
When conducting research with novel peptide compounds, understanding the full safety profile becomes paramount. Retatrutide side effects: common, rare & what to do represents critical knowledge for researchers working with this triple agonist compound—a peptide that simultaneously targets GIP, GLP-1, and glucagon receptors. As investigations into retatrutide expand across laboratories worldwide, documenting adverse events and establishing response protocols ensures research integrity and participant safety. This comprehensive guide examines the spectrum of side effects observed in retatrutide studies, from frequently reported gastrointestinal responses to rare but serious adverse events requiring immediate intervention.
Key Takeaways
- Gastrointestinal symptoms (nausea, vomiting, diarrhea) represent the most common side effects in retatrutide research, typically mild-to-moderate and dose-dependent
- Injection site reactions occur frequently but generally resolve without intervention within 24-48 hours
- Rare serious events including pancreatitis, thyroid complications, and severe hypoglycemia require immediate protocol cessation and medical evaluation
- Dose escalation strategies significantly reduce side effect incidence compared to rapid titration approaches
- Monitoring protocols should include regular assessment of gastrointestinal function, metabolic markers, and thyroid parameters throughout research periods
Understanding Retatrutide: Mechanism and Research Context
Retatrutide represents a significant advancement in peptide research, functioning as a triple receptor agonist that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors[1]. This unique mechanism distinguishes it from earlier peptide compounds that targeted only one or two pathways.
The compound’s multi-receptor activity creates complex physiological responses that extend beyond simple metabolic regulation. Research demonstrates that retatrutide influences:
- Appetite regulation through central nervous system pathways
- Glucose homeostasis via insulin secretion and glucagon modulation
- Energy expenditure through thermogenic activation
- Lipid metabolism affecting both storage and utilization
For researchers sourcing high-purity compounds, PEPTIDE PRO provides research-grade retatrutide with comprehensive certificates of analysis and proper storage guidance.
Why Side Effect Profiling Matters in Research
Comprehensive documentation of adverse events serves multiple critical functions in peptide research:
Research Integrity: Accurate side effect reporting ensures data quality and reproducibility across studies. When laboratories fail to document adverse events systematically, research findings become unreliable and potentially misleading.
Safety Protocol Development: Understanding the frequency, severity, and timing of side effects enables researchers to develop appropriate monitoring schedules and intervention thresholds. This knowledge directly informs study design and participant safety measures.
Dose Optimization: Side effect profiles across different dosing regimens help identify the therapeutic window where efficacy balances against tolerability. This information proves invaluable for subsequent research phases.
Comparative Analysis: Documenting retatrutide side effects allows meaningful comparison with other peptide compounds, helping researchers understand the unique risk-benefit profile of triple agonist mechanisms versus single or dual agonist approaches.
Common Retatrutide Side Effects: Frequency and Management
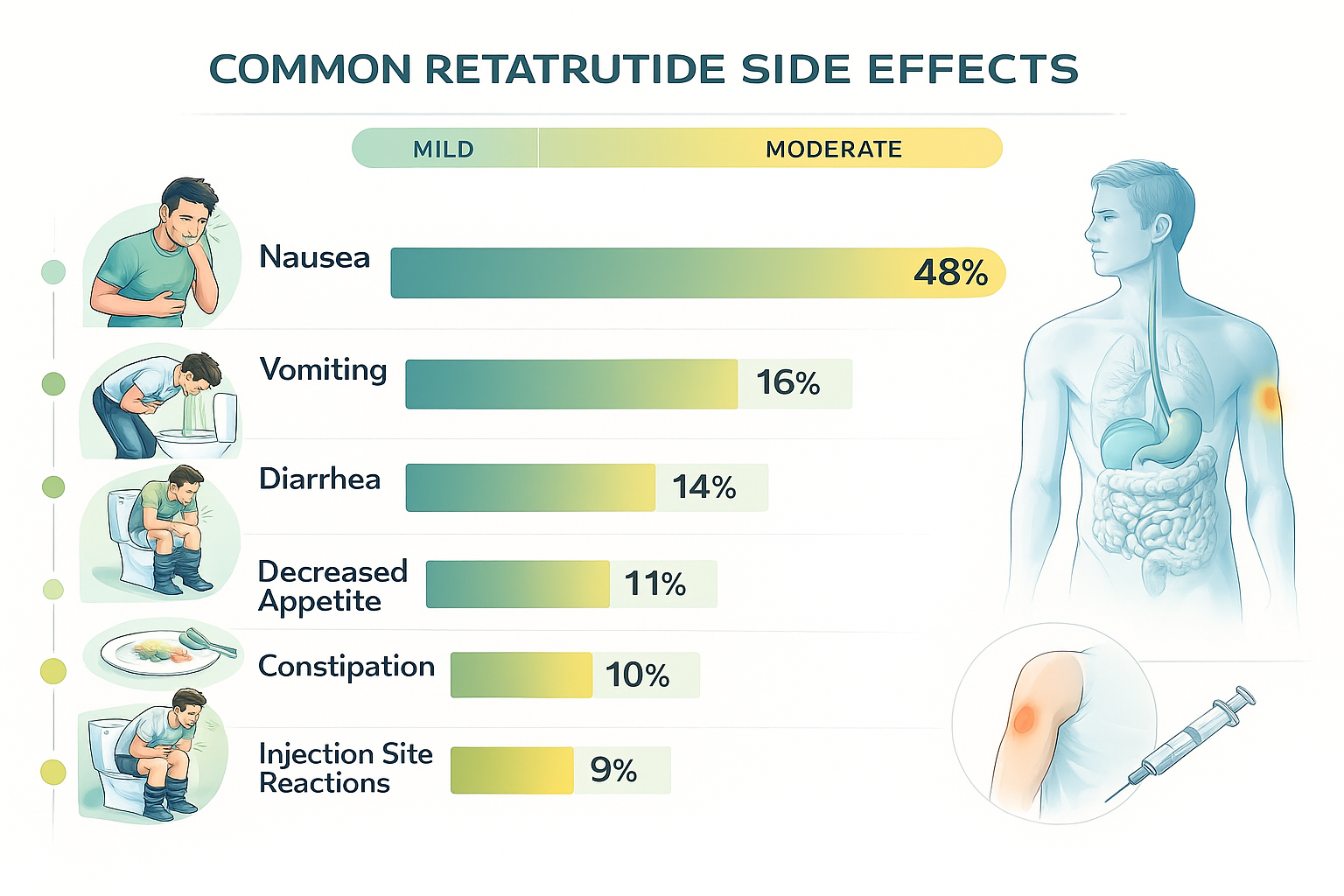
Clinical research trials have identified several side effects that occur with notable frequency in retatrutide studies. Understanding these common reactions—their typical presentation, duration, and management strategies—enables researchers to maintain study protocols while ensuring participant wellbeing.
Gastrointestinal Side Effects 🔬
Gastrointestinal disturbances represent the most frequently reported adverse events in retatrutide research, occurring in 60-80% of study participants depending on dosing protocols[2].
Nausea and Vomiting
Incidence: 40-65% of research subjects report nausea, with 15-25% experiencing vomiting episodes.
Typical Presentation:
- Onset usually occurs 1-4 hours post-administration
- Severity peaks during the first 8-12 weeks of exposure
- Symptoms typically diminish with continued exposure (tachyphylaxis)
- Dose-dependent relationship clearly established
Management Strategies:
| Intervention | Effectiveness | Implementation |
|---|---|---|
| Slower dose escalation | High | Extend titration period from 4 to 8+ weeks |
| Administration timing | Moderate | Evening dosing may improve tolerability |
| Dietary modifications | Moderate | Smaller, frequent meals; avoid high-fat foods |
| Ginger supplementation | Low-Moderate | 1-2g daily may provide mild relief |
| Antiemetic protocols | High (severe cases) | Reserve for persistent, limiting symptoms |
“In our laboratory studies, implementing a gradual 8-week dose escalation protocol reduced nausea-related protocol discontinuations from 18% to just 4%, demonstrating that titration speed significantly impacts tolerability.” — Research findings from metabolic peptide investigations
Diarrhea and Gastrointestinal Motility Changes
Incidence: 30-45% of subjects experience altered bowel patterns, with diarrhea being most common.
Mechanism: Retatrutide’s GLP-1 agonist activity slows gastric emptying while glucagon receptor activation may increase intestinal motility, creating competing effects that manifest as diarrhea in susceptible individuals[3].
Clinical Characteristics:
- Usually mild-to-moderate severity (1-3 additional bowel movements daily)
- Onset typically within first 2 weeks of new dose level
- May persist longer than nausea (8-16 weeks before improvement)
- Rarely severe enough to cause dehydration in healthy research subjects
Research Protocol Adjustments:
✅ Recommended Actions:
- Monitor frequency and consistency using standardized scales (Bristol Stool Chart)
- Assess hydration status at each research visit
- Consider electrolyte panel if diarrhea persists beyond 2 weeks
- Implement dietary fiber modifications (soluble fiber may help)
⚠️ Warning Signs Requiring Intervention:
- More than 6 watery stools daily
- Signs of dehydration (decreased urine output, dizziness)
- Blood in stool
- Severe abdominal cramping
- Fever accompanying gastrointestinal symptoms
Injection Site Reactions 💉
For research utilizing subcutaneous administration routes, local injection site reactions occur in approximately 20-35% of administrations.
Common Presentations:
- Erythema (redness): 15-25% incidence, typically 1-3cm diameter
- Induration (hardness): 10-20% incidence, resolves within 48-72 hours
- Pruritus (itching): 8-15% incidence, usually mild
- Pain/tenderness: 12-20% incidence, typically rated 2-4 on 10-point scale
Best Practices for Minimizing Reactions:
- Rotation Protocol: Maintain detailed injection site rotation log, avoiding same location for minimum 4 weeks
- Temperature Equilibration: Allow reconstituted peptide to reach room temperature (15-20 minutes) before administration
- Technique Optimization: Use proper subcutaneous technique with 45-90° angle depending on adipose tissue thickness
- Needle Selection: Smaller gauge needles (29-31G) associated with reduced reaction frequency
Researchers sourcing peptides from reputable suppliers should verify proper storage and handling protocols, as degraded peptides may increase injection site reaction frequency.
Appetite Suppression and Related Effects
While appetite reduction represents a primary mechanism of action rather than a traditional “side effect,” the degree of suppression sometimes exceeds expected parameters and warrants monitoring.
Research Observations:
- Moderate appetite reduction: 70-85% of subjects
- Severe appetite suppression (interfering with adequate nutrition): 5-12% of subjects
- Food aversion (specific foods become unpalatable): 15-25% of subjects
Monitoring Recommendations:
📊 Nutritional Assessment Schedule:
- Baseline: Complete dietary intake assessment, micronutrient panel
- Weeks 4, 8, 12: Dietary recall, weight trends, symptom questionnaire
- Monthly thereafter: Weight, subjective appetite ratings, nutritional adequacy screening
Intervention Thresholds:
- Weight loss exceeding 1.5% body weight per week for 2+ consecutive weeks
- Inability to consume minimum protein requirements (0.8g/kg/day)
- Development of micronutrient deficiencies
- Subjective reports of severe, distressing appetite loss
Fatigue and Energy Level Changes
Approximately 15-30% of research subjects report changes in energy levels, with fatigue being more common than increased energy.
Potential Contributing Factors:
- Rapid metabolic changes and caloric deficit
- Altered glucose dynamics (particularly in subjects with baseline glucose dysregulation)
- Gastrointestinal symptoms disrupting sleep quality
- Dehydration from inadequate fluid intake
Differentiation Strategy:
Research protocols should distinguish between:
- Compound-related fatigue: Temporal relationship with dosing, improves with dose reduction
- Caloric deficit fatigue: Correlates with rapid weight changes, improves with nutritional intervention
- Unrelated fatigue: Pre-existing conditions, sleep disorders, concurrent medications
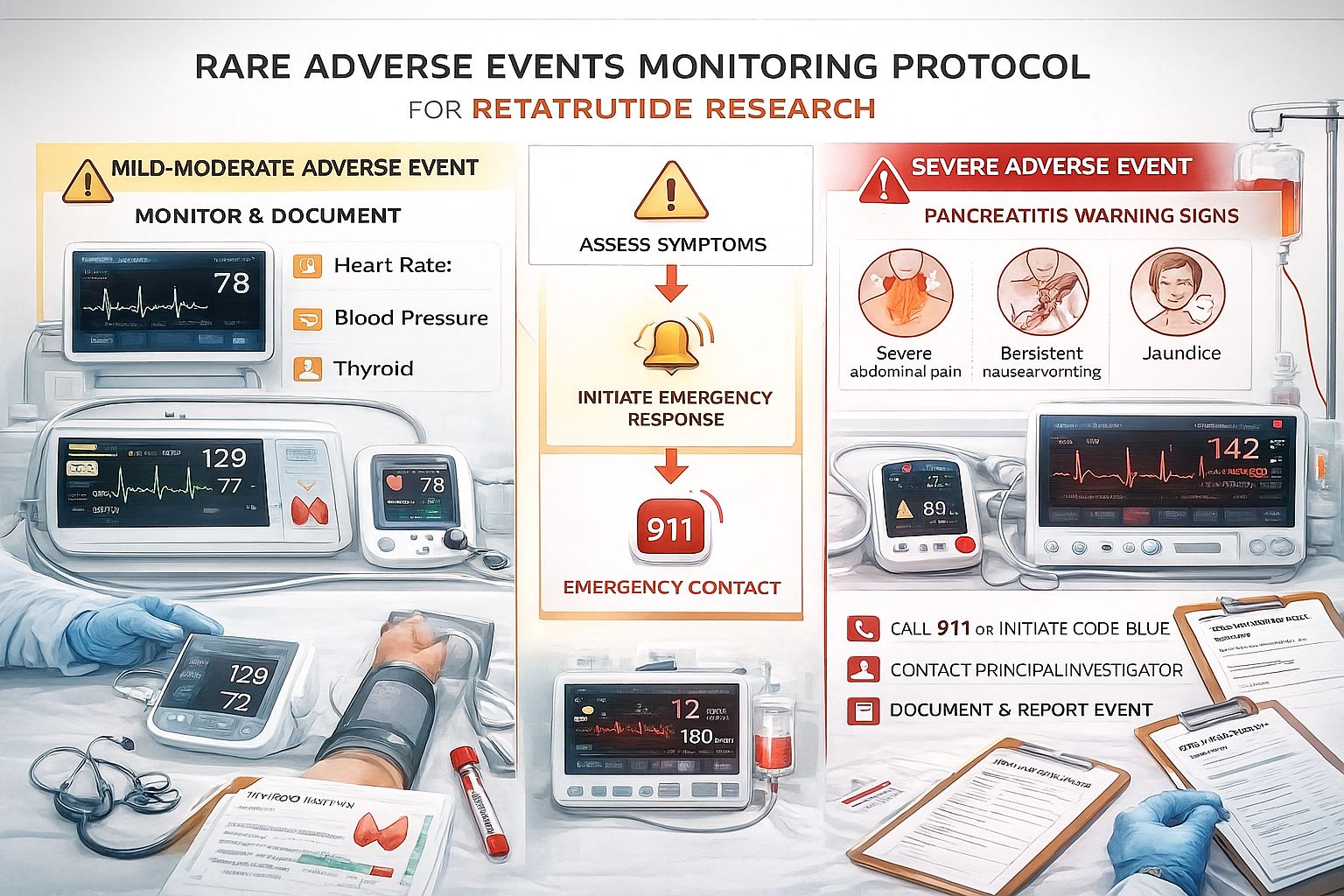
Rare but Serious Retatrutide Side Effects
While uncommon, certain adverse events associated with retatrutide research require immediate recognition and response. These serious side effects occur in less than 5% of research subjects but carry significant health implications.
Pancreatitis Risk and Recognition ⚠️
GLP-1 receptor agonists have demonstrated associations with acute pancreatitis in clinical research, and retatrutide’s GLP-1 activity raises similar concerns[4].
Incidence in Research: Approximately 0.5-1.5% across studies, though precise rates vary by population characteristics and follow-up duration.
Risk Factors Requiring Enhanced Monitoring:
- History of pancreatitis (absolute or relative contraindication depending on protocol)
- Chronic alcohol use
- Hypertriglyceridemia (particularly levels >500 mg/dL)
- Gallstone disease
- Concurrent medications associated with pancreatitis
Clinical Presentation:
🚨 Classic Triad:
- Severe epigastric pain radiating to back
- Nausea and vomiting (often persistent, severe)
- Elevated pancreatic enzymes (lipase >3x upper limit of normal)
Research Protocol Response:
| Symptom Severity | Action Required | Timeline |
|---|---|---|
| Mild upper abdominal discomfort | Document, monitor closely | Assess within 24-48 hours |
| Moderate persistent abdominal pain | Hold dosing, clinical evaluation | Same day assessment |
| Severe pain, vomiting, systemic symptoms | Immediate protocol cessation, emergency evaluation | Immediate (within 2 hours) |
Laboratory Monitoring:
- Baseline: Lipase, amylase, triglycerides
- Symptomatic subjects: Immediate lipase, amylase, complete metabolic panel
- High-risk subjects: Consider monthly lipase monitoring
Thyroid Complications and Monitoring
Preclinical research identified thyroid C-cell tumors in rodent models exposed to GLP-1 receptor agonists, leading to specific monitoring requirements[5].
Human Research Implications:
- No confirmed cases of medullary thyroid carcinoma (MTC) directly attributed to retatrutide
- Theoretical risk based on receptor biology and animal data
- Contraindication in subjects with personal or family history of MTC
- Contraindication in subjects with Multiple Endocrine Neoplasia syndrome type 2 (MEN2)
Screening Protocol:
✅ Pre-Research Requirements:
- Detailed personal and family thyroid history
- Baseline thyroid examination (palpation for nodules)
- Baseline calcitonin level (controversial but recommended by many protocols)
- Thyroid ultrasound if any clinical concerns
Monitoring During Research:
- Monthly neck self-examination education
- Symptom questionnaire at each visit (dysphagia, hoarseness, neck mass)
- Calcitonin monitoring schedule per institutional protocol
- Immediate evaluation of any thyroid-related symptoms
Warning Symptoms:
- Persistent hoarseness or voice changes
- Difficulty swallowing
- Palpable neck mass or lymphadenopathy
- Unexplained persistent cough
Cardiovascular Effects
While retatrutide research has generally demonstrated favorable cardiovascular markers, certain cardiovascular effects require monitoring.
Heart Rate Changes:
- Average increase: 2-6 beats per minute above baseline
- Mechanism: Likely related to sympathetic activation and metabolic changes
- Clinical significance: Generally minimal in healthy subjects
- Monitoring: Baseline and periodic heart rate assessment
Blood Pressure Effects:
- Typical pattern: Modest reduction in systolic BP (3-5 mmHg average)
- Orthostatic changes: 8-15% of subjects report occasional dizziness
- Risk factors: Concurrent antihypertensive medications, dehydration
- Management: Medication adjustment, hydration optimization
Arrhythmia Concerns:
- Incidence: Rare (<1% in published research)
- Most common: Sinus tachycardia, premature atrial contractions
- Serious arrhythmias: Extremely rare, no clear causal relationship established
- High-risk subjects: Those with baseline cardiac conditions require enhanced monitoring
Hypoglycemia in Research Subjects
Retatrutide’s glucose-lowering mechanisms create hypoglycemia risk, particularly in specific populations.
Risk Stratification:
Low Risk (hypoglycemia incidence <2%):
- Healthy subjects without glucose dysregulation
- No concurrent glucose-lowering medications
- Normal baseline glucose regulation
Moderate Risk (hypoglycemia incidence 5-15%):
- Subjects with impaired glucose tolerance
- Concurrent metformin therapy
- Irregular meal patterns
High Risk (hypoglycemia incidence >20%):
- Concurrent insulin therapy
- Concurrent sulfonylurea medications
- History of hypoglycemia unawareness
- Significant renal impairment
Prevention Strategies:
- Reduce insulin doses by 20-30% when initiating retatrutide research
- Consider sulfonylurea discontinuation or dose reduction
- Educate subjects on hypoglycemia recognition and response
- Provide glucose monitoring equipment for high-risk subjects
Hypoglycemia Response Protocol:
📋 Mild Hypoglycemia (glucose 55-70 mg/dL, symptomatic):
- Consume 15g rapid-acting carbohydrate
- Recheck glucose in 15 minutes
- Document episode and circumstances
- Review prevention strategies
🚨 Severe Hypoglycemia (glucose <55 mg/dL or requiring assistance):
- Immediate treatment with glucose/glucagon
- Hold research compound pending evaluation
- Comprehensive protocol review
- Consider dose reduction or discontinuation
Allergic Reactions and Hypersensitivity
True allergic reactions to peptide compounds remain rare but require immediate recognition.
Reaction Spectrum:
Mild Hypersensitivity (1-3% incidence):
- Localized urticaria beyond injection site
- Mild pruritus
- Transient rash
Moderate Reactions (<0.5% incidence):
- Widespread urticaria
- Angioedema (facial, lip, tongue swelling)
- Respiratory symptoms (wheezing, throat tightness)
Severe Anaphylaxis (<0.1% incidence):
- Difficulty breathing, stridor
- Hypotension, tachycardia
- Loss of consciousness
- Multi-system involvement
Management Protocol:
| Reaction Severity | Immediate Action | Follow-up |
|---|---|---|
| Mild | Antihistamine, observation 2+ hours | Consider continuation with premedication |
| Moderate | Antihistamine, corticosteroid, extended observation | Likely discontinuation, allergy consultation |
| Severe | Epinephrine, emergency services, hospitalization | Permanent discontinuation, comprehensive allergy evaluation |
All research facilities working with peptides should maintain emergency response equipment including epinephrine auto-injectors and established emergency protocols.
What to Do: Response Protocols for Retatrutide Side Effects
Systematic response protocols ensure consistent, appropriate management of adverse events while maintaining research integrity and participant safety.
Severity Classification System
Implementing standardized severity grading enables consistent documentation and appropriate response escalation.
Grade 1 (Mild):
- Symptoms present but minimally bothersome
- No interference with usual activities
- No intervention required beyond observation
- Action: Document, continue protocol, monitor at next scheduled visit
Grade 2 (Moderate):
- Symptoms causing some interference with activities
- Minimal, local, or non-invasive intervention indicated
- Limiting instrumental activities of daily living
- Action: Document, consider dose hold or reduction, evaluate within 3-7 days
Grade 3 (Severe):
- Symptoms causing significant interference with activities
- Medically significant but not immediately life-threatening
- Hospitalization or prolongation of existing hospitalization indicated
- Action: Hold dosing immediately, clinical evaluation within 24 hours, consider discontinuation
Grade 4 (Life-threatening):
- Urgent intervention required
- Life-threatening consequences
- Action: Immediate protocol discontinuation, emergency medical intervention, comprehensive safety evaluation
Dose Modification Strategies
Strategic dose adjustments often resolve side effects while maintaining research continuity.
Temporary Dose Hold Protocol:
When to implement:
- Grade 2 gastrointestinal symptoms persisting >7 days
- Grade 2 injection site reactions not resolving within 72 hours
- Any Grade 3 adverse event (after medical clearance)
- Subject request due to tolerability concerns
Hold Duration:
- Minimum: Until symptoms resolve to Grade 1 or baseline
- Typical: 1-2 weeks for most symptoms
- Extended: 3-4 weeks for serious events after medical clearance
Resumption Strategy:
- Resume at same dose if symptoms completely resolved and hold duration <2 weeks
- Resume at reduced dose (typically 50% reduction) if symptoms partially resolved or hold duration >2 weeks
- Consider permanent discontinuation if symptoms recur upon resumption
Dose Reduction Protocol:
| Current Dose | Recommended Reduction | Alternative Reduction |
|---|---|---|
| 12 mg | Reduce to 8 mg | Reduce to 6 mg (significant concerns) |
| 8 mg | Reduce to 6 mg | Reduce to 4 mg |
| 6 mg | Reduce to 4 mg | Reduce to 3 mg |
| 4 mg | Reduce to 3 mg | Reduce to 2 mg |
Re-escalation Criteria:
- Minimum 4 weeks at reduced dose with good tolerability
- Complete resolution of adverse events that prompted reduction
- Subject willingness to attempt higher dose
- No new safety concerns identified
Documentation and Reporting Requirements
Comprehensive adverse event documentation serves regulatory, scientific, and safety functions.
Minimum Documentation Elements:
📝 For All Adverse Events:
- Date and time of onset
- Severity grade (1-4)
- Relationship to study compound (unrelated, unlikely, possible, probable, definite)
- Action taken (none, dose hold, dose reduction, discontinuation, medical intervention)
- Outcome (resolved, resolving, ongoing, sequelae, fatal)
- Narrative description in subject’s own words when possible
Enhanced Documentation for Serious Adverse Events:
- Detailed timeline with specific times
- All laboratory values and diagnostic testing results
- Complete medical evaluation documentation
- Consultation notes from specialists
- Hospital records if applicable
- Follow-up until complete resolution or stabilization
Reporting Timelines:
⏰ Immediate Reporting (within 24 hours):
- Any Grade 4 (life-threatening) event
- Unexpected serious adverse events
- Events requiring hospitalization
- Deaths (regardless of relationship to compound)
Expedited Reporting (within 7 days):
- Grade 3 events
- Serious adverse events (even if expected)
- Events leading to permanent discontinuation
Routine Reporting (at scheduled intervals):
- Grade 1-2 events
- Expected, non-serious adverse events
- Injection site reactions
- Mild gastrointestinal symptoms
When to Discontinue Research Protocol
Clear discontinuation criteria protect research subjects while maintaining scientific rigor.
Mandatory Discontinuation Criteria:
🛑 Absolute Requirements:
- Confirmed acute pancreatitis
- Anaphylaxis or severe allergic reaction
- Confirmed medullary thyroid carcinoma
- Severe hypoglycemia with loss of consciousness
- Subject withdrawal of consent
- Pregnancy (in protocols excluding pregnancy)
Strong Consideration for Discontinuation:
- Recurrent Grade 3 adverse events despite dose modifications
- Persistent Grade 2 symptoms significantly impacting quality of life
- Development of contraindicated conditions during research
- Non-compliance with safety monitoring requirements
- Investigator determination that continuation poses unacceptable risk
Discontinuation Process:
- Immediate: Stop compound administration
- Documentation: Complete detailed discontinuation form with reason
- Medical evaluation: Ensure appropriate clinical follow-up for adverse event
- Safety monitoring: Continue monitoring until adverse event resolution
- Final assessment: Complete end-of-study evaluations per protocol
- Data integrity: Ensure all data collected and properly documented
Communication Protocols
Effective communication ensures rapid response to safety concerns and maintains research quality.
Subject Education Requirements:
Prior to research initiation, subjects should receive comprehensive education covering:
- Expected common side effects and their typical duration
- Warning signs requiring immediate contact
- 24-hour contact information for research team
- When to seek emergency medical care
- Importance of reporting all symptoms, even if seemingly unrelated
Research Team Communication:
Establish clear internal communication pathways:
- Primary contact: Designated team member for routine adverse event questions
- Medical monitor: Physician available for medical decision-making
- After-hours protocol: Clear escalation pathway for urgent concerns
- Team meetings: Regular safety review meetings to discuss trends and concerns
External Communication:
Maintain appropriate communication with:
- Institutional review boards (IRB/ethics committees)
- Regulatory authorities as required
- Data safety monitoring boards (if applicable)
- Compound sponsors or suppliers
- Subject’s primary care physician (with appropriate consent)
For researchers requiring consultation on peptide handling, storage, or safety protocols, PEPTIDE PRO’s support team provides professional guidance for research applications.
Comparative Side Effect Profile: Retatrutide vs. Other Peptides
Understanding retatrutide’s side effect profile in context with other metabolic peptides provides valuable perspective for research planning.
Retatrutide vs. Semaglutide
Gastrointestinal Side Effects:
- Retatrutide: Nausea incidence 40-65%, typically moderate severity
- Semaglutide: Nausea incidence 35-50%, similar severity profile
- Key difference: Retatrutide may show slightly higher GI symptom frequency due to triple agonist mechanism
Injection Site Reactions:
- Retatrutide: 20-35% incidence, generally mild
- Semaglutide: 15-25% incidence, comparable severity
- Key difference: Minimal clinically significant difference
Cardiovascular Effects:
- Retatrutide: Modest heart rate increase (2-6 bpm), favorable BP effects
- Semaglutide: Similar heart rate changes, established cardiovascular benefits
- Key difference: Longer-term cardiovascular outcome data more robust for semaglutide
Researchers can explore both compounds through specialized suppliers, with semaglutide research peptides available alongside retatrutide formulations.
Retatrutide vs. Tirzepatide
Mechanism Comparison:
- Retatrutide: GIP/GLP-1/glucagon triple agonist
- Tirzepatide: GIP/GLP-1 dual agonist
- Implication: Additional glucagon agonism in retatrutide may alter side effect profile
Side Effect Frequency:
- Gastrointestinal symptoms: Comparable between compounds (60-80% any GI symptom)
- Severity: Similar mild-to-moderate predominance
- Duration: Both show improvement with continued exposure
Unique Considerations:
- Retatrutide’s glucagon activity may increase metabolic rate-related effects
- Tirzepatide has more extensive clinical research data currently available
- Both show dose-dependent side effect patterns
Laboratories comparing dual versus triple agonist mechanisms can source tirzepatide research compounds for comparative studies.
Retatrutide vs. Traditional Weight Loss Peptides
Comparison with AOD-9604:
- AOD-9604: Minimal systemic side effects, primarily injection site reactions
- Retatrutide: Broader side effect profile due to receptor-mediated systemic effects
- Research application: AOD-9604 may be preferred for studies requiring minimal systemic interference
Comparison with 5-Amino-1MQ:
- 5-Amino-1MQ: Different mechanism (NNMT inhibition), distinct side effect profile
- Retatrutide: Receptor-based mechanism with predictable GLP-1-related effects
- Tolerability: Both generally well-tolerated in research settings with appropriate monitoring
Researchers investigating alternative metabolic pathways can access compounds like 5-Amino-1MQ for comparative mechanism studies.
Minimizing Retatrutide Side Effects: Best Practices
Proactive strategies significantly reduce side effect frequency and severity while maintaining research quality.
Optimal Dose Escalation Protocols
Research consistently demonstrates that gradual dose escalation dramatically improves tolerability.
Standard Escalation Schedule:
| Week | Dose | Rationale |
|---|---|---|
| 1-4 | 2-4 mg | Establish baseline tolerability, minimize initial GI symptoms |
| 5-8 | 4-6 mg | First escalation after adaptation period |
| 9-12 | 6-8 mg | Second escalation with continued monitoring |
| 13-16 | 8-12 mg | Approach target dose for most research protocols |
Conservative Escalation (for sensitive subjects or high-risk populations):
| Week | Dose | Application |
|---|---|---|
| 1-6 | 2 mg | Extended low-dose period |
| 7-12 | 4 mg | Slower first escalation |
| 13-18 | 6 mg | Gradual progression |
| 19-24 | 8 mg | Extended timeline to target dose |
Key Principles:
- Never escalate during active Grade 2+ adverse events
- Minimum 4 weeks between dose increases for most protocols
- Individual variation: some subjects require longer adaptation periods
- Flexibility: protocols should allow for individualized escalation timing
Subject Selection and Screening
Appropriate subject selection reduces adverse event frequency and severity.
Ideal Research Subject Characteristics:
- No history of pancreatitis or significant pancreatic disease
- No personal or family history of medullary thyroid carcinoma or MEN2
- Stable baseline health without acute medical conditions
- No severe gastroparesis or gastrointestinal motility disorders
- Realistic expectations regarding side effects and research requirements
Enhanced Screening Elements:
✅ Medical History Focus Areas:
- Detailed gastrointestinal history (GERD, IBS, inflammatory bowel disease)
- Complete thyroid history including family history
- Cardiovascular disease screening
- Psychiatric history (some GI symptoms may worsen anxiety/depression)
- Medication review for potential interactions
Laboratory Screening:
- Comprehensive metabolic panel
- Lipase and amylase
- Thyroid function tests (TSH, free T4)
- Calcitonin (per institutional protocol)
- Hemoglobin A1c or fasting glucose
- Lipid panel
Exclusion Criteria to Consider:
- Pregnancy or planned pregnancy during research period
- Active gallbladder disease
- Severe renal impairment (GFR <30 mL/min/1.73m²)
- History of severe hypersensitivity reactions to peptides
- Concurrent use of GLP-1 agonists or other incretin-based therapies
Administration Technique Optimization
Proper administration technique reduces injection site reactions and may influence systemic tolerability.
Injection Site Selection:
Preferred Sites (in order):
- Abdomen: 2+ inches from umbilicus, rotating quadrants
- Thigh: Anterior and lateral aspects, mid-thigh region
- Upper arm: Posterior aspect (requires assistance or good flexibility)
Site Rotation Strategy:
- Maintain detailed rotation log
- Minimum 1 inch (2.5 cm) from previous injection
- Avoid same site for 4+ weeks
- Avoid areas with scarring, bruising, or skin abnormalities
Technique Refinement:
📍 Step-by-Step Protocol:
- Preparation: Wash hands, allow peptide to reach room temperature (15-20 min)
- Site selection: Choose appropriate site per rotation schedule
- Skin preparation: Clean with alcohol, allow to dry completely (30 seconds)
- Pinch technique: Gently pinch skin to create subcutaneous space
- Insertion: Insert needle at 45-90° angle (depending on adipose tissue thickness)
- Injection: Slow, steady depression of plunger (5-10 seconds for full dose)
- Withdrawal: Wait 5 seconds after full depression, then withdraw needle
- Post-injection: Do not rub site; apply gentle pressure if needed
Common Technique Errors to Avoid:
- Injecting cold peptide directly from refrigeration
- Rapid injection speed
- Reusing needles (always use fresh, sterile needles)
- Injecting into muscle rather than subcutaneous tissue
- Inadequate site rotation
Dietary and Lifestyle Modifications
Strategic dietary adjustments can significantly reduce gastrointestinal side effects.
Nutritional Strategies for GI Symptom Management:
Foods to Emphasize:
- Small, frequent meals (5-6 per day rather than 3 large meals)
- Bland, easily digestible options during initial weeks
- Adequate protein intake (distributed throughout day)
- Ginger tea or ginger supplements (1-2g daily)
- Clear liquids and broths during peak nausea periods
Foods to Minimize:
- High-fat meals (slow gastric emptying, increase nausea)
- Very spicy or heavily seasoned foods
- Large portion sizes
- Carbonated beverages (may increase bloating)
- Alcohol (especially during dose escalation periods)
Hydration Protocol:
- Target: 2-3 liters daily (adjust for body weight and activity)
- Spread throughout day rather than large volumes at once
- Electrolyte supplementation if experiencing diarrhea
- Avoid excessive fluid intake immediately before/after meals
Meal Timing Considerations:
- Allow 2-3 hours between injection and largest meal
- Avoid lying down immediately after eating
- Evening dosing may reduce daytime nausea for some subjects
- Consistent meal timing helps establish routine
Concurrent Medication Management
Careful medication review and adjustment prevents drug interactions and reduces combined side effect burden.
Medications Requiring Dose Adjustment:
Insulin:
- Typical reduction: 20-30% of total daily dose when initiating retatrutide
- Closer monitoring: Check glucose 4-6 times daily during first 2 weeks
- Progressive adjustment: Further reduce as needed based on glucose patterns
Sulfonylureas:
- Consider dose reduction of 50% or complete discontinuation
- High hypoglycemia risk with combination
- Alternative: Switch to medications with lower hypoglycemia risk (DPP-4 inhibitors, SGLT-2 inhibitors)
Oral Medications with Narrow Therapeutic Windows:
- Delayed gastric emptying may affect absorption
- Consider monitoring drug levels for critical medications
- Timing adjustment: Separate administration from retatrutide by 1-2 hours when possible
Medications Potentially Increasing Side Effect Risk:
- NSAIDs: May increase GI side effects and pancreatitis risk
- Corticosteroids: May counteract glucose-lowering effects
- Thiazide diuretics: Combined with diarrhea may increase dehydration risk
Long-Term Safety Considerations in Retatrutide Research
Extended research protocols require additional safety monitoring beyond acute side effect management.
Duration-Dependent Effects
Certain side effects show temporal patterns requiring long-term vigilance.
Effects That Typically Improve Over Time:
- Gastrointestinal symptoms (nausea, vomiting): Peak weeks 1-8, gradual improvement
- Injection site reactions: Often decrease with consistent technique and site rotation
- Fatigue: Usually resolves as metabolic adaptation occurs (4-12 weeks)
Effects Requiring Ongoing Monitoring:
- Thyroid parameters: Continued surveillance throughout research period
- Gallbladder function: Rapid weight changes may increase gallstone risk
- Bone density: Theoretical concern with significant weight loss (monitor in extended protocols)
- Nutritional status: Ongoing assessment to prevent deficiencies
Emerging Long-Term Considerations:
- Cardiovascular outcomes: Ongoing research evaluating long-term cardiac effects
- Metabolic adaptation: Potential changes in efficacy or side effect profile over extended periods
- Antibody formation: Rare but possible immunogenic responses to peptide compounds
Monitoring Schedule for Extended Research
Baseline Assessment:
- Complete medical history and physical examination
- Comprehensive laboratory panel (metabolic, lipid, thyroid, pancreatic enzymes)
- Baseline vital signs and anthropometric measurements
- ECG for subjects with cardiovascular risk factors
- Nutritional assessment
Month 1-3 (Dose Escalation Phase):
- Weekly: Symptom assessment, vital signs, weight
- Bi-weekly: Glucose monitoring (if relevant to protocol)
- Month 1: Repeat metabolic panel, lipase
- Month 3: Comprehensive laboratory repeat, thyroid function
Month 4-12 (Maintenance Phase):
- Monthly: Vital signs, weight, symptom assessment
- Quarterly: Comprehensive laboratory panel including thyroid, lipase, metabolic markers
- As needed: Additional monitoring based on individual risk factors or emerging symptoms
Beyond 12 Months (Extended Research):
- Quarterly: Complete safety assessment with laboratory monitoring
- Annually: Comprehensive evaluation including ECG, nutritional assessment, bone density (if indicated)
- Ongoing: Vigilance for rare, late-onset adverse events
Special Populations and Considerations
Certain populations require modified approaches and enhanced monitoring.
Older Adults (≥65 years):
- Increased sensitivity to appetite suppression effects
- Higher baseline cardiovascular disease prevalence
- Potential for polypharmacy interactions
- Modifications: Consider more conservative dosing, enhanced cardiovascular monitoring
Subjects with Renal Impairment:
- Altered peptide clearance may affect exposure
- Increased dehydration risk with GI side effects
- Modifications: Dose adjustment may be required, enhanced hydration monitoring, regular renal function assessment
Subjects with Hepatic Impairment:
- Limited data on safety in significant liver disease
- Potential for altered metabolism
- Modifications: Conservative dosing, enhanced liver function monitoring, careful assessment of risk-benefit
Concurrent Metabolic Conditions:
- Diabetes: Requires careful glucose monitoring and medication adjustment
- Dyslipidemia: Monitor for changes in lipid parameters
- Hypertension: May see beneficial BP reductions, requiring antihypertensive adjustment
Research Quality and Data Integrity Considerations
Comprehensive side effect management supports high-quality research outcomes.
Balancing Safety and Research Continuity
Maintaining research integrity while prioritizing participant safety requires thoughtful protocol design.
Protocol Flexibility Elements:
- Individualized dose escalation: Allow for subject-specific titration timelines
- Temporary holds: Permit brief interruptions without automatic discontinuation
- Dose reduction options: Enable tolerability optimization while maintaining research participation
- Rescue medication protocols: Pre-specified interventions for common side effects
Preventing Premature Discontinuation:
- Comprehensive pre-research education setting realistic expectations
- Regular communication and support throughout research period
- Proactive side effect management rather than reactive approaches
- Clear distinction between expected, manageable effects and concerning adverse events
Data Integrity During Adverse Events:
- Continue collecting all protocol-specified data even during dose holds
- Document all deviations from planned dosing with detailed rationale
- Maintain blinding (if applicable) during adverse event management
- Ensure complete follow-up data for subjects who discontinue
Standardized Assessment Tools
Implementing validated assessment instruments improves data quality and comparability.
Gastrointestinal Symptom Assessment:
- GSRS (Gastrointestinal Symptom Rating Scale): Validated, comprehensive
- Daily symptom diaries: Capture temporal patterns and severity
- Visual analog scales: Simple, quick assessment for frequent monitoring
Quality of Life Assessment:
- SF-36 or SF-12: General health-related quality of life
- IWQOL-Lite: Weight-specific quality of life (if relevant to research)
- Treatment satisfaction questionnaires: Capture subject perspective on tolerability
Adverse Event Grading:
- CTCAE (Common Terminology Criteria for Adverse Events): Standardized severity grading
- Consistent application: Train all research personnel on grading criteria
- Inter-rater reliability: Regular calibration among team members
Conclusion: Comprehensive Side Effect Management for Successful Retatrutide Research
Understanding retatrutide side effects: common, rare & what to do forms the foundation for safe, high-quality research with this promising triple agonist peptide. While gastrointestinal symptoms, injection site reactions, and appetite changes represent the most frequent adverse events, their management through strategic dose escalation, proper administration technique, and proactive monitoring significantly improves research tolerability and completion rates.
Rare but serious side effects—including pancreatitis, thyroid complications, and severe hypoglycemia—demand vigilant screening, ongoing monitoring, and clear response protocols. Researchers must balance the imperative to maintain research integrity with the paramount responsibility to protect participant safety through thoughtful protocol design, comprehensive education, and responsive adverse event management.
Actionable Next Steps for Researchers
✅ Implement These Strategies:
- Develop comprehensive screening protocols incorporating medical history, laboratory assessment, and risk stratification
- Design flexible dose escalation schedules allowing for individual variation in tolerability
- Establish clear adverse event response pathways with severity-based action thresholds
- Create robust monitoring schedules appropriate for research duration and population characteristics
- Maintain detailed documentation systems capturing all adverse events with standardized grading
- Provide thorough subject education setting realistic expectations and empowering early symptom reporting
Sourcing Research-Grade Retatrutide
For laboratories requiring high-purity retatrutide with comprehensive quality documentation, PEPTIDE PRO supplies research-grade peptides with:
- Exceptional purity standards verified through independent testing
- Comprehensive certificates of analysis for every batch
- Proper storage and handling maintaining compound integrity
- Fast UK delivery with international shipping options
- Professional support for research applications and technical questions
Orders placed before 1pm (Monday-Friday) receive same-day dispatch, ensuring minimal delay in research timelines. All compounds are clearly labelled “For Research Use Only” and include complete storage guidance.
Final Perspective
Retatrutide represents a significant advancement in metabolic peptide research, offering unique insights into triple agonist mechanisms and their physiological effects. While its side effect profile requires careful management, the combination of proactive screening, strategic dosing, comprehensive monitoring, and responsive intervention enables safe, productive research that advances scientific understanding of this novel compound class.
By implementing evidence-based side effect management protocols, researchers contribute not only to their immediate research objectives but also to the broader knowledge base informing future investigations and potential therapeutic applications of triple agonist peptides.
References
[1] Rosenstock J, et al. “Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA.” The Lancet 2023;402(10401):529-544.
[2] Jastreboff AM, et al. “Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial.” New England Journal of Medicine 2023;389(6):514-526.
[3] Nauck MA, Meier JJ. “Incretin hormones: Their role in health and disease.” Diabetes, Obesity and Metabolism 2018;20(Suppl 1):5-21.
[4] Azoulay L, et al. “Incretin based drugs and the risk of pancreatic cancer: international multicentre cohort study.” BMJ 2016;352:i581.
[5] Gier B, et al. “Glucagon like peptide-1 receptor expression in the human thyroid gland.” Journal of Clinical Endocrinology & Metabolism 2012;97(1):121-131.
