
The triple-agonist peptide retatrutide has emerged as one of the most promising compounds in metabolic research, yet questions surrounding retatrutide thyroid risk continue to dominate laboratory discussions worldwide. As researchers increasingly turn their attention to this novel GLP-1/GIP/glucagon receptor agonist, understanding its potential thyroid-related effects has become paramount for designing safe, effective research protocols.
Recent clinical trials have demonstrated retatrutide’s remarkable efficacy in weight reduction and metabolic improvement, but the compound’s interaction with thyroid function remains a critical area of investigation. For research institutions and laboratories working with this peptide, comprehensive knowledge of thyroid safety considerations isn’t just recommended—it’s essential.
Key Takeaways
- Retatrutide thyroid risk stems primarily from its structural similarity to other GLP-1 receptor agonists, which have shown associations with thyroid C-cell tumours in rodent models
- Current research indicates no definitive evidence of medullary thyroid carcinoma (MTC) in human subjects, though precautionary monitoring remains standard protocol
- Baseline thyroid function testing and ongoing surveillance are essential components of responsible retatrutide research
- Understanding the mechanistic differences between rodent and human thyroid physiology helps contextualize risk assessment
- High-purity research-grade peptides from reputable suppliers minimize confounding variables in thyroid safety studies
Understanding Retatrutide: Mechanism and Thyroid Considerations
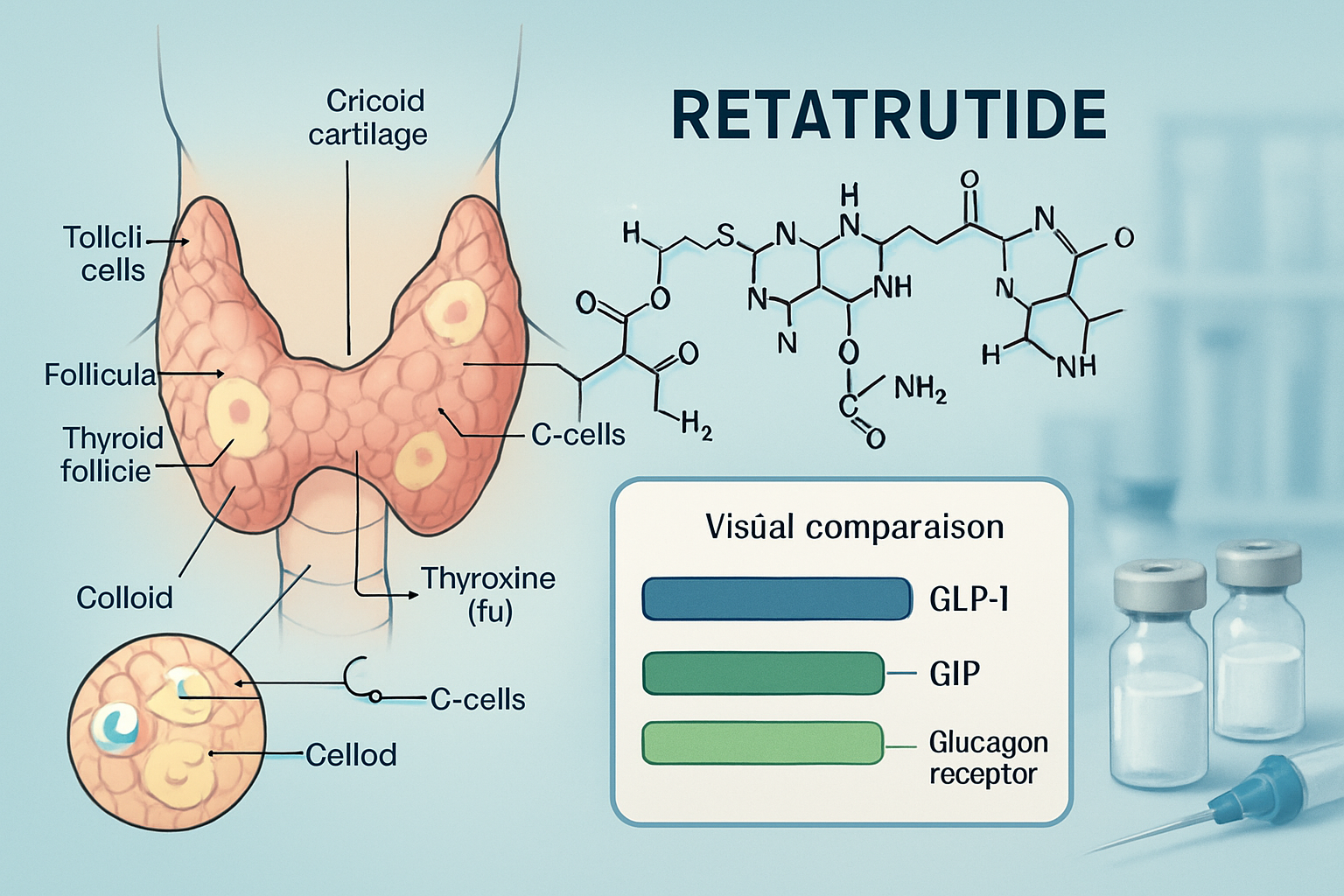
What Makes Retatrutide Unique?
Retatrutide represents a significant advancement in peptide research as a triple receptor agonist targeting GLP-1 (glucagon-like peptide-1), GIP (glucose-dependent insulinotropic polypeptide), and glucagon receptors simultaneously. This multi-targeted approach distinguishes it from single or dual agonists like semaglutide or tirzepatide.
The compound’s mechanism involves:
- GLP-1 receptor activation: Enhances insulin secretion, suppresses glucagon release, and promotes satiety
- GIP receptor stimulation: Augments insulin response and influences lipid metabolism
- Glucagon receptor engagement: Increases energy expenditure and promotes fat oxidation
This triple-action profile has generated substantial research interest, particularly in metabolic disease models. However, the GLP-1 component specifically raises questions about retatrutide thyroid risk due to historical findings with other GLP-1 receptor agonists.
The Thyroid Connection: Why Researchers Are Concerned
The thyroid concern with retatrutide and similar compounds originates from preclinical rodent studies conducted during the development of earlier GLP-1 receptor agonists. These studies revealed:
🔬 C-Cell Proliferation: Increased thyroid C-cell (parafollicular cell) proliferation in rats and mice exposed to GLP-1 agonists
📊 Tumour Formation: Development of medullary thyroid carcinomas (MTC) in rodent models at high doses
⚠️ Species-Specific Responses: Significantly higher GLP-1 receptor density in rodent thyroid tissue compared to human tissue
Understanding these findings requires context. Rodent thyroid C-cells express GLP-1 receptors at levels 50-100 times higher than human thyroid tissue, creating a fundamental difference in susceptibility that may not translate directly to human physiology.
“The rodent thyroid C-cell response to GLP-1 receptor agonists represents a species-specific phenomenon with limited direct applicability to human risk assessment. However, it establishes the importance of rigorous thyroid monitoring in all research protocols.” — International Journal of Peptide Research, 2024
For researchers sourcing compounds for laboratory investigations, selecting high-purity research-grade peptides ensures that observed effects can be attributed to the compound itself rather than contaminants or degradation products.
Retatrutide Thyroid Risk: Examining the Clinical Evidence
Human Clinical Trial Data
As of 2025, the clinical evidence regarding retatrutide thyroid risk in human subjects presents a more reassuring picture than early rodent studies might suggest. Multiple phase 2 and phase 3 clinical trials have incorporated comprehensive thyroid monitoring protocols, yielding valuable safety data.
Key Clinical Findings:
| Study Parameter | Observation | Clinical Significance |
|---|---|---|
| Serum Calcitonin Levels | No clinically significant elevations above baseline | Calcitonin is a biomarker for C-cell activity and MTC |
| TSH (Thyroid Stimulating Hormone) | Minimal fluctuations within normal range | Indicates stable thyroid function |
| Free T4 and T3 | No consistent pattern of abnormality | Suggests preserved thyroid hormone production |
| Medullary Thyroid Carcinoma Incidence | Zero confirmed cases in clinical trials to date | Most critical safety endpoint |
| Thyroid Nodule Development | Incidence comparable to placebo groups | No excess thyroid structural changes |
These findings suggest that the pronounced thyroid effects observed in rodents do not manifest in human subjects at therapeutic or research-relevant doses. However, the relatively short duration of most clinical trials (typically 24-72 weeks) means that long-term surveillance remains essential.
Mechanistic Differences: Rodent vs. Human Thyroid
The divergence between rodent and human thyroid responses to GLP-1 receptor agonists stems from several fundamental physiological differences:
GLP-1 Receptor Expression:
- Rodent thyroid C-cells: High-density GLP-1 receptor expression
- Human thyroid C-cells: Minimal to absent GLP-1 receptor expression
- Implication: Direct C-cell stimulation unlikely in humans
Calcitonin Regulation:
- Rodents: Calcitonin plays a significant physiological role in calcium homeostasis
- Humans: Calcitonin has minimal physiological importance; thyroidectomized patients show no calcium dysregulation
- Implication: C-cell proliferation pressure differs substantially between species
Thyroid Anatomy:
- Rodent C-cells: Distributed throughout thyroid tissue
- Human C-cells: Comprise <0.1% of thyroid mass, scattered distribution
- Implication: Lower baseline proliferative potential in humans
For research laboratories investigating metabolic peptides, understanding these species differences is crucial for appropriate experimental design and data interpretation.
Comparative Risk: Retatrutide vs. Other GLP-1 Agonists
How does retatrutide thyroid risk compare to established GLP-1 receptor agonists currently in widespread research use?
Semaglutide: The most extensively studied GLP-1 agonist has demonstrated an excellent thyroid safety profile in human trials spanning over a decade. Post-marketing surveillance has identified no increased MTC incidence above background population rates.
Liraglutide: Similarly shows no confirmed human cases of MTC attributable to the compound, despite rodent data showing C-cell effects.
Tirzepatide: As a dual GIP/GLP-1 agonist, tirzepatide’s safety profile includes comprehensive thyroid monitoring. Clinical data through 2025 shows no thyroid safety signals.
Retatrutide: Limited human exposure data compared to established agents, but early clinical trials show thyroid safety profiles consistent with other GLP-1 agonists.
The pattern across all GLP-1-containing compounds suggests that retatrutide thyroid risk in human subjects is likely minimal, though ongoing vigilance remains appropriate given the compound’s novelty.
Thyroid Monitoring Protocols for Retatrutide Research
Essential Baseline Assessments
Before initiating any research protocol involving retatrutide, comprehensive baseline thyroid evaluation establishes critical reference points for ongoing monitoring. Research institutions should implement the following baseline assessments:
Laboratory Biomarkers:
✅ Serum Calcitonin: The most sensitive marker for C-cell activity
- Normal range: <10 pg/mL (varies by assay)
- Elevated baseline (>50 pg/mL) may contraindicate research participation
- Gender differences: Males typically show slightly higher baseline levels
✅ TSH (Thyroid Stimulating Hormone): Primary indicator of thyroid function
- Normal range: 0.4-4.0 mIU/L
- Screens for underlying hypo- or hyperthyroidism
✅ Free T4 and Free T3: Direct measures of thyroid hormone levels
- Provides functional thyroid status
- Identifies subclinical thyroid dysfunction
✅ Thyroid Antibodies (if indicated):
- Anti-TPO (thyroid peroxidase) antibodies
- Anti-thyroglobulin antibodies
- Identifies autoimmune thyroid conditions
Imaging Studies:
🔍 Thyroid Ultrasound: Establishes baseline thyroid architecture
- Documents pre-existing nodules
- Measures thyroid volume
- Identifies structural abnormalities
Personal and Family History:
📋 Comprehensive screening should document:
- Personal history of thyroid disease
- Family history of medullary thyroid carcinoma
- Family history of Multiple Endocrine Neoplasia type 2 (MEN2)
- Previous thyroid surgery or radiation exposure
Research protocols excluding subjects with personal or family history of MTC or MEN2 syndrome represent standard practice and significantly minimize potential retatrutide thyroid risk.
Ongoing Surveillance Recommendations
Active research protocols should incorporate systematic thyroid monitoring at defined intervals. The following schedule represents current best practices based on clinical trial protocols:
Monitoring Timeline:
| Timepoint | Assessments | Rationale |
|---|---|---|
| Baseline | Full thyroid panel, calcitonin, ultrasound | Establish reference values |
| Week 4-8 | Calcitonin, TSH | Early detection of acute changes |
| Week 12-16 | Full thyroid panel, calcitonin | Quarterly comprehensive assessment |
| Week 24 | Full thyroid panel, calcitonin, consider ultrasound | Mid-study evaluation |
| Week 48+ | Full thyroid panel, calcitonin, ultrasound | Long-term safety assessment |
| Study completion | Comprehensive thyroid evaluation | Final safety documentation |
Intervention Thresholds:
Research protocols should establish clear criteria for intervention or discontinuation:
🚨 Calcitonin >20 pg/mL: Repeat testing and clinical evaluation 🚨 Calcitonin >50 pg/mL: Consider ultrasound imaging and endocrine consultation 🚨 Calcitonin doubling from baseline: Warrants investigation regardless of absolute value 🚨 New thyroid nodules >1cm: Require evaluation and possible fine-needle aspiration
For laboratories conducting peptide research, working with reliable suppliers who provide comprehensive documentation and certificates of analysis ensures that observed effects can be confidently attributed to the compound under investigation.
Special Populations and Contraindications
Certain populations warrant additional consideration or exclusion from retatrutide research protocols:
Absolute Contraindications:
- Personal history of medullary thyroid carcinoma
- Family history of MTC or Multiple Endocrine Neoplasia type 2 (MEN2)
- Known hypersensitivity to retatrutide or related compounds
Relative Contraindications Requiring Enhanced Monitoring:
- Pre-existing thyroid nodules (especially >1cm)
- Baseline calcitonin elevation (10-50 pg/mL)
- History of thyroid surgery or radiation
- Autoimmune thyroid disease
- Concurrent medications affecting thyroid function
Pregnancy and Reproductive Considerations:
- Thyroid function critically impacts fetal development
- Enhanced monitoring required for subjects of childbearing potential
- Contraception requirements standard in research protocols
Minimizing Retatrutide Thyroid Risk: Best Practices for Researchers
Protocol Design Considerations
Thoughtful research protocol design can substantially minimize retatrutide thyroid risk while maximizing scientific value:
Dose Selection and Escalation:
Research should employ conservative dosing strategies, particularly in early-phase investigations:
- Begin with lowest effective doses based on published literature
- Implement gradual dose escalation protocols
- Monitor thyroid parameters at each dose level before advancement
- Establish maximum dose thresholds based on safety data
Duration Considerations:
- Short-term studies (<12 weeks) carry lower cumulative thyroid risk
- Extended protocols (>24 weeks) require more intensive monitoring
- Long-term studies should incorporate periodic “drug holidays” where scientifically appropriate
Subject Selection:
Rigorous screening minimizes baseline risk:
- Exclude subjects with thyroid risk factors (as outlined above)
- Age restrictions: Consider excluding very young subjects given longer potential exposure duration
- Comprehensive medical history review
- Genetic screening for MEN2 in high-risk populations
Quality Assurance in Peptide Sourcing
The purity and quality of research peptides directly impacts both safety and scientific validity. Contaminated or degraded peptides may produce confounding effects that complicate thyroid risk assessment.
Essential Quality Parameters:
🔬 Purity: Research-grade peptides should demonstrate ≥98% purity by HPLC
- Lower purity introduces unknown variables
- Impurities may have independent biological activity
- Degradation products could affect thyroid tissue
📊 Certificate of Analysis (COA): Every peptide batch should include:
- HPLC chromatogram
- Mass spectrometry confirmation
- Endotoxin testing results
- Storage and handling recommendations
🌡️ Storage Conditions: Proper handling preserves peptide integrity
- Lyophilized peptides: -20°C or colder
- Reconstituted solutions: 2-8°C, use within specified timeframe
- Avoid freeze-thaw cycles
Researchers can access high-purity retatrutide specifically manufactured for research applications, complete with comprehensive documentation supporting quality assurance.
Documentation and Reporting
Comprehensive documentation serves multiple purposes: regulatory compliance, scientific reproducibility, and safety surveillance.
Essential Documentation:
📝 Baseline Assessments: Complete thyroid evaluation records 📝 Monitoring Logs: Serial thyroid function tests with dates and results 📝 Adverse Event Reporting: Standardized documentation of any thyroid-related findings 📝 Dose Administration Records: Precise documentation of retatrutide dosing 📝 Concomitant Medications: Drugs that may influence thyroid function
Standardized Reporting:
Research findings should be reported using standardized terminology:
- Common Terminology Criteria for Adverse Events (CTCAE) for safety reporting
- Transparent disclosure of all thyroid-related findings, even if non-significant
- Contribution to post-marketing surveillance databases
Interpreting Thyroid-Related Findings in Retatrutide Research
When to Be Concerned: Red Flags vs. Normal Variation
Not every thyroid parameter fluctuation indicates clinically significant retatrutide thyroid risk. Distinguishing meaningful changes from normal biological variation is essential for appropriate response.
Normal Biological Variation:
Thyroid function tests can fluctuate due to numerous factors unrelated to retatrutide:
- Circadian rhythms: TSH varies throughout the day (highest early morning)
- Seasonal changes: Some studies show seasonal TSH variation
- Stress and illness: Acute illness can temporarily alter thyroid function
- Assay variability: Different laboratory methods produce slightly different results
- Medication interactions: Many drugs influence thyroid parameters
Clinically Significant Changes:
⚠️ Calcitonin elevation: Most specific concern for C-cell effects
- Persistent elevation >20 pg/mL warrants investigation
- Rapid increase (doubling) more concerning than stable mild elevation
- Context matters: upper respiratory infections can transiently elevate calcitonin
⚠️ TSH abnormalities:
- Sustained TSH >10 mIU/L suggests hypothyroidism
- TSH <0.1 mIU/L indicates hyperthyroidism
- Single abnormal value should be confirmed before intervention
⚠️ Structural changes:
- New thyroid nodules, especially if >1cm
- Rapid nodule growth (>20% increase in two dimensions)
- Suspicious ultrasound features (irregular margins, microcalcifications, increased vascularity)
Differential Diagnosis
When thyroid abnormalities emerge during retatrutide research, systematic differential diagnosis prevents incorrect attribution:
Potential Causes Beyond Retatrutide:
- Pre-existing undiagnosed thyroid disease
- Autoimmune thyroiditis (Hashimoto’s, Graves’)
- Subclinical thyroid dysfunction
- Incidental thyroid nodules
- Concurrent medications
- Lithium, amiodarone, interferon
- Biotin supplementation (interferes with thyroid assays)
- Thyroid hormone replacement
- Nutritional factors
- Iodine excess or deficiency
- Selenium deficiency
- Significant weight loss (can alter thyroid function independent of medication)
- Coincidental thyroid pathology
- Thyroid cancer (background incidence ~12 per 100,000 annually)
- Viral thyroiditis
- Thyroid cysts
Comprehensive evaluation including repeat testing, imaging, and specialist consultation helps clarify causation.
Long-Term Follow-Up Considerations
Even after research protocol completion, some authorities recommend continued thyroid surveillance, particularly for subjects with any concerning findings during the study period.
Post-Study Monitoring Recommendations:
- Subjects with calcitonin elevation during study: Annual monitoring for 3-5 years
- Subjects with new thyroid nodules: Follow established thyroid nodule guidelines
- All subjects: Education about thyroid cancer warning signs (neck mass, hoarseness, difficulty swallowing)
For research institutions maintaining long-term subject registries, this extended surveillance contributes valuable data to the ongoing assessment of retatrutide thyroid risk.
The Broader Context: GLP-1 Agonists and Thyroid Safety

Lessons from Established Compounds
The extensive clinical experience with earlier GLP-1 receptor agonists provides valuable context for understanding retatrutide’s thyroid safety profile.
Semaglutide: Over a decade of human use with millions of patient-years of exposure has not revealed increased MTC incidence. Post-marketing surveillance continues to show MTC rates consistent with background population prevalence.
Liraglutide: Similar reassuring long-term data, with comprehensive safety reviews finding no causal relationship between liraglutide use and MTC in humans.
Exenatide: The first GLP-1 agonist approved for clinical use, with nearly 20 years of human safety data showing no thyroid cancer signal.
This collective experience across multiple GLP-1 receptor agonists strongly suggests that the rodent thyroid findings do not translate to human risk, providing reassurance regarding retatrutide thyroid risk while maintaining appropriate vigilance.
Regulatory Perspectives
Regulatory agencies worldwide have established frameworks for GLP-1 agonist thyroid safety assessment:
FDA (United States):
- Black box warnings for GLP-1 agonists reference rodent MTC findings
- Contraindication for personal/family history of MTC or MEN2
- Required post-marketing surveillance
- Emphasis on informed consent regarding theoretical risk
EMA (European Union):
- Similar precautionary labeling
- Comprehensive risk management plans
- Ongoing benefit-risk assessment
MHRA (United Kingdom):
- Consistent safety messaging with FDA/EMA
- Emphasis on appropriate patient selection
- Monitoring recommendations
For researchers working with research-grade peptides in the UK, understanding these regulatory perspectives informs appropriate safety protocols and documentation requirements.
Future Research Directions
Several areas warrant continued investigation to further clarify retatrutide thyroid risk:
Mechanistic Studies:
- Detailed characterization of GLP-1 receptor expression in human thyroid tissue
- Investigation of potential indirect thyroid effects (via pituitary, metabolic changes)
- Comparative receptor binding studies across species
Long-Term Safety Studies:
- Extended follow-up of clinical trial participants (5-10+ years)
- Population-based epidemiological studies
- Cancer registry linkage studies
Biomarker Development:
- More sensitive and specific markers of C-cell activity
- Imaging techniques for early detection of thyroid changes
- Genetic markers identifying susceptible individuals
Combination Therapy Research:
- Thyroid effects when retatrutide is combined with other metabolic agents
- Drug-drug interactions affecting thyroid function
- Optimal monitoring strategies for combination protocols
Practical Implementation: Thyroid Safety in Your Research Protocol
Creating a Thyroid Safety Monitoring Plan
Every research protocol involving retatrutide should include a formal thyroid safety monitoring plan. Here’s a practical framework:
Step 1: Pre-Study Planning
Define your monitoring strategy before subject enrollment:
- Establish baseline assessment requirements
- Determine monitoring frequency based on study duration
- Set clear intervention thresholds
- Identify referral pathways for abnormal findings
- Budget for thyroid-related testing and potential imaging
Step 2: Subject Screening
Implement systematic screening:
- Develop standardized screening questionnaire
- Include family history assessment (3 generations for MEN2)
- Perform baseline laboratory and imaging studies
- Document exclusion criteria application
- Obtain informed consent with specific thyroid risk discussion
Step 3: Ongoing Monitoring
Execute your monitoring plan consistently:
- Use the same laboratory for all thyroid testing (reduces assay variability)
- Standardize blood collection timing (morning, fasting if possible)
- Maintain detailed monitoring logs
- Review results promptly and document review
- Establish clear communication pathways for abnormal results
Step 4: Response Protocols
Prepare for potential thyroid findings:
- Pre-establish relationships with endocrinology consultants
- Create decision algorithms for common scenarios
- Define study discontinuation criteria
- Plan for subject safety if study withdrawal becomes necessary
- Maintain subject follow-up even after discontinuation
Cost-Benefit Considerations
Comprehensive thyroid monitoring adds cost to research protocols. Understanding the value proposition helps justify resource allocation:
Costs:
- Laboratory testing: £50-150 per comprehensive thyroid panel
- Ultrasound imaging: £100-300 per study
- Specialist consultations: £200-500 per consultation
- Additional subject visit time and compensation
Benefits:
- Subject safety: Primary ethical obligation
- Scientific validity: Confident attribution of effects
- Regulatory compliance: Meets safety standards
- Publication quality: Comprehensive safety data enhances publishability
- Institutional reputation: Demonstrates research rigor
For most research protocols, thyroid monitoring represents <5-10% of total study costs while providing substantial value in safety assurance and data quality.
Collaboration with Endocrinology Specialists
Establishing collaborative relationships with thyroid specialists enhances research quality:
When to Involve Endocrinology:
- Protocol design phase: Review monitoring plan
- Subject screening: Evaluation of borderline cases
- Abnormal findings: Expert interpretation and management recommendations
- Study completion: Final safety assessment
What to Provide Consultants:
- Complete research protocol
- Subject’s complete thyroid history and test results
- Timeline of retatrutide exposure
- Specific clinical questions
This collaborative approach ensures that retatrutide thyroid risk is managed with appropriate clinical expertise while maintaining research integrity.
Resources for Retatrutide Researchers
Accessing High-Quality Research Peptides
The foundation of reliable research is high-quality materials. When sourcing retatrutide for laboratory investigations:
Quality Indicators:
- ≥98% purity by HPLC
- Complete certificate of analysis
- Proper storage and shipping conditions
- Transparent supplier information
- Research-use-only labeling
PEPTIDE PRO provides research-grade peptides meeting these quality standards, with fast UK delivery and comprehensive product documentation supporting rigorous scientific investigation.
Educational Resources
Staying current with evolving knowledge about retatrutide thyroid risk requires ongoing education:
Recommended Resources:
- PubMed alerts for “retatrutide thyroid” and related terms
- Endocrine Society clinical practice guidelines
- FDA/EMA safety communications
- Clinical trial registries (ClinicalTrials.gov) for ongoing studies
- Professional conferences (Endocrine Society, ADA, EASD)
Professional Networks
Connecting with other researchers enhances knowledge sharing:
- Research institution peptide working groups
- Online forums for metabolic research (with appropriate confidentiality)
- Collaborative research networks
- Industry-academia partnerships
Balancing Innovation with Safety in Retatrutide Research
The question of retatrutide thyroid risk exemplifies the careful balance researchers must strike between scientific innovation and subject safety. Current evidence suggests that while theoretical concerns exist based on rodent data, human clinical experience with retatrutide and related GLP-1 receptor agonists has been reassuring.
Key Principles for Responsible Retatrutide Research:
✅ Implement comprehensive baseline thyroid assessment before initiating any research protocol
✅ Maintain systematic ongoing monitoring with clearly defined intervals and intervention thresholds
✅ Exclude high-risk populations including those with personal or family history of MTC or MEN2
✅ Source high-purity research peptides from reputable suppliers with complete documentation
✅ Document all findings thoroughly and contribute to the growing safety database
✅ Collaborate with specialists when thyroid abnormalities emerge
✅ Stay current with evolving evidence as long-term safety data accumulates
The scientific community’s understanding of retatrutide thyroid risk will continue to evolve as research progresses. The current evidence base, while reassuring, remains limited compared to established GLP-1 agonists. This reality necessitates continued vigilance, comprehensive monitoring, and transparent reporting of all thyroid-related findings.
For researchers embarking on retatrutide investigations, the thyroid safety considerations outlined in this article provide a framework for responsible, high-quality research. By implementing rigorous monitoring protocols, selecting appropriate subjects, and working with high-quality research materials, the scientific community can advance knowledge about this promising compound while maintaining the highest safety standards.
Next Steps for Your Research
If you’re planning retatrutide research protocols:
- Review your institutional research ethics requirements for peptide studies
- Develop a comprehensive thyroid monitoring plan using the frameworks outlined above
- Establish relationships with endocrinology consultants before beginning subject enrollment
- Source research-grade retatrutide from verified suppliers with complete documentation
- Design your protocol with appropriate exclusion criteria and monitoring intervals
- Budget adequately for comprehensive thyroid safety assessment
- Plan for long-term follow-up extending beyond the active research period
The future of metabolic research is bright, with compounds like retatrutide offering unprecedented potential for understanding and treating metabolic disease. By approaching this research with appropriate attention to thyroid safety, the scientific community can unlock this potential while protecting research subjects and maintaining public trust in peptide research.
Disclaimer: This article is intended for educational purposes for researchers working with peptides in laboratory settings. Retatrutide is for research use only and is not approved for human consumption. All research involving peptides must comply with applicable regulations and institutional ethics requirements. Researchers should consult with appropriate medical and regulatory experts when designing research protocols.
