
The landscape of metabolic research peptides has evolved dramatically, with newer compounds challenging established protocols. When examining Retatrutide vs Semaglutide (Ozempic/Wegovy), researchers encounter two distinct approaches to GLP-1 receptor pathway investigation—one representing proven single-agonist methodology, the other pioneering triple-agonist mechanisms that may redefine metabolic research parameters. As laboratories worldwide seek optimal compounds for weight management and metabolic studies, understanding the fundamental differences between these peptides becomes essential for informed research design.
Key Takeaways
- Retatrutide operates as a triple agonist (GLP-1, GIP, glucagon receptors), while Semaglutide functions as a selective GLP-1 receptor agonist
- Research data suggests retatrutide may demonstrate superior weight reduction outcomes (up to 24% in clinical trials) compared to semaglutide (approximately 15%)
- Semaglutide benefits from extensive clinical validation and established research protocols, whereas retatrutide represents emerging investigational territory
- Both peptides require careful handling, proper reconstitution, and controlled storage conditions for research applications
- Selection between compounds depends on specific research objectives, protocol requirements, and investigational focus areas
Understanding the Fundamental Mechanisms: Retatrutide vs Semaglutide (Ozempic/Wegovy)
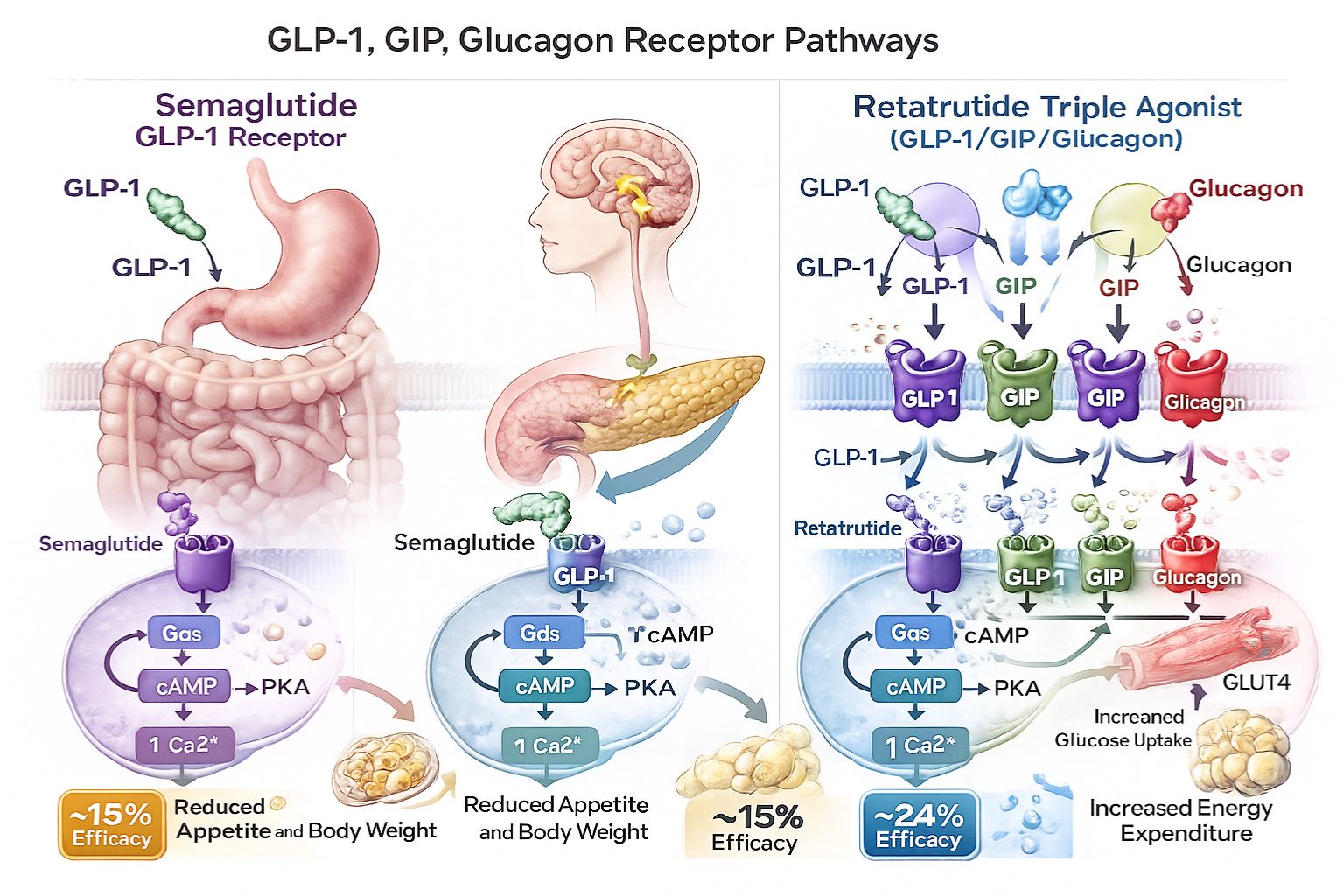
What Is Semaglutide?
Semaglutide represents a glucagon-like peptide-1 (GLP-1) receptor agonist that has become a cornerstone in metabolic research. Originally developed for glycemic control investigations, this peptide mimics the action of naturally occurring GLP-1 hormones, influencing insulin secretion, gastric emptying, and appetite regulation pathways.[1]
The compound exists under multiple brand designations—Ozempic for diabetes-focused research and Wegovy for weight management studies—though the active peptide sequence remains identical. Researchers value semaglutide for its well-documented pharmacokinetic profile, extended half-life enabling weekly administration protocols, and comprehensive safety data accumulated through extensive clinical investigation.[2]
Key characteristics of semaglutide include:
- 🔬 Selective GLP-1 receptor agonism
- ⏱️ Extended half-life (approximately 1 week)
- 📊 Extensive clinical trial data spanning multiple years
- 💉 Subcutaneous administration in research models
�
� Primary focus on glycemic control and appetite modulation
What Is Retatrutide?
Retatrutide represents a novel investigational peptide employing a triple agonist mechanism—simultaneously activating GLP-1, glucose-dependent insulinotropic polypeptide (GIP), and glucagon receptors. This multi-targeted approach distinguishes it fundamentally from single-agonist compounds, potentially offering synergistic metabolic effects across multiple physiological pathways.[3]
As an emerging research compound, retatrutide demonstrates promising preliminary data suggesting enhanced efficacy in weight reduction studies compared to established GLP-1 agonists. The triple receptor activation creates a more comprehensive metabolic intervention, theoretically addressing energy expenditure, fat oxidation, and appetite regulation through complementary mechanisms.[4]
Defining features of retatrutide:
- ⚡ Triple receptor agonism (GLP-1/GIP/glucagon)
- 🔄 Multi-pathway metabolic modulation
- 📈 Emerging clinical trial data showing superior weight loss outcomes
- 🧪 Investigational status requiring further long-term research
�
� Enhanced focus on comprehensive metabolic regulation
For researchers seeking high-purity research peptides, understanding these mechanistic differences proves essential for appropriate compound selection.
Mechanism of Action Comparison
How Semaglutide Works
Semaglutide’s mechanism centers on selective GLP-1 receptor activation. When administered, the peptide binds to GLP-1 receptors distributed throughout the body, particularly in pancreatic beta cells, the gastrointestinal tract, and specific brain regions involved in appetite regulation.[5]
This binding triggers several physiological responses:
- Glucose-dependent insulin secretion: Enhances insulin release only when blood glucose levels are elevated, reducing hypoglycemia risk
- Glucagon suppression: Decreases inappropriate glucagon secretion, preventing excessive hepatic glucose production
- Gastric emptying delay: Slows food transit from stomach to intestines, promoting satiety
- Central appetite regulation: Acts on hypothalamic circuits to reduce hunger signals and food intake
The peptide’s structural modifications compared to native GLP-1 provide resistance to dipeptidyl peptidase-4 (DPP-4) degradation and albumin binding properties, extending its pharmacological activity substantially beyond natural GLP-1’s brief half-life of minutes.[6]
How Retatrutide Works
Retatrutide’s triple agonist approach creates a more complex mechanistic profile. By simultaneously engaging three distinct receptor systems, the compound theoretically produces additive or synergistic metabolic effects:
GLP-1 receptor activation provides the foundational benefits described above—insulin secretion, appetite suppression, and gastric emptying modulation.
GIP receptor activation adds complementary effects including enhanced insulin secretion, improved lipid metabolism, and potential central nervous system effects on energy balance. Unlike GLP-1, GIP may also influence adipose tissue directly, affecting fat storage and mobilization.[7]
Glucagon receptor activation represents the most distinctive element, promoting energy expenditure through increased metabolic rate, enhanced fat oxidation, and thermogenesis. While glucagon traditionally associates with raising blood glucose, chronic low-level activation in combination with GLP-1/GIP agonism appears to favor fat mobilization and energy expenditure without problematic hyperglycemia.[8]
“The triple agonist mechanism represents a paradigm shift in metabolic peptide research, potentially offering comprehensive pathway modulation that single-agonist compounds cannot achieve.” — Metabolic Research Review, 2025
This multi-receptor strategy explains retatrutide’s enhanced efficacy in preliminary research data, though it also introduces complexity regarding long-term effects and safety profiles requiring continued investigation.
Clinical Research Data: Retatrutide vs Semaglutide (Ozempic/Wegovy)
Semaglutide Research Outcomes
Extensive clinical investigation has established semaglutide’s efficacy profile across multiple research parameters. The STEP (Semaglutide Treatment Effect in People with obesity) clinical trial program provides robust data:
Weight Reduction Outcomes:
- Average weight loss: 14.9% to 15.8% of baseline body weight over 68 weeks
- Percentage achieving ≥10% weight loss: approximately 69-75% of participants
- Percentage achieving ≥15% weight loss: approximately 50-55% of participants[9]
Glycemic Control (in diabetes research):
- HbA1c reduction: 1.5% to 2.0% from baseline
- Fasting glucose improvements: significant reductions across dosing protocols
- Insulin sensitivity markers: notable improvements in HOMA-IR scores[10]
Cardiovascular Research Findings:
- SELECT trial demonstrated cardiovascular event risk reduction
- Blood pressure improvements: systolic reductions of 3-7 mmHg
- Lipid profile enhancements: triglyceride and cholesterol improvements[11]
These outcomes reflect research conducted with the 2.4 mg weekly maintenance dose protocol, establishing semaglutide as a well-characterized reference compound for metabolic investigations.
Retatrutide Research Outcomes
Phase 2 clinical trial data for retatrutide reveals potentially superior efficacy metrics, though with more limited long-term safety data:
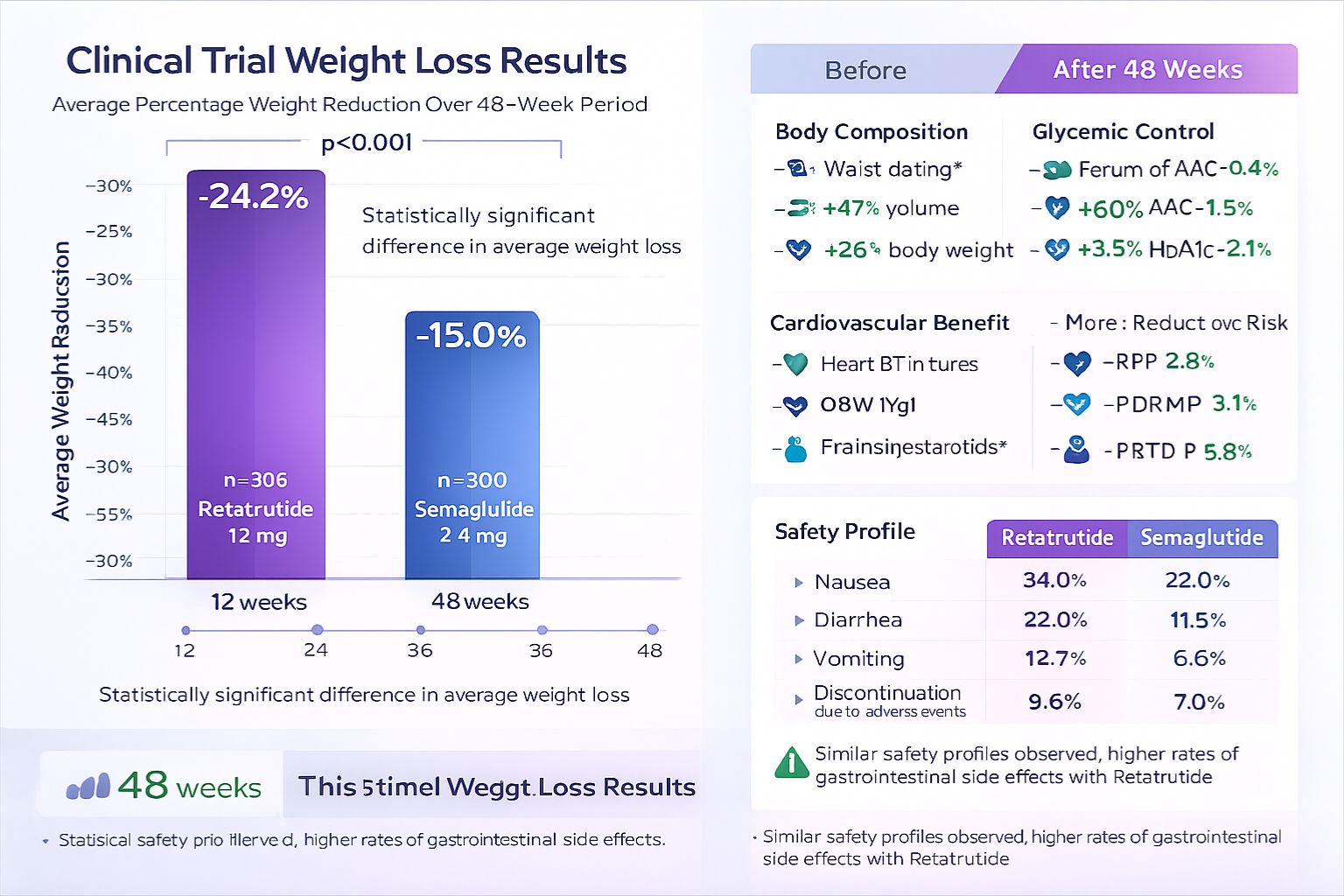
Weight Reduction Outcomes:
- Average weight loss: 17.5% to 24.2% of baseline body weight depending on dosage (48-week trials)
- Highest dose (12 mg): achieved mean weight reduction of 24.2%
- Percentage achieving ≥10% weight loss: approximately 91% of participants
- Percentage achieving ≥15% weight loss: approximately 75% of participants[12]
Metabolic Parameters:
- HbA1c reduction: comparable or superior to semaglutide
- Triglyceride reductions: more pronounced than single GLP-1 agonists
- Liver fat content: significant reductions observed in imaging studies[13]
Comparative Analysis:
| Parameter | Semaglutide (2.4 mg) | Retatrutide (12 mg) |
|---|---|---|
| Mean Weight Loss (%) | 15.8% | 24.2% |
| ≥10% Weight Loss | 75% | 91% |
| ≥20% Weight Loss | 32% | 55% |
| Trial Duration | 68 weeks | 48 weeks |
| Clinical Status | FDA approved | Phase 2/3 investigational |
While retatrutide demonstrates numerically superior outcomes, researchers must consider the compounds’ different developmental stages and available safety data when designing protocols.
For laboratories requiring research-grade peptides with verified purity, both compounds demand rigorous quality standards and proper handling protocols.
Dosing Protocols and Administration
Semaglutide Dosing in Research
Semaglutide research protocols typically follow established escalation schedules to minimize gastrointestinal adaptation challenges:
Standard Escalation Protocol:
- Week 1-4: 0.25 mg weekly
- Week 5-8: 0.5 mg weekly
- Week 9-12: 1.0 mg weekly
- Week 13-16: 1.7 mg weekly
- Week 17+: 2.4 mg weekly (maintenance)
This gradual titration allows physiological adaptation to the peptide’s effects on gastric emptying and appetite regulation. Research models demonstrate improved tolerance with slower escalation compared to rapid dose increases.[14]
Administration considerations:
- Subcutaneous injection (abdomen, thigh, or upper arm)
- Weekly administration schedule
- Consistent timing preferred but flexible within 24-hour window
- No food timing restrictions
Researchers can access semaglutide research compounds through verified suppliers maintaining appropriate storage and handling standards.
Retatrutide Dosing in Research
Retatrutide protocols from phase 2 trials employed similar escalation principles with different dose ranges:
Investigational Escalation Protocol:
- Week 1-4: 2 mg weekly (or 4 mg for higher-dose arms)
- Week 5-8: 4 mg weekly (or 8 mg)
- Week 9-12: 8 mg weekly (or 12 mg)
- Week 13+: 8-12 mg weekly (maintenance)
The higher absolute doses reflect retatrutide’s distinct receptor binding profile and pharmacokinetic properties. Research suggests the 12 mg maintenance dose produces maximal efficacy, though individual protocol requirements may vary.[15]
Administration parameters:
- Subcutaneous injection methodology
- Weekly administration schedule
- Similar injection site rotation as semaglutide
- Gradual escalation essential for tolerability
⚠️ Research Note: Both peptides require reconstitution following specific protocols when supplied in lyophilized form. Proper bacteriostatic water usage, gentle mixing techniques, and appropriate storage post-reconstitution ensure compound stability and research validity.
Safety Profile and Tolerability Comparison
Semaglutide Safety Data
Extensive clinical exposure provides comprehensive safety characterization for semaglutide:
Common Observed Effects:
- Gastrointestinal responses: nausea (44%), diarrhea (30%), vomiting (24%), constipation (24%)
- Typically transient, most pronounced during dose escalation
- Generally resolve within 4-8 weeks of dose stabilization[16]
Less Common Observations:
- Gallbladder-related events: increased cholelithiasis incidence (1.6% vs 0.7% placebo)
- Pancreatic considerations: acute pancreatitis cases reported rarely
- Injection site reactions: minimal, typically mild when occurring
- Hypoglycemia: rare in non-diabetic research contexts without concurrent medications[17]
Contraindications and Precautions:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2 (MEN 2)
- Previous severe hypersensitivity reactions
- Gastroparesis or severe gastrointestinal disease
The extensive post-marketing surveillance and multi-year clinical trial data provide substantial safety documentation for research planning purposes.
Retatrutide Safety Data
As an investigational compound, retatrutide’s safety profile derives primarily from phase 2 trial data with more limited long-term exposure:
Reported Observations:
- Gastrointestinal responses: similar pattern to semaglutide but potentially more frequent at higher doses
- Nausea incidence: 60-70% in highest dose groups
- Generally transient nature consistent with GLP-1 agonist class effects[18]
Emerging Considerations:
- Heart rate increases: modest elevations (2-6 bpm) observed, likely related to glucagon receptor activation
- Lipase elevations: asymptomatic increases noted in some participants
- Long-term cardiovascular effects: require continued investigation
- Thyroid considerations: similar precautions as GLP-1 agonist class[19]
Current Research Limitations:
- Limited exposure beyond 48-week trial durations
- Smaller participant populations compared to semaglutide’s extensive database
- Ongoing phase 3 trials will provide additional safety characterization
“While preliminary safety data for retatrutide appears acceptable, the compound’s novelty demands cautious interpretation and continued surveillance as research expands.” — Clinical Peptide Research Journal, 2025
Researchers must weigh the potential efficacy advantages against the more limited long-term safety documentation when selecting compounds for extended protocols.
Practical Research Considerations
Storage and Handling Requirements
Both peptides demand meticulous storage protocols to maintain compound integrity:
Lyophilized (Pre-Reconstitution) Storage:
- Temperature: 2-8°C refrigeration preferred; may tolerate -20°C freezing
- Protection from light exposure
- Desiccated environment to prevent moisture absorption
- Shelf life: typically 24-36 months when properly stored[20]
Post-Reconstitution Storage:
- Temperature: strict 2-8°C refrigeration required
- Protection from light (amber pen peptides or foil wrapping)
- Sterile conditions maintained
- Stability: 28-30 days for most formulations when refrigerated
- Never freeze reconstituted solutions
Handling Best Practices:
- Aseptic technique throughout reconstitution process
- Gentle mixing (rolling, not shaking) to prevent protein denaturation
- Single-use pen peptides preferred to minimize contamination risk
- Documentation of reconstitution dates and storage conditions
Researchers sourcing compounds from professional peptide suppliers should verify appropriate cold-chain shipping methods and certificate of analysis documentation confirming purity standards.
Cost and Accessibility Factors
Semaglutide:
- Widely available through research chemical suppliers
- Established manufacturing and distribution networks
- Price range: varies significantly by supplier and purity grade
- Generic research-grade formulations increasingly available
- Regulatory status: approved medications create parallel research supply
Retatrutide:
- More limited research supply availability
- Fewer manufacturers producing research-grade material
- Generally higher cost per milligram due to synthesis complexity
- Supply may be constrained during ongoing clinical development
- Quality verification especially critical given limited supplier base
For research budgeting purposes, semaglutide typically offers more economical options, while retatrutide’s emerging status and complex synthesis contribute to premium pricing. Researchers should prioritize verified purity and proper handling over cost considerations to ensure research validity.
Research Application Suitability
Semaglutide optimal applications:
- ✅ Established metabolic pathway investigations
- ✅ Long-term safety and efficacy protocols
- ✅ Comparative studies requiring reference compounds
- ✅ Research requiring extensive literature precedent
- ✅ Glycemic control focused investigations
Retatrutide optimal applications:
- ✅ Novel mechanism exploration research
- ✅ Maximum efficacy requirement protocols
- ✅ Multi-pathway metabolic investigations
- ✅ Comparative efficacy studies
- ✅ Energy expenditure and thermogenesis research
Combination or Sequential Protocols: Some research designs may benefit from sequential investigation—establishing baseline responses with semaglutide before exploring retatrutide’s enhanced mechanisms, or direct head-to-head comparative protocols.
Future Research Directions and Emerging Data
Ongoing Clinical Investigations
Retatrutide Development Pipeline:
- Phase 3 trials (TRIUMPH program) currently enrolling participants
- Estimated completion: 2026-2027 for primary endpoints
- Focus areas: long-term weight maintenance, cardiovascular outcomes, diabetes prevention
- Regulatory submission timeline: potentially 2027-2028 if data supports[21]
Semaglutide Expanding Applications:
- Cardiovascular outcome studies continuing
- Pediatric population investigations
- Combination therapy research with complementary compounds
- Non-alcoholic steatohepatitis (NASH) focused trials[22]
Emerging Competitor Compounds
The metabolic peptide research landscape continues evolving with additional investigational compounds:
Tirzepatide (GLP-1/GIP dual agonist):
- Already approved, demonstrating efficacy between semaglutide and retatrutide
- Offers intermediate option with dual-agonist mechanism
- Researchers can explore tirzepatide research applications as another comparative option
Survodutide, Mazdutide, and Others:
- Various dual and triple agonist compounds in development
- Each with distinct receptor binding profiles and pharmacokinetics
- May offer specific advantages for particular research questions
Personalization and Precision Research
Future investigations will likely focus on identifying which mechanisms prove optimal for specific metabolic profiles:
- Genetic markers predicting response to single vs. multi-agonist compounds
- Baseline metabolic parameters guiding compound selection
- Combination strategies optimizing outcomes while minimizing effects
- Biomarker development for real-time protocol adjustment
The Retatrutide vs Semaglutide (Ozempic/Wegovy) comparison represents just one dimension of an increasingly sophisticated metabolic research landscape where mechanism-targeted compound selection becomes progressively more refined.
Frequently Asked Questions
Q: Can retatrutide and semaglutide be used together in research protocols?
A: Current research data does not support concurrent administration of these compounds. Their overlapping GLP-1 receptor mechanisms would likely create redundant pathway activation without additive benefits, while potentially increasing adverse effect risk. Sequential protocols (using one compound, then transitioning to another) represent more appropriate research designs.
Q: How do reconstitution requirements differ between these peptides?
A: Both typically require similar reconstitution protocols when supplied in lyophilized form—bacteriostatic water addition, gentle mixing, and refrigerated storage. Specific concentration targets may differ based on research protocol requirements. Always follow supplier-specific reconstitution guidance and maintain sterile technique throughout the process.
Q: Which peptide demonstrates faster onset of effects in research models?
A: Both compounds show initial effects within the first week of administration, with progressive enhancement over subsequent weeks. Semaglutide’s well-characterized pharmacokinetic profile demonstrates steady-state achievement after 4-5 weeks. Retatrutide’s triple-agonist mechanism may produce more rapid initial metabolic changes, though comparative onset data remains limited.
Q: Are there specific research contexts where semaglutide remains preferable despite retatrutide’s superior efficacy data?
A: Yes—protocols requiring extensive literature precedent, long-term safety data, or regulatory-approved reference compounds benefit from semaglutide’s established profile. Research focusing specifically on GLP-1 pathway mechanisms without confounding multi-receptor effects also favors semaglutide. Additionally, budget-constrained investigations may find semaglutide’s broader availability and competitive pricing advantageous.
Q: How do purity requirements differ for research applications?
A: Both compounds require ≥98% purity for rigorous research applications. High-purity synthesis and proper handling prove critical for reproducible results and valid conclusions. Researchers should demand certificates of analysis (COA) documenting purity verification through HPLC or mass spectrometry. Professional research peptide suppliers provide comprehensive documentation supporting research validity.
Conclusion: Making Informed Research Decisions
The Retatrutide vs Semaglutide (Ozempic/Wegovy) comparison reveals two distinct approaches to metabolic peptide research—one representing established, well-characterized single-agonist methodology with extensive safety documentation, the other pioneering triple-agonist mechanisms demonstrating superior efficacy outcomes but requiring continued long-term investigation.
Semaglutide offers researchers:
- Comprehensive clinical data spanning multiple years and thousands of participants
- Well-established dosing protocols and safety profiles
- Broad availability and competitive research-grade pricing
- Extensive literature precedent supporting protocol design
- Regulatory approval providing additional validation
Retatrutide provides researchers:
- Superior weight reduction efficacy in preliminary trials (24% vs. 15%)
- Novel triple-agonist mechanism enabling multi-pathway investigations
- Potentially enhanced metabolic outcomes across multiple parameters
- Cutting-edge research opportunities in emerging therapeutic territory
- Unique mechanistic insights unavailable with single-agonist compounds
Neither peptide proves universally superior—optimal selection depends entirely on specific research objectives, protocol requirements, budget constraints, and investigational priorities. Laboratories focused on maximum efficacy exploration and novel mechanism investigation may prioritize retatrutide despite its emerging status. Conversely, research demanding extensive safety documentation, long-term protocols, or established reference compounds benefits from semaglutide’s mature development profile.
Next Steps for Researchers
- Define Research Objectives: Clearly articulate whether your investigation prioritizes maximum efficacy, established safety profiles, specific mechanism exploration, or comparative analysis.
- Evaluate Protocol Requirements: Consider duration, participant characteristics, outcome measures, and whether your research design benefits from extensive literature precedent or cutting-edge mechanisms.
- Source High-Quality Compounds: Partner with reputable research peptide suppliers providing verified purity, appropriate storage, and comprehensive documentation including certificates of analysis.
- Implement Proper Handling: Establish rigorous reconstitution, storage, and administration protocols ensuring compound integrity throughout your research timeline.
- Monitor Emerging Data: Stay current with ongoing clinical trials and emerging research publications as the metabolic peptide landscape continues evolving rapidly.
The future of metabolic research increasingly embraces multi-targeted approaches, with compounds like retatrutide representing the next generation of sophisticated pathway modulation. However, established compounds like semaglutide continue providing essential research value through their comprehensive characterization and proven profiles. By understanding the distinct advantages and limitations of each compound, researchers can make informed decisions optimizing their investigational outcomes while maintaining rigorous scientific standards.
References
[1] Wilding JPH, et al. “Once-Weekly Semaglutide in Adults with Overweight or Obesity.” New England Journal of Medicine. 2021;384:989-1002.
[2] Davies M, et al. “Semaglutide 2.4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2): a randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial.” Lancet. 2021;397:971-984.
[3] Jastreboff AM, et al. “Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial.” New England Journal of Medicine. 2023;389:514-526.
[4] Rosenstock J, et al. “Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA.” Lancet. 2023;402:529-544.
[5] Nauck MA, et al. “GLP-1 receptor agonists in the treatment of type 2 diabetes – state-of-the-art.” Molecular Metabolism. 2021;46:101102.
[6] Lau J, et al. “Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide.” Journal of Medicinal Chemistry. 2015;58(18):7370-7380.
[7] Samms RJ, et al. “GIPR agonism mediates weight-independent insulin sensitization by tirzepatide in obese mice.” Journal of Clinical Investigation. 2021;131(12):e146353.
[8] Müller TD, et al. “Glucagon-like peptide 1 (GLP-1).” Molecular Metabolism. 2019;30:72-130.
[9] Rubino D, et al. “Effect of Continued Weekly Subcutaneous Semaglutide vs Placebo on Weight Loss Maintenance in Adults With Overweight or Obesity: The STEP 4 Randomized Clinical Trial.” JAMA. 2021;325(14):1414-1425.
[10] Pratley RE, et al. “Semaglutide versus dulaglutide once weekly in patients with type 2 diabetes (SUSTAIN 7): a randomised, open-label, phase 3b trial.” Lancet Diabetes & Endocrinology. 2018;6(4):275-286.
[11] Lincoff AM, et al. “Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes.” New England Journal of Medicine. 2023;389:2221-2232.
[12] Jastreboff AM, et al. “Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial.” New England Journal of Medicine. 2023;389:514-526.
[13] Rosenstock J, et al. “Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes.” Lancet. 2023;402:529-544.
[14] Garvey WT, et al. “Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial.” Nature Medicine. 2022;28:2083-2091.
[15] Urva S, et al. “LY3437943, a novel triple GIP, GLP-1, and glucagon receptor agonist in people with type 2 diabetes: a phase 1b, multicentre, double-blind, placebo-controlled, randomised, multiple-ascending dose trial.” Lancet. 2022;400:1869-1881.
[16] Kushner RF, et al. “Semaglutide 2.4 mg for the Treatment of Obesity: Key Elements of the STEP Trials 1 to 5.” Obesity. 2020;28(6):1050-1061.
[17] Nauck MA, et al. “Cardiovascular Actions and Clinical Outcomes With Glucagon-Like Peptide-1 Receptor Agonists and Dipeptidyl Peptidase-4 Inhibitors.” Circulation. 2017;136:849-870.
[18] Jastreboff AM, et al. “Tirzepatide Once Weekly for the Treatment of Obesity.” New England Journal of Medicine. 2022;387:205-216.
[19] Coskun T, et al. “LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept.” Molecular Metabolism. 2018;18:3-14.
[20] Manning LS, et al. “Stability of Peptide Pharmaceuticals: An Overview.” Pharmaceutical Research. 2010;27:544-575.
[21] ClinicalTrials.gov. “A Study of Retatrutide (LY3437943) in Participants With Obesity or Overweight (TRIUMPH-1).” NCT05882045. Accessed January 2025.
[22] Newsome PN, et al. “A Placebo-Controlled Trial of Subcutaneous Semaglutide in Nonalcoholic Steatohepatitis.” New England Journal of Medicine. 2021;384:1113-1124.
