The landscape of metabolic research peptides has evolved dramatically, with retatrutide vs cagrisema emerging as one of the most compelling comparisons in contemporary peptide science. As researchers and laboratories seek the most effective compounds for metabolic studies, understanding the fundamental differences between these two advanced peptides has become essential. Both represent cutting-edge approaches to metabolic pathway investigation, yet they operate through distinctly different mechanisms that warrant careful examination.
Key Takeaways
- Retatrutide functions as a triple agonist targeting GLP-1, GIP, and glucagon receptors simultaneously, whilst cagrisema combines semaglutide with cagrilintide for dual-pathway activation
- Research applications differ significantly: retatrutide offers triple-receptor modulation in single-compound studies, whereas cagrisema provides combination therapy investigation opportunities
- Dosing protocols, reconstitution requirements, and storage parameters vary considerably between these two research peptides
- Both compounds are strictly for research use only and must be handled under appropriate laboratory conditions with proper documentation
- Selection between retatrutide and cagrisema should be based on specific research objectives, receptor pathway targets, and experimental design requirements
Understanding Retatrutide: Triple Agonist Mechanism
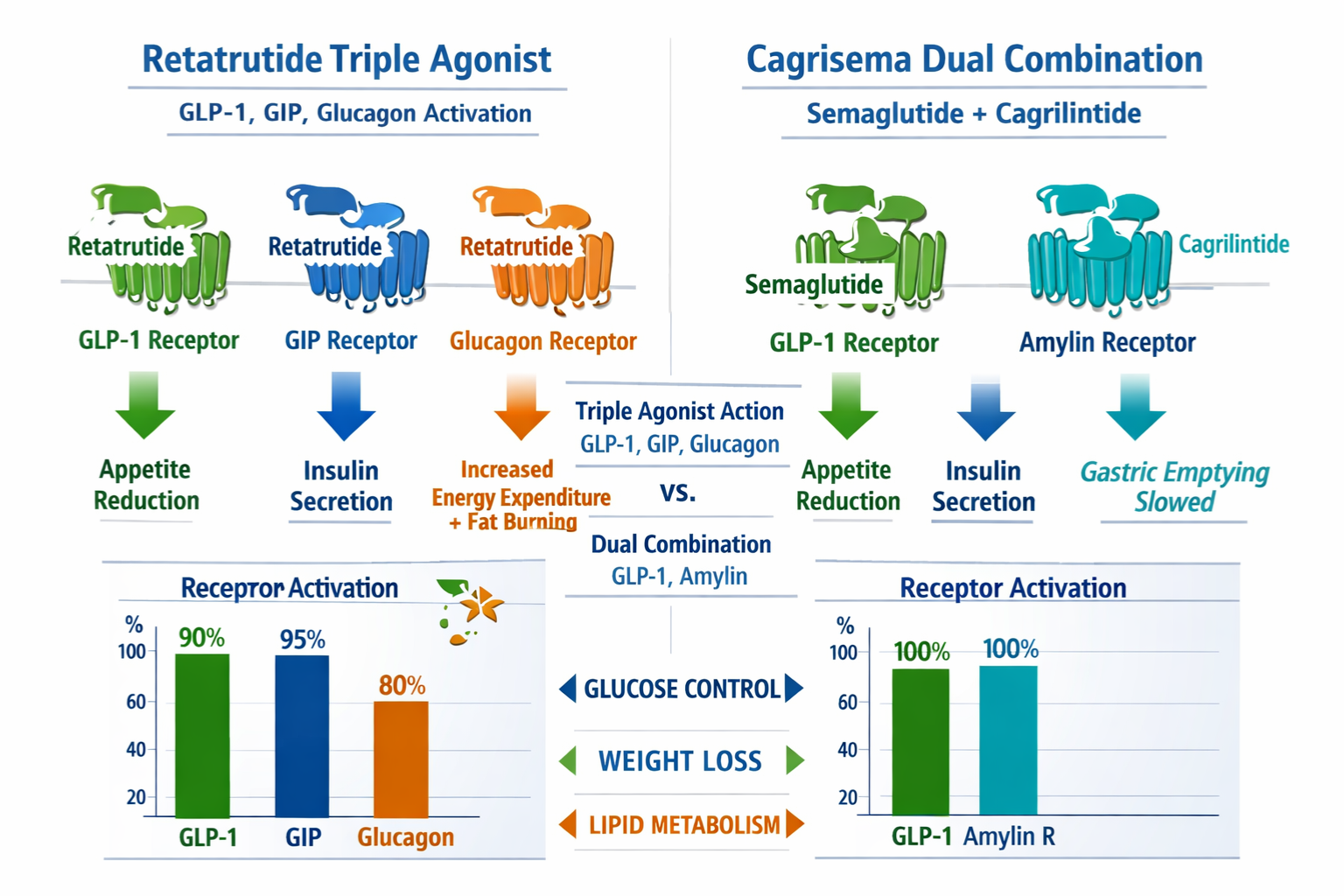
Retatrutide represents a sophisticated advancement in peptide research, functioning as a triple agonist that simultaneously activates three distinct receptor pathways: glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon receptors. This tri-agonist approach provides researchers with a unique tool for investigating complex metabolic pathway interactions.
Molecular Structure and Receptor Binding
The molecular architecture of retatrutide enables it to bind effectively to all three target receptors with varying degrees of affinity. This multi-receptor engagement creates a comprehensive metabolic modulation profile that researchers can study across multiple physiological pathways simultaneously.
Key structural characteristics include:
- Peptide backbone optimised for triple-receptor recognition
- Molecular modifications enhancing stability and half-life
- Specific amino acid sequences facilitating receptor selectivity
- Formulation designed for research-grade purity standards
Research institutions studying metabolic pathways often select retatrutide for its comprehensive receptor coverage, allowing investigation of synergistic effects across multiple systems.
Research Applications and Study Protocols
Laboratories utilising retatrutide in 2026 typically focus on:
- Metabolic pathway mapping – Understanding how simultaneous receptor activation influences cellular signalling cascades
- Receptor interaction studies – Investigating cross-talk between GLP-1, GIP, and glucagon pathways
- Dose-response characterisation – Establishing concentration-dependent effects across different receptor systems
- Comparative pharmacology – Benchmarking against single or dual agonist compounds
The versatility of retatrutide makes it particularly valuable for researchers examining complex metabolic regulation mechanisms that cannot be adequately studied with single-receptor compounds.
Cagrisema Explained: Combination Peptide Approach
Unlike retatrutide’s single-compound triple agonism, cagrisema represents a deliberate combination of two distinct peptides: semaglutide (a GLP-1 receptor agonist) and cagrilintide (an amylin analogue). This combination strategy offers researchers a different investigational paradigm focused on complementary pathway activation.
Component Analysis: Semaglutide and Cagrilintide
Semaglutide operates primarily through GLP-1 receptor activation, providing well-characterised effects on incretin signalling pathways. Researchers have extensively documented its receptor binding profile, making it a reliable component for controlled studies.
Cagrilintide functions as a long-acting amylin receptor agonist, targeting a distinct pathway that complements GLP-1 activity. The amylin pathway plays crucial roles in:
- Gastric emptying regulation
- Satiety signal modulation
- Glucagon secretion inhibition
- Central nervous system metabolic signalling
When combined as cagrisema, these two peptides create a dual-pathway research tool that allows investigation of synergistic mechanisms between incretin and amylin systems.
Combination Ratios and Formulation Considerations
The specific ratio of semaglutide to cagrilintide in cagrisema formulations has been optimised for research applications, with typical preparations maintaining precise proportions to ensure consistent experimental outcomes. Researchers must account for:
- Individual peptide stability requirements
- Differential reconstitution protocols
- Separate degradation pathways
- Potential interaction effects during storage
This combination approach provides laboratories with the ability to study both individual component effects and synergistic interactions within the same experimental framework.
Retatrutide vs Cagrisema: Mechanism Comparison
The fundamental distinction in the retatrutide vs cagrisema comparison centres on their mechanistic approaches to metabolic pathway modulation. Understanding these differences is critical for appropriate research design and compound selection.
Receptor Pathway Coverage
| Receptor Target | Retatrutide | Cagrisema |
|---|---|---|
| GLP-1 |
✅ Direct agonist |
✅ Via semaglutide | | GIP |
✅ Direct agonist |
❌ Not targeted | | Glucagon |
✅ Direct agonist |
❌ Not targeted | | Amylin |
❌ Not targeted |
✅ Via cagrilintide |
This table illustrates the complementary yet distinct receptor coverage, highlighting why researchers might select one compound over the other based on specific pathway investigation requirements.
Pharmacokinetic Profiles
Retatrutide exhibits:
- Single-compound pharmacokinetics
- Unified absorption and distribution profile
- Consistent half-life across all receptor activities
- Simplified metabolic pathway analysis
Cagrisema demonstrates:
- Dual-component pharmacokinetics requiring separate analysis
- Potentially different absorption rates for each component
- Variable half-lives between semaglutide and cagrilintide
- Complex metabolic considerations for combination effects
Researchers conducting pharmacokinetic studies must account for these fundamental differences when designing experimental protocols and interpreting results.
Synergistic vs Additive Effects
A critical research question in the retatrutide vs cagrisema comparison involves whether observed effects represent true synergy or simple additive activity:
Retatrutide’s triple agonism may produce synergistic effects through:
- Simultaneous receptor activation creating pathway cross-talk
- Coordinated metabolic responses across multiple systems
- Potential receptor heterodimer formation
- Integrated signalling cascade amplification
Cagrisema’s combination approach enables researchers to study:
- Independent pathway contributions from each component
- Temporal dynamics of dual-pathway activation
- Dose-titration effects for individual components
- Mechanistic separation of incretin vs amylin effects
Research Applications: When to Choose Each Peptide
Selecting between retatrutide and cagrisema for specific research applications requires careful consideration of experimental objectives, pathway targets, and methodological requirements.
Optimal Research Scenarios for Retatrutide
Laboratories should consider retatrutide when research priorities include:
🔬 Comprehensive metabolic pathway studies requiring simultaneous multi-receptor modulation without the confounding variables of combination therapy.
🔬 Receptor cross-talk investigations examining how GLP-1, GIP, and glucagon pathways interact at the cellular and molecular levels.
🔬 Simplified dosing protocols where single-compound administration reduces experimental complexity and improves reproducibility.
🔬 Novel mechanism exploration investigating whether triple agonism produces unique effects not achievable through dual or single receptor targeting.
Research institutions focusing on integrated metabolic regulation often find retatrutide’s comprehensive receptor coverage advantageous for hypothesis-driven investigation.
Optimal Research Scenarios for Cagrisema
Cagrisema becomes the preferred choice for research programmes emphasising:
🔬 Incretin-amylin pathway interactions specifically examining how GLP-1 and amylin systems coordinate metabolic regulation.
🔬 Component contribution analysis where researchers need to isolate individual effects of semaglutide versus cagrilintide.
🔬 Combination therapy modelling investigating synergistic potential of dual-pathway activation with separately optimised components.
🔬 Established pathway research leveraging the well-characterised profiles of both semaglutide and cagrilintide for predictable experimental outcomes.
Laboratories with specific interest in amylin pathway biology find cagrisema’s inclusion of cagrilintide particularly valuable, as this receptor system is not addressed by retatrutide.
Experimental Design Considerations
When planning research protocols involving either compound, investigators should evaluate:
- Endpoint measurements – Which physiological parameters align with each compound’s receptor profile?
- Temporal dynamics – Do research questions require rapid onset or sustained effects?
- Dose-response relationships – Will single-compound or dual-component titration better serve experimental objectives?
- Control group design – How will appropriate comparators be structured for each peptide type?
- Statistical power – What sample sizes are required given each compound’s effect variability?
Dosing, Reconstitution, and Handling Protocols
Proper handling of research peptides is essential for maintaining compound integrity and ensuring experimental validity. The retatrutide vs cagrisema comparison extends to practical laboratory procedures that differ significantly between these compounds.
Retatrutide Handling Guidelines
Storage requirements:
- Lyophilised powder: Store at -20°C to -80°C in original sealed pen peptides
- Protect from light and moisture exposure
- Avoid repeated freeze-thaw cycles
- Maintain detailed storage logs for quality assurance
Reconstitution protocol:
- Allow pen peptide to reach room temperature (approximately 15-20 minutes)
- Use appropriate bacteriostatic water or sterile water for injection
- Add solvent slowly down the pen peptide wall to minimise foaming
- Gently swirl (never shake vigorously) until completely dissolved
- Inspect solution for clarity and absence of particulates
- Label with reconstitution date and concentration
Post-reconstitution storage:
- Refrigerate at 2-8°C immediately after reconstitution
- Use within timeframe specified by stability data (typically 14-28 days)
- Protect from light during storage
- Never freeze reconstituted solutions
Cagrisema Handling Guidelines
Due to its dual-component nature, cagrisema requires particular attention to ensure both semaglutide and cagrilintide remain stable and active:
Storage requirements:
- Similar temperature requirements as retatrutide (-20°C to -80°C for lyophilised form)
- May require specific packaging to protect both components
- Consider potential differential stability of components
- Maintain separate documentation for combination products
Reconstitution considerations:
- Follow manufacturer-specific protocols for combination peptides
- Ensure solvent compatibility with both components
- Verify complete dissolution of all peptide components
- Account for potential differential solubility
- Document reconstitution parameters meticulously
Concentration calculations: When working with cagrisema, researchers must account for the presence of two active components. If a pen peptide contains 10mg total (for example, 2.4mg semaglutide + 7.6mg cagrilintide), concentration calculations must reflect this combination ratio for accurate dosing in experimental protocols.
Quality Control and Documentation
Both compounds require rigorous quality control measures:
✅ Certificate of Analysis (COA) verification – Confirm purity specifications before use
✅ Visual inspection – Check for discolouration, precipitation, or particulates
✅ pH verification – Ensure reconstituted solution falls within acceptable range
✅ Concentration validation – Verify actual concentration matches intended preparation
✅ Sterility maintenance – Use aseptic technique throughout all handling procedures
PEPTIDE PRO provides comprehensive documentation and certificates of analysis with all research peptides, supporting proper laboratory protocols and regulatory compliance.
Safety Profiles and Research Considerations
When evaluating retatrutide vs cagrisema for research applications, understanding their respective safety profiles and handling precautions is paramount for responsible laboratory practice.
Laboratory Safety Protocols
Personal protective equipment (PPE) requirements:
- Nitrile gloves (latex-free to prevent allergic reactions)
- Laboratory coat or protective clothing
- Safety glasses or face shield when handling powders
- Work within appropriate ventilation (fume hood when indicated)
Spill management procedures:
- Contain spill immediately with absorbent materials
- Clean affected area with appropriate disinfectant
- Dispose of contaminated materials according to institutional protocols
- Document incident per laboratory safety requirements
- Decontaminate equipment and surfaces thoroughly
Research-Only Status and Compliance
⚠️ Critical reminder: Both retatrutide and cagrisema are strictly for research use only. These compounds are not approved for human consumption, veterinary use, or any application outside controlled laboratory environments.
Institutional compliance requirements:
- Appropriate research authorisation and oversight
- Documented research protocols and ethical approvals
- Secure storage with restricted access
- Detailed usage logs and inventory tracking
- Proper waste disposal following regulatory guidelines
Researchers must ensure their institutions maintain full compliance with applicable regulations governing peptide research. Contact PEPTIDE PRO for guidance on documentation requirements and certificates of analysis.
Monitoring and Reporting
Research programmes utilising either compound should implement:
Experimental monitoring:
- Regular assessment of compound stability
- Documentation of any unexpected observations
- Systematic recording of dosing and outcomes
- Quality control checks at predetermined intervals
Incident reporting:
- Protocols for reporting adverse events or unexpected results
- Communication channels with institutional safety officers
- Documentation systems for deviations from standard procedures
- Review processes for continuous improvement
Clinical Trial Data and Research Outcomes (2026 Update)

As of 2026, both retatrutide and cagrisema have generated substantial research interest, with ongoing investigations providing valuable comparative data for the scientific community.
Retatrutide Research Findings
Recent research programmes investigating retatrutide have focused on:
Metabolic pathway characterisation:
- Triple-receptor activation produces coordinated metabolic responses
- Dose-dependent effects observed across GLP-1, GIP, and glucagon pathways
- Receptor selectivity profiles confirm multi-targeted engagement
- Pharmacodynamic studies reveal sustained receptor activation
Efficacy parameters in research models:
- Significant metabolic modulation observed in appropriate research systems
- Dose-response curves established for various experimental endpoints
- Comparative studies demonstrate advantages over single-agonist approaches
- Long-term stability studies support extended research protocols
Cagrisema Research Findings
Investigations into cagrisema have emphasised:
Dual-pathway synergy analysis:
- Complementary effects of GLP-1 and amylin pathway activation
- Component contribution studies isolate individual peptide effects
- Combination ratios influence overall research outcomes
- Temporal dynamics differ between semaglutide and cagrilintide components
Comparative effectiveness research:
- Direct comparisons with single-agent semaglutide demonstrate additive benefits
- Amylin pathway contribution provides distinct mechanistic advantages
- Combination approach enables flexible dose optimisation
- Research models show sustained effects with appropriate dosing protocols
Head-to-Head Comparative Studies
Limited direct retatrutide vs cagrisema comparisons exist in published literature as of 2026, though research institutions have begun conducting parallel investigations:
Preliminary comparative observations suggest:
📊 Receptor coverage – Retatrutide’s glucagon agonism vs cagrisema’s amylin agonism represent fundamentally different pathway targets, making direct efficacy comparisons context-dependent
📊 Dosing complexity – Single-compound retatrutide offers simplified protocols, whilst cagrisema’s dual components provide greater flexibility for component-specific optimisation
📊 Mechanistic insights – Each compound enables investigation of distinct biological questions, with selection depending on research priorities
📊 Reproducibility – Both demonstrate consistent performance when handled according to proper protocols, with quality-controlled preparations from reputable suppliers like PEPTIDE PRO
Cost, Availability, and Sourcing Considerations
Practical considerations including cost, availability, and reliable sourcing significantly influence research planning when selecting between retatrutide and cagrisema.
Pricing Structures and Research Budgeting
Retatrutide pricing factors:
- Single-compound synthesis may offer cost advantages
- Pricing typically based on total peptide mass
- Larger pen peptide sizes often provide better per-milligram value
- Research-grade purity commands premium pricing
Cagrisema pricing factors:
- Combination formulation involves two distinct peptides
- Pricing reflects both semaglutide and cagrilintide components
- Formulation complexity may influence overall cost
- Component ratios affect final pricing structure
Research institutions should request detailed quotations that specify:
- Cost per milligram of active peptide
- Minimum order quantities
- Volume discounts for larger research programmes
- Shipping and handling fees (particularly for temperature-controlled transport)
Supplier Selection and Quality Assurance
Choosing a reputable peptide supplier is critical for research validity and reproducibility. Essential supplier criteria include:
✓ Purity verification – Comprehensive certificates of analysis (COA) with HPLC, mass spectrometry, and purity percentage
✓ Regulatory compliance – Proper labelling as “Research Use Only” with appropriate documentation
✓ Storage and shipping – Temperature-controlled logistics maintaining peptide integrity
✓ Customer support – Technical expertise to answer research-specific questions
✓ Consistency – Batch-to-batch reproducibility for long-term research programmes
PEPTIDE PRO maintains rigorous quality standards across all research peptides, including both retatrutide and cagrisema, with comprehensive documentation and rapid UK delivery supporting research timelines.
Availability and Lead Times
Current availability status (2026):
Both compounds are available through specialised research peptide suppliers, though availability may fluctuate based on:
- Manufacturing schedules and synthesis capacity
- Raw material sourcing for peptide production
- Regulatory changes affecting research compound distribution
- Geographic location and import/export considerations
Planning for research continuity:
- Order peptides well in advance of experimental timelines
- Maintain appropriate inventory for ongoing studies
- Establish relationships with reliable suppliers for priority access
- Consider backup sourcing options for critical research programmes
Researchers in the UK benefit from PEPTIDE PRO’s same-day dispatch for orders placed before 1pm Monday-Friday, ensuring minimal delays in research progression.
Frequently Asked Questions: Retatrutide vs Cagrisema
What is the primary difference between retatrutide and cagrisema?
The fundamental distinction lies in their mechanistic approaches: retatrutide is a single peptide functioning as a triple agonist (GLP-1, GIP, and glucagon receptors), whilst cagrisema is a combination of two separate peptides (semaglutide and cagrilintide) targeting GLP-1 and amylin receptors respectively. This difference influences receptor coverage, experimental design, and research applications.
Which peptide is more suitable for metabolic pathway research?
The answer depends on specific research objectives:
- Choose retatrutide for comprehensive multi-receptor studies examining GLP-1, GIP, and glucagon pathway interactions
- Choose cagrisema for investigations focusing on GLP-1 and amylin pathway synergy, or when amylin receptor biology is central to research questions
Both offer valuable research capabilities within their respective mechanistic frameworks.
Can these peptides be used in combination research protocols?
Whilst theoretically possible, combining retatrutide and cagrisema in the same experimental system would create significant complexity due to overlapping GLP-1 receptor activation and the presence of five distinct receptor pathways. Such approaches would require extremely careful experimental design and sophisticated analytical methods to interpret results meaningfully. Most research programmes select one compound based on pathway priorities.
How do storage requirements differ between these compounds?
Both require similar fundamental storage conditions:
- Lyophilised form: -20°C to -80°C
- Reconstituted solutions: 2-8°C refrigeration
- Protection from light and moisture
- Avoidance of freeze-thaw cycles
However, cagrisema’s dual-component nature may require additional considerations to ensure both peptides remain stable. Always follow supplier-specific storage recommendations and maintain detailed storage logs.
What documentation is required for research use?
Research institutions must maintain:
- Certificates of Analysis (COA) from the peptide supplier
- Institutional research protocols and approvals
- Detailed usage logs and inventory records
- Safety data sheets (SDS) for laboratory personnel
- Storage and handling documentation
- Waste disposal records
PEPTIDE PRO provides comprehensive documentation with all research peptides to support institutional compliance requirements.
Are there any contraindications for laboratory use?
As research-only compounds, retatrutide and cagrisema must never be used for:
- Human consumption or administration
- Veterinary applications
- Any purpose outside controlled laboratory research
- Self-experimentation or non-approved studies
Researchers must ensure proper institutional oversight, ethical approvals, and adherence to all applicable regulations governing peptide research.
How quickly can I obtain these peptides for my research?
Availability varies by supplier and location. PEPTIDE PRO offers same-day dispatch for orders placed before 1pm Monday-Friday, with fast UK delivery options ensuring researchers receive high-purity peptides promptly. International orders may require additional time for customs clearance and temperature-controlled shipping logistics.
What quality control measures should laboratories implement?
Essential quality control practices include:
- Verification of COA specifications upon receipt
- Visual inspection before and after reconstitution
- Proper storage temperature monitoring
- Aseptic technique during all handling procedures
- Regular equipment calibration and validation
- Detailed documentation of all experimental parameters
- Periodic review of handling protocols and procedures
Future Directions and Emerging Research (2026 Perspective)
The retatrutide vs cagrisema comparison continues to evolve as research programmes generate new data and insights into these advanced metabolic peptides.
Emerging Research Trends
Novel application areas under investigation:
🔬 Receptor heterodimer studies – Examining whether triple agonism produces unique receptor complex formations not achievable with dual or single agonists
🔬 Tissue-specific effects – Investigating differential responses across various metabolic tissues and cell types
🔬 Long-term stability research – Characterising extended storage conditions and formulation optimisation
🔬 Biomarker identification – Developing specific markers for pathway activation and metabolic modulation
🔬 Comparative pharmacology – Expanding head-to-head research comparing retatrutide, cagrisema, and other metabolic peptides
Technological Advances Supporting Research
Recent technological developments enhancing peptide research include:
Advanced analytical methods:
- High-resolution mass spectrometry for precise characterisation
- Real-time receptor binding assays
- Multi-parameter flow cytometry for cellular responses
- Sophisticated pharmacokinetic modelling software
Improved formulation technologies:
- Enhanced stability formulations extending shelf life
- Novel delivery systems for experimental applications
- Lyophilisation optimisation reducing degradation
- Quality control automation improving consistency
Collaborative Research Opportunities
The complexity of multi-receptor peptide research creates opportunities for collaborative investigation:
- Multi-institutional studies pooling resources and expertise
- Data sharing initiatives improving reproducibility
- Standardised protocols enhancing cross-study comparisons
- International research networks advancing mechanistic understanding
Researchers interested in collaborative opportunities or seeking high-quality peptides for their investigations can explore PEPTIDE PRO’s extensive catalogue of research-grade compounds.
Conclusion: Making the Right Choice for Your Research
The retatrutide vs cagrisema decision ultimately depends on specific research objectives, pathway targets, and experimental design requirements. Neither compound is universally “superior”—rather, each offers distinct advantages for particular research applications.
Decision Framework for Researchers
Select retatrutide when your research priorities include:
- Comprehensive triple-receptor pathway investigation (GLP-1, GIP, glucagon)
- Single-compound simplicity in experimental design
- Novel mechanism exploration through simultaneous multi-receptor activation
- Studies requiring coordinated metabolic pathway modulation
Select cagrisema when your research priorities include:
- Specific investigation of GLP-1 and amylin pathway interactions
- Component contribution analysis separating individual peptide effects
- Research leveraging well-characterised semaglutide and cagrilintide profiles
- Studies where amylin receptor biology is central to research questions
Ensuring Research Success
Regardless of which peptide you select, research success depends on:
✓ Quality sourcing – Obtaining research-grade peptides from reputable suppliers with comprehensive documentation
✓ Proper handling – Following rigorous storage, reconstitution, and handling protocols
✓ Experimental rigor – Implementing appropriate controls, statistical methods, and reproducibility measures
✓ Regulatory compliance – Maintaining all required documentation and institutional approvals
✓ Continuous learning – Staying current with emerging research and methodological advances
Next Steps for Researchers
Ready to advance your metabolic peptide research? Consider these actionable steps:
- Define research objectives clearly – Identify which receptor pathways are central to your scientific questions
- Review available literature – Examine published research on both compounds relevant to your field
- Consult with colleagues – Discuss experiences and recommendations from other researchers
- Source high-quality peptides – Partner with reputable suppliers offering comprehensive quality assurance
- Develop detailed protocols – Create standardised operating procedures for handling and experimental use
- Implement quality controls – Establish verification and validation processes throughout your research
PEPTIDE PRO provides premium research-grade peptides including both retatrutide and cagrisema, supported by comprehensive certificates of analysis, expert customer service, and fast UK delivery. Our commitment to exceptional purity, transparent documentation, and professional service ensures your research progresses with confidence.
For questions about peptide selection, handling protocols, or research applications, our knowledgeable team is available to provide guidance tailored to your specific requirements. Contact PEPTIDE PRO today to discuss your research needs and explore our extensive catalogue of high-purity research peptides.
Remember: All peptides discussed in this article are strictly for research use only. Not for human or animal consumption. Always maintain proper laboratory protocols, institutional oversight, and regulatory compliance in all research activities.
