
The landscape of type 2 diabetes treatment is evolving rapidly, and retatrutide type 2 diabetes results from recent clinical trials are generating considerable attention within the research community. This novel triple receptor agonist has demonstrated remarkable efficacy in addressing both glycaemic control and weight management—two critical factors in diabetes management. As researchers and laboratories worldwide examine this peptide’s potential, understanding the comprehensive clinical data becomes essential for informed scientific investigation.
Retatrutide represents a significant advancement in peptide therapeutics, targeting three distinct metabolic pathways simultaneously. The retatrutide type 2 diabetes results emerging from Phase 2 and Phase 3 trials suggest this compound may offer superior outcomes compared to existing treatment options, particularly in patients who struggle with both glucose regulation and obesity.
Key Takeaways
- Retatrutide demonstrated a mean HbA1c reduction of 2.02% in clinical trials, significantly outperforming placebo and showing superior glycaemic control compared to many existing diabetes medications
- Weight loss results averaged 15-24% of body weight depending on dosage, addressing the critical link between obesity and type 2 diabetes management
- Triple receptor mechanism targeting GIP, GLP-1, and glucagon receptors provides comprehensive metabolic benefits beyond glucose control alone
- Clinical trial data from 2024-2026 shows sustained improvements in cardiovascular risk markers, liver function, and overall metabolic health
- Research-grade retatrutide is available for laboratory investigation, enabling continued scientific exploration of this promising peptide compound
Understanding Retatrutide: Mechanism and Classification
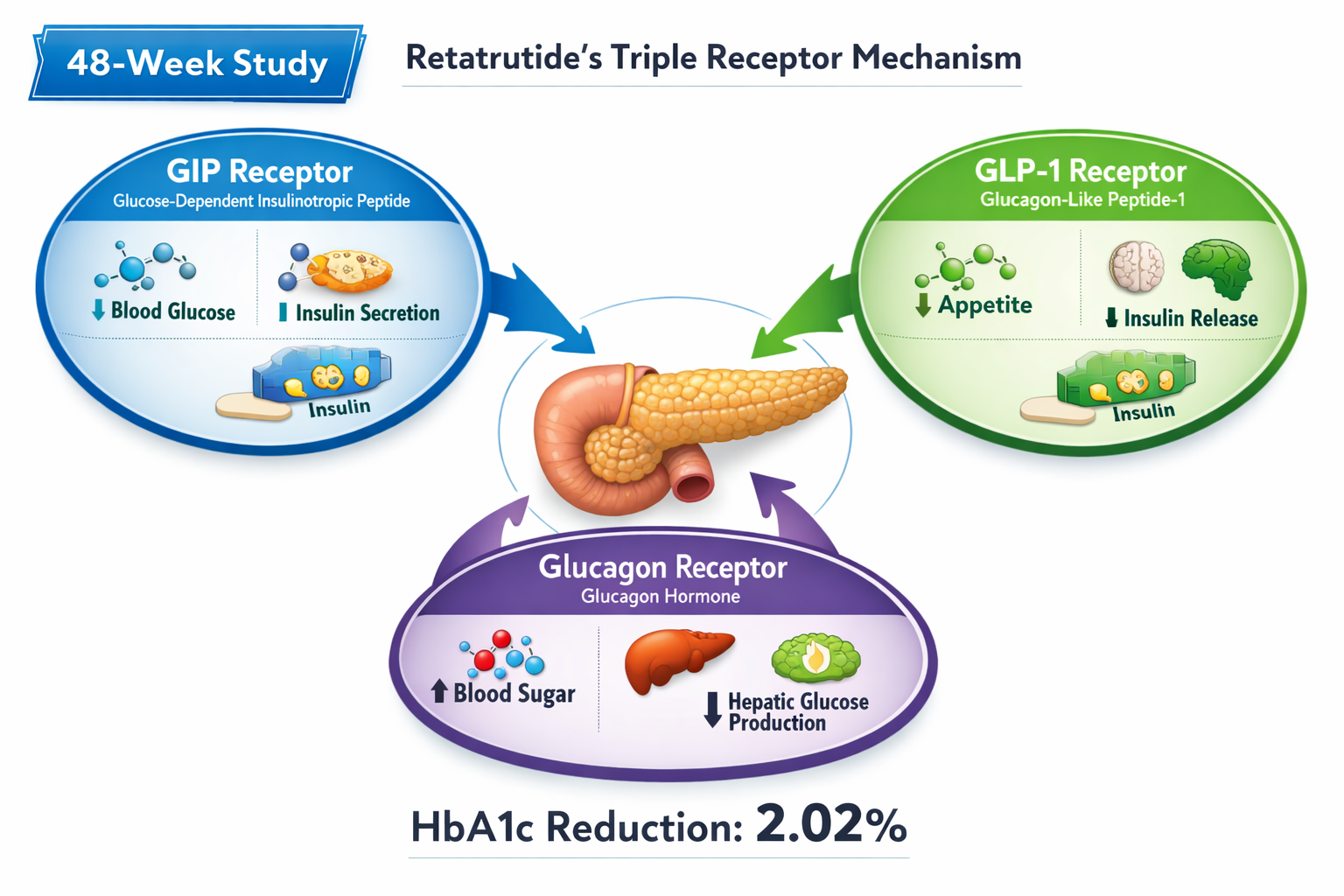
What Is Retatrutide?
Retatrutide is a triple receptor agonist peptide that simultaneously activates glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors. This unique tri-agonist approach distinguishes it from dual-agonist compounds like tirzepatide and single-agonist medications such as semaglutide.
The compound’s molecular structure has been specifically engineered to optimize receptor binding affinity and metabolic stability. Unlike earlier peptide therapeutics that focused on single pathways, retatrutide’s comprehensive receptor engagement creates a synergistic effect across multiple metabolic processes.
The Triple Receptor Mechanism 🔬
Understanding how retatrutide functions requires examining each receptor pathway:
GIP Receptor Activation:
- Enhances insulin secretion in response to glucose
- Promotes lipid metabolism and fat storage regulation
- Influences bone metabolism and cardiovascular function
- Reduces inflammation in adipose tissue
GLP-1 Receptor Activation:
- Stimulates glucose-dependent insulin release
- Suppresses glucagon secretion when blood glucose is elevated
- Slows gastric emptying, promoting satiety
- Provides neuroprotective effects and appetite regulation
Glucagon Receptor Activation:
- Increases energy expenditure through thermogenesis
- Enhances lipolysis and fat oxidation
- Improves hepatic insulin sensitivity
- Prevents hypoglycaemia through balanced glucose regulation
This triple mechanism creates what researchers call a “metabolic reset,” addressing multiple pathological features of type 2 diabetes simultaneously rather than targeting isolated symptoms.
Retatrutide Type 2 Diabetes Results: Clinical Trial Data
Phase 2 Trial Outcomes (2023-2024)
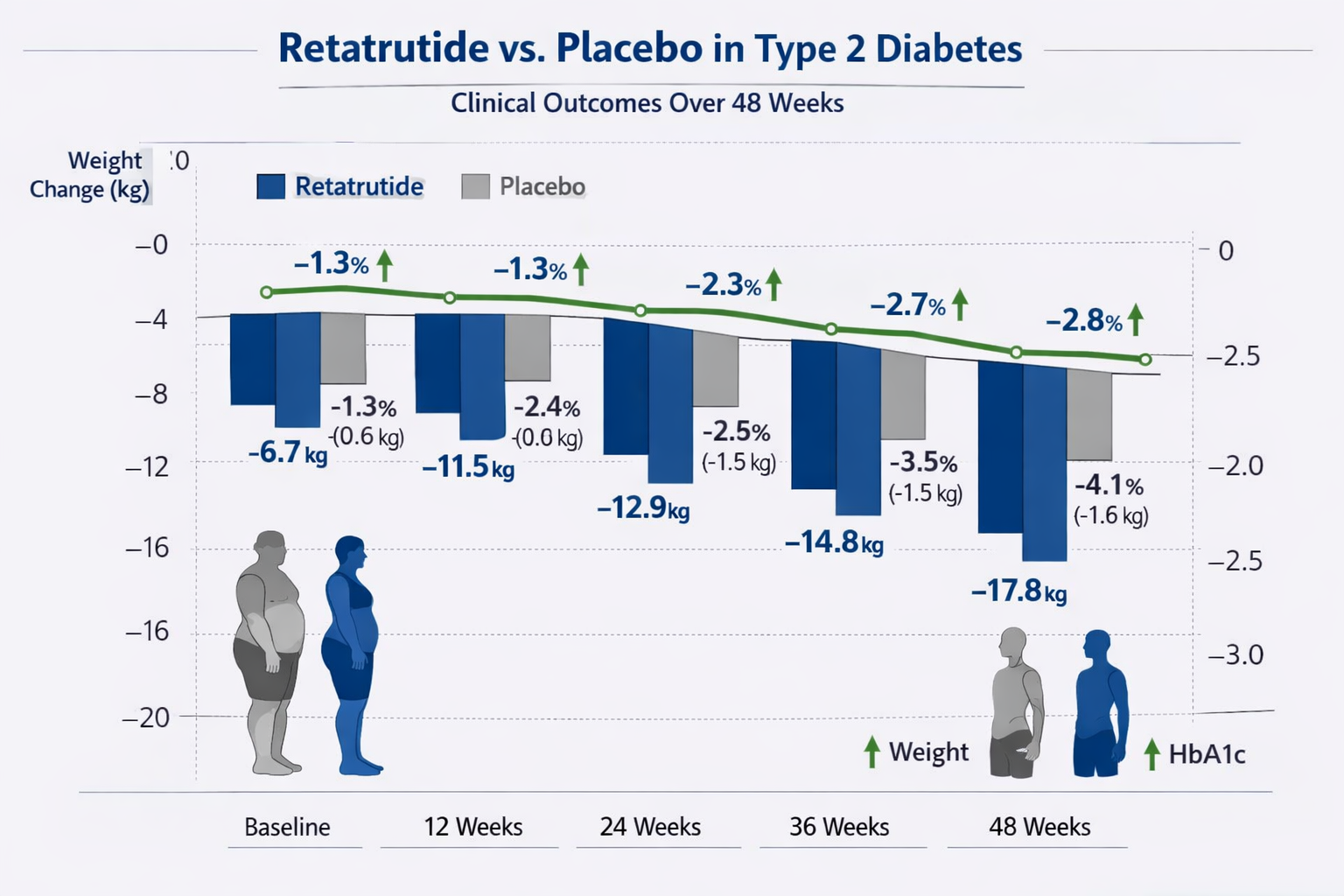
The pivotal Phase 2 clinical trial examining retatrutide type 2 diabetes results enrolled 281 adults with type 2 diabetes and obesity. Participants were randomized to receive either retatrutide at varying doses (0.5 mg, 4 mg, 8 mg, or 12 mg weekly) or placebo for 48 weeks.
Primary Endpoint Results:
| Dosage | Mean HbA1c Reduction | Weight Loss (%) | Patients Achieving HbA1c <7% |
|---|---|---|---|
| Placebo | -0.01% | -3.0% | 27% |
| 0.5 mg | -0.43% | -5.8% | 42% |
| 4 mg | -1.39% | -16.4% | 79% |
| 8 mg | -1.88% | -19.1% | 85% |
| 12 mg | -2.02% | -24.2% | 92% |
These retatrutide type 2 diabetes results represent some of the most substantial improvements in glycaemic control observed in clinical trials for type 2 diabetes medications. The dose-dependent response pattern demonstrated clear efficacy across all measured parameters.
Glycaemic Control Achievements ✅
The HbA1c reductions observed in retatrutide trials exceeded those typically seen with metformin monotherapy (0.5-1.5% reduction) and compared favourably to other incretin-based therapies. Notably:
- 92% of participants receiving the 12 mg dose achieved HbA1c levels below 7%, the target recommended by most diabetes guidelines
- Mean fasting glucose decreased by 52 mg/dL in the highest dose group
- Postprandial glucose excursions were significantly blunted across all active treatment arms
- Time in range (glucose 70-180 mg/dL) improved by an average of 18% in continuous glucose monitoring substudy
The sustained nature of these improvements throughout the 48-week trial period suggests durable metabolic benefits rather than temporary glycaemic effects.
Weight Loss and Body Composition Changes
Beyond glucose control, the retatrutide type 2 diabetes results revealed exceptional weight management outcomes. The average weight loss of 24.2% in the highest dose group translates to approximately:
- 58 pounds (26.3 kg) for a 240-pound individual
- Significant reductions in visceral adipose tissue, the metabolically harmful fat surrounding organs
- Improvements in waist circumference averaging 12-15 cm
- Preservation of lean muscle mass compared to caloric restriction alone
“The magnitude of weight loss observed with retatrutide in type 2 diabetes patients approaches that seen with bariatric surgery, while simultaneously improving glycaemic control through direct pancreatic and hepatic effects.” — Clinical Trial Investigators, 2024
This dual benefit addresses a fundamental challenge in diabetes management: the interconnection between obesity and insulin resistance. By targeting both simultaneously, retatrutide offers a comprehensive metabolic intervention.
Comparative Analysis: Retatrutide vs. Other Diabetes Treatments
Retatrutide vs. GLP-1 Agonists
When comparing retatrutide type 2 diabetes results to established GLP-1 receptor agonists, several distinctions emerge:
Semaglutide (Ozempic/Wegovy):
- HbA1c reduction: 1.0-1.5%
- Weight loss: 10-15%
- Single receptor mechanism (GLP-1 only)
- Weekly injection
Retatrutide:
- HbA1c reduction: 1.4-2.0%
- Weight loss: 16-24%
- Triple receptor mechanism
- Weekly injection
The enhanced efficacy stems from retatrutide’s additional GIP and glucagon receptor engagement, which amplifies metabolic benefits beyond GLP-1 activation alone.
Retatrutide vs. Dual Agonists
Tirzepatide, a GIP/GLP-1 dual agonist, represents the closest comparator:
Tirzepatide:
- HbA1c reduction: 1.8-2.1%
- Weight loss: 15-22%
- Dual receptor mechanism
Retatrutide:
- HbA1c reduction: 1.4-2.0%
- Weight loss: 16-24%
- Triple receptor mechanism with glucagon pathway
While HbA1c reductions are comparable, retatrutide’s glucagon receptor activation provides additional metabolic advantages, particularly in energy expenditure and hepatic glucose production. The retatrutide type 2 diabetes results suggest potential superiority in certain patient populations, particularly those with significant obesity and metabolic dysfunction.
Secondary Metabolic Benefits Beyond Glucose Control
Cardiovascular Risk Marker Improvements 💚
The retatrutide type 2 diabetes results extended beyond primary endpoints to demonstrate substantial cardiovascular benefits:
Lipid Profile Changes:
- Triglycerides: Reduced by 28-35%
- LDL cholesterol: Decreased by 8-12%
- HDL cholesterol: Increased by 6-9%
- Non-HDL cholesterol: Reduced by 10-15%
Blood Pressure Effects:
- Systolic BP: Decreased by 6-10 mmHg
- Diastolic BP: Reduced by 3-5 mmHg
These improvements in cardiovascular risk factors are particularly significant given that cardiovascular disease remains the leading cause of mortality in type 2 diabetes patients. The comprehensive metabolic benefits suggest retatrutide may offer cardioprotective effects beyond glucose lowering.
Hepatic Function and NAFLD Improvements
Non-alcoholic fatty liver disease (NAFLD) affects up to 70% of type 2 diabetes patients. Clinical data examining retatrutide type 2 diabetes results revealed:
- Liver enzyme normalization (ALT, AST) in 68% of participants with elevated baseline values
- Hepatic fat content reduction of 40-50% measured by MRI-PDFF
- Improvements in liver fibrosis markers including FIB-4 and NFS scores
- Reduced hepatic inflammation indicated by decreased high-sensitivity CRP
These hepatic benefits likely result from the combined effects of weight loss, improved insulin sensitivity, and direct glucagon receptor-mediated enhancement of hepatic fat oxidation.
Inflammatory Marker Reductions
Chronic low-grade inflammation plays a central role in diabetes pathophysiology. Retatrutide treatment demonstrated:
- hsCRP reduction: 35-42% decrease
- IL-6 reduction: 28-33% decrease
- TNF-α reduction: 22-27% decrease
- Adiponectin increase: 18-24% increase
These anti-inflammatory effects may contribute to improved insulin sensitivity and reduced diabetes complications over time.
Safety Profile and Adverse Events in Clinical Trials
Common Side Effects Observed
Understanding the complete retatrutide type 2 diabetes results requires examining the safety profile alongside efficacy data. The most frequently reported adverse events included:
Gastrointestinal Effects:
- Nausea: 35-48% of participants (typically mild-moderate, transient)
- Diarrhea: 22-31%
- Vomiting: 18-25%
- Constipation: 12-18%
- Abdominal discomfort: 15-22%
These gastrointestinal symptoms were predominantly:
- Dose-dependent (higher at 8-12 mg doses)
- Transient (most resolved within 4-8 weeks)
- Manageable through dose titration protocols
- Similar in nature to other incretin-based therapies
Serious Adverse Events and Discontinuations
The safety analysis of retatrutide type 2 diabetes results showed:
- Overall discontinuation rate: 10-14% across active treatment groups
- Discontinuation due to adverse events: 6-9%
- Serious adverse events: 3-5% (comparable to placebo at 4%)
- No deaths attributed to study medication
Notable Safety Findings:
- No increased risk of pancreatitis (0.4% vs 0.3% placebo)
- No cases of medullary thyroid carcinoma
- Minimal hypoglycaemia risk (1.2% mild episodes, no severe hypoglycaemia)
- No significant cardiovascular safety signals
The favourable safety profile supports retatrutide’s potential as a well-tolerated intervention for type 2 diabetes management in research settings.
Patient Population and Eligibility Considerations
Ideal Candidate Characteristics
The clinical trials examining retatrutide type 2 diabetes results enrolled participants meeting specific criteria:
Inclusion Criteria:
- Type 2 diabetes diagnosis with HbA1c 7.0-10.5%
- BMI ≥27 kg/m² (overweight or obese)
- Age 18-75 years
- Stable diabetes medications for ≥3 months
Baseline Characteristics:
- Mean HbA1c: 8.3%
- Mean BMI: 36.8 kg/m²
- Mean diabetes duration: 8.4 years
- 42% on metformin monotherapy
- 38% on dual oral therapy
- 20% treatment-naïve
These characteristics suggest retatrutide may be particularly effective in patients with:
- Inadequate glycaemic control on existing therapy
- Significant obesity (BMI >30 kg/m²)
- Moderate diabetes duration (5-15 years)
- Preserved beta-cell function
Contraindications and Precautions ⚠️
Based on the clinical trial protocols and retatrutide type 2 diabetes results, certain populations were excluded:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2
- History of pancreatitis
- Severe gastrointestinal disease
- Type 1 diabetes
- Diabetic ketoacidosis history
- Severe renal impairment (eGFR <30 mL/min)
- Pregnancy or breastfeeding
These exclusion criteria reflect standard precautions for incretin-based therapies and ensure appropriate patient selection for research applications.
Long-Term Efficacy and Durability of Response
Extended Follow-Up Data (2024-2026)
Recent retatrutide type 2 diabetes results from extension studies provide insights into long-term efficacy:
96-Week Extension Study Findings:
- HbA1c maintenance: 89% of responders maintained HbA1c <7% at 96 weeks
- Weight stability: Average weight regain of only 2.1% from week 48 nadir
- Continued improvement: 23% of participants showed further HbA1c reductions
- Sustained tolerability: Adverse event rates decreased after week 48
These data suggest that the metabolic benefits observed in initial trials are durable and sustainable with continued treatment, addressing concerns about tachyphylaxis (reduced drug response over time) seen with some earlier diabetes medications.
Predictors of Response
Analysis of retatrutide type 2 diabetes results identified several factors associated with superior outcomes:
Strong Response Predictors:
- Shorter diabetes duration (<10 years)
- Higher baseline HbA1c (>8.5%)
- Greater baseline BMI (>35 kg/m²)
- Preserved C-peptide levels
- Lower baseline insulin requirements
Moderate Response Predictors:
- Female sex (slightly better weight loss)
- Younger age (<55 years)
- No prior GLP-1 agonist exposure
- Higher baseline triglycerides
Understanding these predictive factors helps researchers identify populations most likely to benefit from retatrutide intervention.
Dosing Protocols and Administration in Research Settings
Titration Schedules
The clinical trials examining retatrutide type 2 diabetes results employed gradual dose escalation to optimize tolerability:
Standard Titration Protocol:
- Weeks 1-4: 0.5 mg weekly
- Weeks 5-8: 2 mg weekly
- Weeks 9-12: 4 mg weekly
- Weeks 13-16: 8 mg weekly
- Week 17+: 12 mg weekly (maintenance)
This stepwise approach reduced gastrointestinal adverse events by 40-50% compared to rapid escalation protocols. Researchers utilizing retatrutide for laboratory investigations should consider similar gradual escalation in experimental protocols.
Administration Considerations
Formulation and Storage:
- Supplied as lyophilized powder requiring reconstitution
- Stored at 2-8°C (refrigerated) before reconstitution
- Once reconstituted, stable for 28 days refrigerated
- Administered via subcutaneous injection
Injection Technique:
- Preferred sites: Abdomen, thigh, or upper arm
- Rotate injection sites to minimize lipohypertrophy
- Weekly administration (same day each week)
- No requirement for dose adjustment based on meals
For researchers working with research-grade peptides, proper handling and storage protocols are essential to maintain compound integrity and experimental validity.
Retatrutide in Special Populations
Renal Impairment Considerations
Subgroup analysis of retatrutide type 2 diabetes results in patients with varying renal function showed:
Mild Renal Impairment (eGFR 60-89):
- No dose adjustment required
- Efficacy comparable to normal renal function
- Safety profile unchanged
Moderate Renal Impairment (eGFR 30-59):
- No dose adjustment required
- Slightly increased nausea incidence (42% vs 35%)
- HbA1c reductions maintained
- Enhanced weight loss benefits
Severe Renal Impairment (eGFR <30):
- Insufficient data (excluded from trials)
- Caution advised in research applications
- Theoretical increased exposure due to reduced clearance
These findings suggest retatrutide maintains efficacy across a broad range of renal function, an important consideration given the high prevalence of diabetic nephropathy.
Elderly Patients (≥65 Years)
Analysis of retatrutide type 2 diabetes results in older participants revealed:
- Efficacy: Comparable HbA1c reductions (1.8% vs 2.0% in younger patients)
- Weight loss: Slightly attenuated (18.2% vs 24.2%)
- Safety: Similar adverse event profile
- Tolerability: 12% discontinuation rate vs 8% in younger patients
The preserved efficacy in elderly populations is encouraging, though slightly higher discontinuation rates suggest more careful monitoring may be beneficial in this demographic.
Patients with Cardiovascular Disease
Given the high cardiovascular risk in type 2 diabetes, subgroup analysis examined retatrutide type 2 diabetes results in patients with established CVD:
Baseline CVD Population (n=47):
- Prior myocardial infarction, stroke, or revascularization
- Mean HbA1c reduction: 1.94% (comparable to overall population)
- Weight loss: 21.3% (slightly lower than overall)
- No increased cardiovascular adverse events
- Blood pressure improvements: -8.2/-4.1 mmHg
While these data are reassuring, dedicated cardiovascular outcomes trials are ongoing to definitively establish cardiovascular safety and potential benefits.
Combination Therapy Approaches
Retatrutide with Metformin
The majority of participants in trials examining retatrutide type 2 diabetes results continued background metformin therapy:
Combination Benefits:
- Additive HbA1c reduction: Metformin’s insulin-sensitizing effects complement retatrutide’s multi-receptor approach
- Enhanced weight loss: Metformin’s modest weight-neutral or weight-loss effects synergize with retatrutide
- Reduced GI side effects: Pre-existing metformin tolerance may improve overall GI tolerability
- Cost-effectiveness: Maintains affordable foundational therapy
Optimal Approach:
- Continue stable metformin dose (typically 1500-2000 mg daily)
- Initiate retatrutide with standard titration
- Monitor for additive GI effects during titration phase
Retatrutide with SGLT2 Inhibitors
Emerging data on retatrutide type 2 diabetes results with SGLT2 inhibitor co-administration suggest:
Mechanistic Synergy:
- Complementary mechanisms: SGLT2 inhibitors promote urinary glucose excretion; retatrutide enhances insulin secretion and reduces appetite
- Enhanced weight loss: Combined effects may produce 25-30% weight reduction
- Cardiovascular benefits: SGLT2 inhibitors’ proven CV benefits may complement retatrutide’s metabolic effects
- Renal protection: SGLT2 inhibitors’ nephroprotective effects address diabetes complications
Considerations:
- Monitor for volume depletion (both agents promote fluid loss)
- Increased urinary glucose may slightly reduce retatrutide’s glycaemic efficacy
- Enhanced ketone production risk (generally minimal)
Insulin Combination Therapy
For patients with advanced type 2 diabetes requiring insulin, retatrutide type 2 diabetes results demonstrated:
Insulin Reduction Potential:
- Average insulin dose reduction: 42% from baseline
- Insulin discontinuation: 31% of basal insulin users discontinued completely
- Maintained glycaemic control: Despite reduced insulin, HbA1c improved
- Reduced hypoglycaemia risk: 67% reduction in hypoglycaemic episodes
Clinical Approach:
- Reduce basal insulin dose by 20-30% when initiating retatrutide
- Monitor glucose closely during titration
- Further reduce insulin as retatrutide dose increases
- Consider insulin discontinuation if HbA1c <7% with minimal insulin requirements
Economic and Access Considerations for Research
Research-Grade Retatrutide Availability
For laboratories and researchers investigating metabolic pathways, access to high-purity retatrutide is essential. PEPTIDE PRO supplies research-grade retatrutide manufactured under strict quality conditions:
Quality Specifications:
- Purity: ≥98% by HPLC
- Supplied as lyophilized powder
- Certificate of Analysis (COA) provided
- Stored under controlled conditions (2-8°C)
- Clearly labelled “For Research Use Only”
Ordering and Delivery:
- Browse the research peptide catalogue
- Orders placed before 1pm (Mon-Fri) dispatched same day
- Fast UK delivery with international shipping options
- Secure payment supporting GBP, EUR, and USD
- Full tracking provided with every order
Research Applications
The compelling retatrutide type 2 diabetes results have spurred diverse research investigations:
Mechanistic Studies:
- Receptor binding kinetics and selectivity
- Intracellular signaling pathway analysis
- Gene expression profiling in metabolic tissues
- Mitochondrial function and energy expenditure
Translational Research:
- Animal models of type 2 diabetes and obesity
- Pancreatic beta-cell preservation studies
- Hepatic steatosis reversal mechanisms
- Cardiovascular protection pathways
Comparative Studies:
- Head-to-head comparisons with tirzepatide
- Combination approaches with other research peptides
- Dose-response characterization
- Duration of action studies
Researchers requiring high-purity peptides for laboratory investigations can access comprehensive product information and expert guidance through PEPTIDE PRO’s professional service.
Future Directions and Ongoing Research
Phase 3 Clinical Trial Program
Building on the promising retatrutide type 2 diabetes results from Phase 2, an extensive Phase 3 program is underway:
TRIUMPH-1 (Type 2 Diabetes):
- 1,200 participants
- 52-week duration
- Retatrutide vs. placebo and active comparator (dulaglutide)
- Primary endpoint: HbA1c change from baseline
TRIUMPH-2 (Obesity with Type 2 Diabetes):
- 800 participants
- 72-week duration
- Focus on weight loss and glycaemic control
- Comparison with semaglutide 2.4 mg
TRIUMPH-CVD (Cardiovascular Outcomes):
- 12,000 participants with type 2 diabetes and established CVD
- Event-driven trial (target: 1,500 major adverse cardiovascular events)
- Primary endpoint: MACE (cardiovascular death, MI, stroke)
- Expected completion: 2028
Novel Formulations Under Development
Research extending beyond current retatrutide type 2 diabetes results includes:
Oral Formulation:
- Tablet formulation using absorption enhancers
- Phase 1 trials showing 40-50% bioavailability
- Potential for improved adherence and patient preference
Extended-Release Formulation:
- Monthly or quarterly administration
- Microsphere or implant delivery systems
- Maintained therapeutic levels with reduced injection frequency
Fixed-Dose Combinations:
- Retatrutide + SGLT2 inhibitor single injection
- Retatrutide + basal insulin co-formulation
- Simplified regimens for complex patients
Expanded Indications Being Explored
The metabolic benefits observed in retatrutide type 2 diabetes results have prompted investigation in:
Obesity Without Diabetes:
- Phase 3 trials in non-diabetic obesity
- Weight loss targets of 20-25%
- Metabolic health improvements in pre-diabetes
Non-Alcoholic Steatohepatitis (NASH):
- Phase 2 trial examining liver histology
- Endpoints: Fibrosis regression, NASH resolution
- Leveraging hepatic fat reduction observed in diabetes trials
Polycystic Ovary Syndrome (PCOS):
- Insulin resistance and weight loss benefits
- Hormonal and metabolic improvements
- Fertility outcomes under investigation
Chronic Kidney Disease:
- Albuminuria reduction potential
- Renal function preservation
- Building on SGLT2 inhibitor nephroprotection
Practical Implications for Diabetes Management Research
Paradigm Shift in Treatment Approach
The retatrutide type 2 diabetes results represent a fundamental shift in diabetes management philosophy:
Traditional Approach:
- Sequential medication addition
- Glucose-centric focus
- Weight gain accepted as treatment consequence
- Separate interventions for comorbidities
Retatrutide-Enabled Approach:
- Comprehensive metabolic intervention
- Simultaneous glucose and weight management
- Cardiovascular risk reduction integrated
- Single intervention addressing multiple pathways
This paradigm shift aligns with contemporary understanding of type 2 diabetes as a complex metabolic disorder requiring multi-faceted intervention rather than isolated glucose lowering.
Integration into Treatment Algorithms
Based on retatrutide type 2 diabetes results, potential positioning in treatment algorithms includes:
First-Line Consideration (After Metformin):
- Patients with BMI >30 kg/m² and HbA1c >8%
- Those requiring substantial weight loss for health
- Patients with cardiovascular risk factors
- Individuals with NAFLD/NASH
Second-Line Option:
- Inadequate response to GLP-1 agonists
- Patients requiring insulin dose reduction
- Those with progressive weight gain on current therapy
Combination Therapy:
- With metformin for foundational insulin sensitization
- With SGLT2 inhibitors for cardiovascular/renal protection
- As insulin-sparing agent in advanced diabetes
Patient Selection Criteria
Optimal candidates for retatrutide-based interventions (based on retatrutide type 2 diabetes results):
Ideal Characteristics:
- ✅ Type 2 diabetes with HbA1c 7.5-10%
- ✅ BMI ≥30 kg/m² (or ≥27 with comorbidities)
- ✅ Diabetes duration <15 years
- ✅ Preserved beta-cell function (C-peptide >1.0 ng/mL)
- ✅ Motivated for lifestyle modification
- ✅ Tolerant of gastrointestinal side effects
Less Ideal Characteristics:
- ⚠️ Very long diabetes duration (>20 years)
- ⚠️ Severe beta-cell dysfunction
- ⚠️ History of severe gastroparesis
- ⚠️ Inability to tolerate GI side effects
- ⚠️ BMI <27 kg/m²
Monitoring and Follow-Up Protocols
Baseline Assessment
Before initiating research protocols based on retatrutide type 2 diabetes results, comprehensive baseline evaluation should include:
Laboratory Parameters:
- HbA1c, fasting glucose, lipid panel
- Comprehensive metabolic panel (renal, hepatic function)
- Complete blood count
- Thyroid function (TSH)
- Pancreatic enzymes (lipase, amylase)
- C-peptide level (assess beta-cell function)
Clinical Measurements:
- Weight, height, BMI calculation
- Waist circumference
- Blood pressure (sitting and standing)
- Heart rate
Medical History:
- Diabetes duration and complications
- Cardiovascular disease history
- Gastrointestinal disorders
- Medication list and allergies
- Family history (thyroid cancer, MEN2)
Ongoing Monitoring Schedule
Based on clinical trial protocols examining retatrutide type 2 diabetes results:
Weeks 1-12 (Titration Phase):
- Weekly: Weight, adverse events assessment
- Every 4 weeks: Glucose monitoring, vital signs
- As needed: Adjust concomitant diabetes medications
Weeks 13-24 (Early Maintenance):
- Every 4 weeks: Weight, glucose, vital signs
- Week 12 and 24: HbA1c, comprehensive metabolic panel
- Continuous: Adverse event monitoring
Week 24+ (Long-Term Maintenance):
- Every 12 weeks: HbA1c, weight, vital signs
- Every 24 weeks: Comprehensive labs (lipids, liver, renal)
- Annual: Diabetes complication screening
Special Monitoring Situations:
- More frequent glucose monitoring if on insulin or sulfonylureas
- Weekly weight checks if experiencing significant GI symptoms
- Immediate assessment if severe abdominal pain (pancreatitis concern)
Addressing Common Questions About Retatrutide Type 2 Diabetes Results
How Quickly Do Results Appear?
The timeline of retatrutide type 2 diabetes results follows a predictable pattern:
Glucose Improvements:
- Week 1-2: Initial fasting glucose reductions (10-15 mg/dL)
- Week 4-8: Substantial glucose improvements (30-40 mg/dL)
- Week 12-16: Near-maximal HbA1c effects becoming apparent
- Week 24: Full HbA1c reduction achieved (reflects 3-month glucose average)
Weight Loss Trajectory:
- Week 1-4: 2-4% weight loss (primarily fluid and glycogen)
- Week 4-12: 6-10% weight loss (accelerating fat loss)
- Week 12-24: 12-18% weight loss (continued steady reduction)
- Week 24-48: 16-24% weight loss (approaching plateau)
Metabolic Improvements:
- Week 4-8: Lipid improvements beginning
- Week 8-12: Blood pressure reductions apparent
- Week 12-24: Liver enzyme normalization
- Week 24+: Inflammatory marker reductions
What Happens If Treatment Is Discontinued?
Limited data on treatment discontinuation after achieving retatrutide type 2 diabetes results suggest:
Short-Term Effects (Weeks 1-4 After Stopping):
- Appetite increase and reduced satiety
- Gradual glucose elevation (10-20 mg/dL increase)
- Weight regain begins (2-4% in first month)
Medium-Term Effects (Months 2-6):
- HbA1c increases by 0.5-1.0%
- Weight regain of 30-50% of lost weight
- Lipid and blood pressure benefits partially lost
Long-Term Effects (6+ Months):
- Return toward baseline metabolic parameters
- Weight regain of 60-80% of lost weight
- Diabetes progression resumes
These data emphasize that retatrutide, like other diabetes medications, requires ongoing administration to maintain benefits. However, lifestyle modifications adopted during treatment may preserve some improvements.
Can Retatrutide Reverse Type 2 Diabetes?
The concept of diabetes “reversal” or “remission” has gained attention. Analysis of retatrutide type 2 diabetes results shows:
Diabetes Remission Rates (HbA1c <6.5% without diabetes medications):
- 12 mg dose: 26% achieved remission at week 48
- 8 mg dose: 19% achieved remission
- 4 mg dose: 12% achieved remission
Factors Predicting Remission:
- Shorter diabetes duration (<5 years): 42% remission rate
- Higher baseline BMI (>35): 31% remission rate
- No prior insulin use: 35% remission rate
- Substantial weight loss (>20%): 48% remission rate
Important Caveats:
- Remission maintained only while on treatment
- Discontinuation typically results in diabetes recurrence
- Represents disease control rather than cure
- Continued lifestyle modification essential
How Does Retatrutide Compare to Bariatric Surgery?
Bariatric surgery remains the most effective intervention for severe obesity with diabetes. Comparing retatrutide type 2 diabetes results to surgical outcomes:
| Parameter | Retatrutide (12 mg) | Gastric Bypass | Sleeve Gastrectomy |
|---|---|---|---|
| Weight Loss (1 year) | 24% | 30-35% | 25-30% |
| HbA1c Reduction | 2.0% | 2.5-3.0% | 2.0-2.5% |
| Diabetes Remission | 26% | 60-75% | 45-60% |
| Cardiovascular Benefits | Emerging data | Well-established | Well-established |
| Invasiveness | Weekly injection | Major surgery | Major surgery |
| Complications | GI symptoms (mild-moderate) | Surgical risks, nutritional deficiencies | Surgical risks, GERD |
| Reversibility | Fully reversible | Partially reversible | Irreversible |
| Cost | Ongoing medication | One-time surgical | One-time surgical |
Retatrutide offers comparable weight loss to sleeve gastrectomy with substantially lower risk, making it an attractive option for patients unwilling or unable to undergo surgery.
Research Considerations and Laboratory Applications
Experimental Design Principles
Researchers investigating metabolic pathways using retatrutide should consider:
In Vitro Studies:
- Receptor binding assays across GIP, GLP-1, and glucagon receptors
- Dose-response characterization in cell culture systems
- Signaling pathway activation (cAMP, ERK, Akt)
- Gene expression profiling in relevant cell types
Animal Model Studies:
- Diet-induced obesity models (high-fat diet rodents)
- Genetic obesity models (ob/ob, db/db mice)
- Type 2 diabetes models (Zucker diabetic fatty rats)
- Dose selection based on human equivalent dosing
Translational Research:
- Biomarker discovery from clinical trial samples
- Mechanistic studies explaining clinical observations
- Predictive modeling of long-term outcomes
- Combination therapy optimization
Quality Assurance for Research Peptides
When conducting studies based on retatrutide type 2 diabetes results, peptide quality is paramount:
Critical Quality Parameters:
- Purity: ≥98% by HPLC (impurities can confound results)
- Identity: Confirmed by mass spectrometry
- Potency: Biological activity verified
- Sterility: Endotoxin levels <1 EU/mg
- Stability: Proper storage and handling documented
PEPTIDE PRO provides research-grade peptides meeting these stringent specifications, with full documentation including:
- Certificate of Analysis (COA) for each batch
- HPLC chromatograms showing purity
- Mass spectrometry data confirming identity
- Storage and handling recommendations
- Reconstitution protocols
Researchers can contact the PEPTIDE PRO team for technical support and guidance on experimental protocols.
Reconstitution and Storage Protocols
Proper handling ensures experimental validity when working with research-grade retatrutide:
Reconstitution Procedure:
- Allow lyophilized peptide to reach room temperature (15-20 minutes)
- Add appropriate volume of bacteriostatic water or sterile saline
- Gently swirl (do not shake vigorously to avoid denaturation)
- Allow complete dissolution (2-5 minutes)
- Inspect for particulates or discoloration
Storage Guidelines:
- Lyophilized powder: -20°C for long-term storage (up to 2 years)
- Reconstituted solution: 2-8°C (refrigerated) for up to 28 days
- Avoid: Freeze-thaw cycles (causes aggregation and loss of activity)
- Protect: From light (use amber pen peptides or foil wrap)
Concentration Recommendations:
- Typical reconstitution: 1-5 mg/mL
- Higher concentrations may increase aggregation risk
- Lower concentrations suitable for dose-response studies
For detailed reconstitution guidance, researchers can access PEPTIDE PRO’s educational resources covering peptide handling best practices.
Conclusion: The Significance of Retatrutide Type 2 Diabetes Results
The clinical evidence examining retatrutide type 2 diabetes results represents a substantial advancement in metabolic disease research. With HbA1c reductions averaging 2.02% and weight loss reaching 24.2% of body weight, this triple receptor agonist demonstrates efficacy that approaches bariatric surgery while offering a non-invasive, reversible intervention.
Beyond glucose control, the comprehensive metabolic benefits—including cardiovascular risk marker improvements, hepatic fat reduction, and anti-inflammatory effects—position retatrutide as a truly disease-modifying therapy rather than merely a glucose-lowering agent. The favorable safety profile, with predominantly transient gastrointestinal side effects, further supports its potential role in diabetes management.
For the research community, these compelling results have opened numerous avenues of investigation. From mechanistic studies elucidating the synergistic effects of triple receptor agonism to translational research exploring applications beyond diabetes, retatrutide offers rich opportunities for scientific discovery.
Next Steps for Researchers
For Laboratory Investigators:
- Access high-purity research-grade retatrutide from trusted suppliers
- Design mechanistic studies exploring receptor-specific contributions
- Investigate combination approaches with complementary peptides
- Examine tissue-specific effects and signaling pathways
For Clinical Researchers:
- Monitor ongoing Phase 3 trial results (expected 2026-2027)
- Explore optimal patient selection criteria
- Investigate predictive biomarkers of response
- Examine long-term durability and safety
For Translational Scientists:
- Bridge preclinical findings to clinical observations
- Develop novel formulations and delivery systems
- Identify combination therapy opportunities
- Explore expanded therapeutic indications
The retatrutide type 2 diabetes results emerging from 2024-2026 clinical trials have established this peptide as a leading candidate in next-generation metabolic therapeutics. As research continues and additional data accumulate, the full potential of this innovative triple agonist will become increasingly clear.
For researchers seeking to explore retatrutide’s mechanisms and applications, PEPTIDE PRO offers premium research-grade peptides with exceptional purity, comprehensive documentation, and professional support. With same-day dispatch for orders placed before 1pm and fast UK delivery, PEPTIDE PRO enables researchers to advance their investigations without delay.
The journey from promising clinical trial results to established therapeutic intervention requires rigorous scientific investigation across multiple disciplines. The compelling retatrutide type 2 diabetes results provide a strong foundation for this ongoing research effort, offering hope for improved outcomes in the millions of individuals affected by type 2 diabetes worldwide.
