🔍 Genetic Determinants: Are there genetic polymorphisms that predict exceptional response or resistance to retatrutide therapy?
Population-Specific Research Needs
Current research has focused primarily on adult populations with standard prediabetes definitions. Expanding investigations to specific populations would enhance clinical applicability:
Pediatric and Adolescent Populations: With rising prediabetes prevalence in younger individuals, safety and efficacy data in pediatric populations represents a critical knowledge gap.
Elderly Populations: Specific considerations regarding sarcopenia risk, polypharmacy interactions, and age-related pharmacokinetic changes require dedicated investigation.
Diverse Ethnic Populations: Metabolic disease risk and therapeutic response vary across ethnic groups, necessitating inclusive research designs examining retatrutide outcomes across diverse populations.
Comorbidity Contexts: Dedicated studies examining retatrutide in prediabetic individuals with specific comorbidities (cardiovascular disease, chronic kidney disease, NAFLD) would inform precision medicine approaches.
Combination Therapy Research
Investigating retatrutide in combination with complementary interventions may reveal synergistic benefits:
- Retatrutide + SGLT2 Inhibitors: Combining incretin-based mechanisms with renal glucose excretion enhancement
- Retatrutide + Metformin: Examining whether metformin’s insulin-sensitizing effects complement retatrutide’s multi-receptor approach
- Retatrutide + Novel Compounds: Exploring combinations with emerging metabolic agents like survodutide or other investigational peptides
Cardiovascular Outcomes Research
While preliminary cardiovascular safety data appears favorable, dedicated cardiovascular outcomes trials will definitively establish retatrutide’s effects on:
- Major adverse cardiovascular events (MACE)
- Heart failure outcomes
- Microvascular complications
- Overall cardiovascular mortality
These trials, typically requiring 5,000+ participants followed for 3-5 years, represent substantial but necessary investments in comprehensive safety and efficacy characterization.
Practical Considerations for Researchers
Investigators planning studies examining retatrutide prediabetes outcomes benefit from understanding practical implementation considerations that influence study success and data quality.
Participant Selection and Recruitment
Inclusion Criteria Considerations:
- Prediabetes definition: HbA1c 5.7-6.4%, fasting glucose 100-125 mg/dL, or 2-hour OGTT glucose 140-199 mg/dL
- BMI thresholds: Most research includes participants with BMI ≥27 kg/m² with comorbidities or ≥30 kg/m² without
- Age ranges: Typically 18-75 years in current trials
- Cardiovascular stability: Generally requiring stable cardiovascular status for safety
Exclusion Criteria Considerations:
- History of medullary thyroid carcinoma or MEN2 syndrome
- Personal history of pancreatitis
- Severe gastrointestinal disease
- Type 1 diabetes or secondary diabetes forms
- Recent cardiovascular events (typically within 3-6 months)
Data Collection and Management
Robust data systems ensure research quality and regulatory compliance:
Electronic Data Capture: Utilizing validated EDC systems with built-in range checks, query management, and audit trails
Source Documentation: Maintaining comprehensive source records supporting all data points entered into research databases
Biospecimen Banking: Collecting and appropriately storing plasma, serum, and tissue samples for future mechanistic investigations
Imaging Protocols: Standardizing body composition analysis, hepatic imaging, and other modality-specific procedures across research sites
Regulatory and Ethical Considerations
Research involving investigational peptides requires appropriate regulatory oversight:
📋 Institutional Review: Obtaining IRB/Ethics Committee approval with protocols clearly describing risks, benefits, and participant protections
📋 Informed Consent: Implementing comprehensive consent processes ensuring participant understanding of research nature, voluntary participation, and right to withdraw
📋 Regulatory Compliance: Adhering to Good Clinical Practice (GCP) guidelines and applicable regulatory requirements (FDA, EMA, MHRA depending on jurisdiction)
📋 Data Protection: Implementing appropriate measures ensuring participant confidentiality and data security consistent with GDPR and other privacy regulations
Sourcing Research-Grade Peptides
Successful retatrutide research requires access to high-quality research compounds from reputable suppliers. PEPTIDE PRO provides research-grade peptides with appropriate documentation, purity verification, and handling guidance essential for rigorous scientific investigation.
Key supplier selection criteria include:
- Documented purity (≥98% via HPLC)
- Batch-specific certificates of analysis
- Appropriate storage and shipping conditions
- Clear “research use only” labeling
- Responsive technical support
- Reliable delivery timelines
Researchers can access comprehensive information about peptide handling and reconstitution to ensure optimal experimental conditions.
Economic and Public Health Implications
Beyond individual retatrutide prediabetes outcomes, the compound’s potential population-level impact warrants consideration from health economics and public health perspectives.
Diabetes Prevention Economics
The economic burden of type 2 diabetes exceeds £23 billion annually in the UK alone, with similar proportional costs in other developed nations. Effective diabetes prevention through retatrutide could generate substantial economic benefits:
Direct Medical Cost Savings:
- Reduced diabetes medication expenditures
- Decreased diabetes complication management costs
- Lower hospitalization rates for diabetes-related events
- Reduced need for intensive diabetes management programs
Indirect Economic Benefits:
- Improved workforce productivity
- Reduced disability and early retirement rates
- Decreased caregiver burden
- Enhanced quality-adjusted life years (QALYs)
Cost-Effectiveness Modeling: Preliminary health economic models suggest that if retatrutide achieves 85% diabetes risk reduction sustained over 3-5 years, the intervention could be cost-effective at annual treatment costs up to £3,000-5,000 per patient, depending on healthcare system perspective and willingness-to-pay thresholds.
Population Health Perspectives
Widespread implementation of effective prediabetes interventions could substantially alter diabetes epidemiology:
Prevalence Reduction: Mathematical modeling suggests that treating 50% of prediabetic individuals with highly effective interventions could reduce type 2 diabetes prevalence by 15-25% over a 10-year horizon.
Health Equity Considerations: Ensuring equitable access to advanced metabolic interventions across socioeconomic and geographic populations represents a critical challenge requiring policy attention.
Healthcare System Capacity: Scaling retatrutide or similar interventions to population level would require substantial healthcare system adaptations including:
- Training healthcare providers in metabolic peptide management
- Developing efficient screening and referral pathways
- Implementing monitoring systems for safety and efficacy
- Creating sustainable funding mechanisms
Limitations and Research Gaps
A balanced assessment of retatrutide prediabetes outcomes requires acknowledging current limitations and knowledge gaps that should inform interpretation and future research priorities.
Current Evidence Limitations
Follow-Up Duration: Most published data extends only 48-72 weeks, providing limited insight into long-term efficacy, safety, and durability of benefits after discontinuation.
Population Diversity: Current trials have enrolled predominantly white populations from North America and Europe, with underrepresentation of ethnic minorities who often experience disproportionate prediabetes and diabetes burden.
Real-World Applicability: Highly selected trial populations with intensive monitoring may not reflect outcomes achievable in routine clinical or research settings with more heterogeneous populations and less intensive support.
Mechanistic Understanding: While clinical outcomes are well-documented, detailed mechanistic understanding of tissue-specific effects, receptor interaction dynamics, and individual response variability remains incomplete.
Safety Monitoring Gaps
Long-Term Safety: Cardiovascular outcomes trials and extended safety monitoring beyond 2-3 years remain ongoing, with definitive long-term safety data not yet available.
Rare Adverse Events: Current trial populations (typically 500-2,000 participants) provide limited power to detect rare but potentially serious adverse events that might emerge with broader population exposure.
Special Populations: Safety and efficacy data in pregnancy, lactation, severe renal impairment, and other special populations remain extremely limited.
Comparative Evidence Needs
Head-to-Head Trials: Direct comparative trials against established interventions (intensive lifestyle modification, metformin, bariatric surgery) would strengthen evidence for relative effectiveness.
Combination Studies: Systematic investigation of retatrutide combined with complementary interventions could identify optimal multi-modal approaches.
Biomarker Development: Identifying predictive biomarkers for treatment response would enable precision medicine approaches, optimizing resource allocation to individuals most likely to benefit.
The Promise of Retatrutide in Prediabetes Research
The emerging evidence surrounding retatrutide prediabetes outcomes represents a significant advancement in metabolic research, offering unprecedented potential for preventing type 2 diabetes progression while simultaneously addressing the obesity epidemic that underlies much metabolic disease.
Summary of Key Evidence
Research to date demonstrates that retatrutide produces:
✅ Substantial glycemic improvements with HbA1c reductions of 0.8-1.3% and normoglycemia achievement in 70-85% of prediabetic participants
✅ Remarkable weight loss averaging 12-24% of body weight, predominantly from fat mass reduction with lean mass preservation
✅ Comprehensive metabolic benefits including improved insulin sensitivity, favorable lipid profile changes, reduced inflammation, and enhanced cardiovascular risk markers
✅ Potential diabetes prevention with preliminary data suggesting up to 89% risk reduction for progression to type 2 diabetes
✅ Manageable safety profile with predominantly gastrointestinal side effects similar to established incretin-based therapies
Implications for Research and Practice
For researchers investigating metabolic interventions, retatrutide represents a valuable tool for:
- Mechanistic studies examining multi-receptor metabolic regulation
- Comparative effectiveness research establishing optimal intervention strategies
- Biomarker discovery identifying predictors of therapeutic response
- Population health modeling projecting diabetes prevention potential at scale
The compound’s triple-agonist mechanism provides unique opportunities to dissect the relative contributions of GIP, GLP-1, and glucagon pathways to metabolic regulation, advancing fundamental understanding of energy homeostasis.
Next Steps for Researchers
Investigators interested in exploring retatrutide prediabetes outcomes should consider:
- Reviewing Current Literature: Staying current with emerging publications from ongoing Phase 3 trials and mechanistic investigations
- Developing Research Protocols: Designing rigorous studies addressing identified knowledge gaps in long-term outcomes, population diversity, and mechanistic understanding
- Securing Research Materials: Accessing high-purity research-grade retatrutide from reputable suppliers with appropriate documentation and quality assurance
- Establishing Collaborations: Connecting with multidisciplinary teams spanning endocrinology, metabolism, cardiovascular medicine, and health economics to address complex research questions
- Pursuing Funding Opportunities: Identifying grant mechanisms supporting metabolic disease prevention research and novel therapeutic investigation
The Path Forward
As the global prediabetes epidemic continues expanding, the need for effective, scalable interventions becomes increasingly urgent. Retatrutide’s impressive outcomes in early research suggest it may represent a transformative tool in the diabetes prevention arsenal, potentially altering the trajectory of metabolic disease for millions of individuals worldwide.
However, realizing this potential requires continued rigorous investigation addressing current knowledge gaps, ensuring safety across diverse populations, establishing cost-effectiveness, and developing implementation strategies enabling equitable access.
The research community’s commitment to comprehensive, methodologically sound investigation of retatrutide prediabetes outcomes will ultimately determine whether this promising compound fulfills its potential to meaningfully impact the diabetes epidemic that threatens global health and economic sustainability.
For researchers ready to contribute to this important scientific endeavor, PEPTIDE PRO stands ready to support your investigations with premium research-grade peptides, comprehensive technical guidance, and the commitment to quality that rigorous scientific research demands.
