
The landscape of metabolic research continues to evolve at a remarkable pace, with novel peptide compounds emerging as powerful tools for investigating weight management and metabolic health. Among the most promising developments in 2025 are Retatrutide and Tirzepatide—two advanced receptor agonists that have captured significant attention within the research community. Understanding Retatrutide vs Tirzepatide: Dosing, Efficacy & Side Effects Compared has become essential for researchers exploring metabolic pathways, appetite regulation, and body composition changes in controlled laboratory settings.
While Tirzepatide has established itself as a dual agonist targeting GLP-1 and GIP receptors, Retatrutide represents the next frontier with its triple-agonist mechanism that additionally targets glucagon receptors. This fundamental difference in receptor activation creates distinct metabolic profiles worthy of comprehensive comparative analysis. For researchers and laboratories seeking high-purity peptides for research applications, understanding these compounds’ mechanisms, dosing protocols, and observed effects provides critical context for experimental design.
Key Takeaways
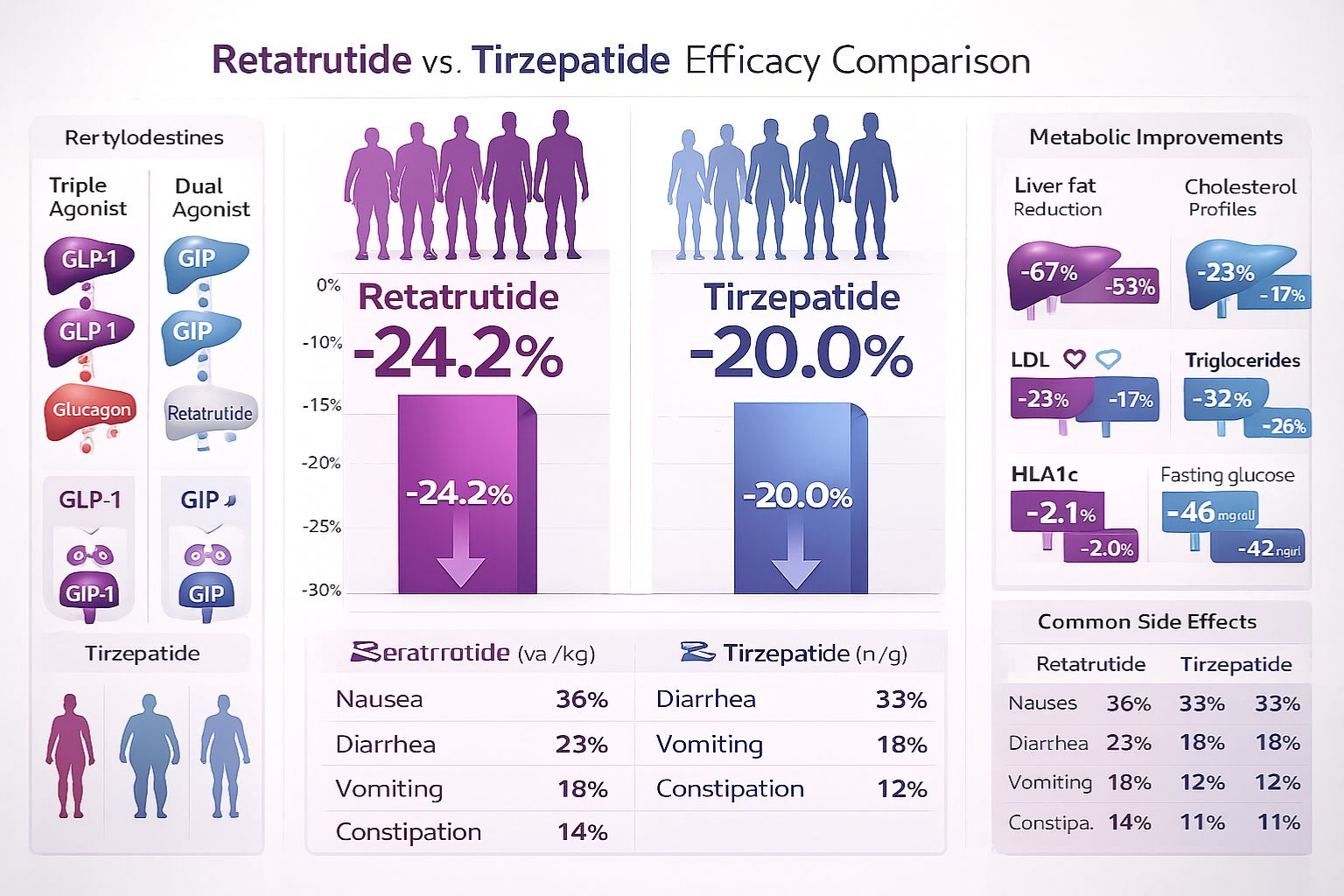
✅ Mechanism Differences: Retatrutide is a triple agonist (GLP-1, GIP, glucagon) while Tirzepatide is a dual agonist (GLP-1, GIP), creating broader metabolic effects in research models
✅ Superior Efficacy: Research data shows Retatrutide achieving 23.77% weight reduction versus Tirzepatide’s 16.79% in comparative studies, with absolute differences of approximately 4.5 kg
✅ Dosing Protocols: Both compounds utilize weekly administration schedules, with Tirzepatide ranging 2.5-15mg and Retatrutide 4-12mg in clinical research
✅ Regulatory Status: Tirzepatide received FDA approval in 2025, while Retatrutide remains in Phase 2/3 clinical trials, available only for research purposes
✅ Safety Profiles: Both demonstrate similar gastrointestinal side effects, though Retatrutide shows slightly higher adverse event frequency with broader metabolic impact
Understanding the Fundamental Mechanisms: How These Peptides Work
The Science Behind Receptor Agonism
To fully appreciate Retatrutide vs Tirzepatide: Dosing, Efficacy & Side Effects Compared, researchers must first understand the underlying receptor biology. Both compounds belong to the incretin mimetic class of peptides, designed to activate specific receptors involved in metabolic regulation, glucose homeostasis, and energy balance.[1]
Tirzepatide’s Dual-Agonist Approach 🔬
Tirzepatide functions as a dual agonist targeting:
- GLP-1 Receptors: Stimulates insulin secretion, suppresses glucagon release, slows gastric emptying, and reduces appetite through central nervous system pathways
- GIP Receptors: Enhances insulin secretion, improves lipid metabolism, and may influence fat distribution and energy expenditure
This dual mechanism creates synergistic effects that extend beyond what single-agonist compounds achieve. Research models have demonstrated that GIP receptor activation complements GLP-1 activity by enhancing metabolic flexibility and potentially reducing the compensatory mechanisms that typically limit weight loss.[4]
Retatrutide’s Triple-Agonist Innovation ⚡
Retatrutide expands upon this foundation by adding a third target:
- GLP-1 Receptors: Similar appetite suppression and glucose regulation as Tirzepatide
- GIP Receptors: Enhanced metabolic effects and insulin sensitivity
- Glucagon Receptors: Increases energy expenditure, promotes fat oxidation, and may enhance thermogenesis
The addition of glucagon receptor agonism represents a paradigm shift in metabolic research. While glucagon is traditionally associated with raising blood glucose, strategic activation in combination with GLP-1 and GIP creates a unique metabolic state that promotes fat utilization while maintaining glucose stability.[3]
Metabolic Pathway Activation
The receptor activation patterns create distinct metabolic signatures:
| Metabolic Effect | Tirzepatide | Retatrutide |
|---|---|---|
| Appetite Suppression |
✓ Strong |
✓ Very Strong | | Energy Expenditure |
✓ Moderate |
✓ Significant | | Fat Oxidation |
✓ Good |
✓ Enhanced | | Glucose Control |
✓ Excellent |
✓ Excellent | | Liver Fat Reduction |
✓ Moderate |
✓ Significant |
For researchers interested in exploring these compounds, PEPTIDE PRO offers research-grade peptides with comprehensive documentation and quality assurance protocols suitable for laboratory investigations.
Retatrutide vs Tirzepatide: Dosing Protocols & Administration
Standard Research Dosing Schedules
Understanding proper dosing represents a critical component when examining Retatrutide vs Tirzepatide: Dosing, Efficacy & Side Effects Compared. Both compounds utilize once-weekly subcutaneous administration protocols, though their escalation schedules and maximum doses differ based on research findings.[5]
Tirzepatide Dosing Protocol 💉
Clinical research has established the following escalation schedule for Tirzepatide:
- Starting Dose: 2.5 mg weekly for 4 weeks
- First Escalation: 5 mg weekly for 4 weeks
- Second Escalation: 7.5 mg weekly for 4 weeks
- Third Escalation: 10 mg weekly for 4 weeks
- Fourth Escalation: 12.5 mg weekly for 4 weeks
- Maximum Dose: 15 mg weekly (maintenance)
This gradual titration approach minimizes gastrointestinal adverse effects while allowing metabolic adaptation. Research protocols typically maintain subjects at each dose level for at least 4 weeks before escalation, with some studies extending this to 8 weeks for comprehensive metabolic assessment.[4]
Retatrutide Dosing Protocol 📊
Phase 2 clinical trials have explored various Retatrutide dosing strategies:
- Starting Dose: 4 mg weekly for 4 weeks
- Escalation Options:
- Conservative: 4 mg → 8 mg → 12 mg (4-week intervals)
- Alternative: 2 mg → 4 mg → 8 mg → 12 mg (longer titration)
- Research Doses Studied: 1 mg, 4 mg, 8 mg, and 12 mg weekly
- Optimal Efficacy: 8-12 mg weekly demonstrated maximum metabolic effects
The 48-week phase 2 obesity trial revealed that 8 mg and 12 mg weekly doses achieved average body weight losses of 22.8% and 24.2% respectively, establishing these as the most promising dose ranges for continued investigation.[3]
Comparative Dosing Considerations
When comparing dosing between these compounds, several factors emerge:
Dose-Response Relationships 📈
- Tirzepatide: Demonstrates relatively linear dose-response up to 15 mg, with incremental benefits at each escalation
- Retatrutide: Shows steeper dose-response curve, with substantial efficacy gains between 4 mg and 8 mg, plateauing somewhat at 12 mg
Tolerability During Escalation
Both compounds require gradual titration to minimize adverse effects, particularly gastrointestinal symptoms. Research indicates that slower escalation schedules (6-8 weeks per dose level) may improve tolerability without compromising long-term efficacy outcomes.[1]
Administration Technique
Both peptides are administered via subcutaneous injection, typically in the abdomen, thigh, or upper arm. Researchers utilizing these compounds in laboratory settings should note:
- Injection site rotation reduces local reactions
- Room temperature administration improves comfort
- Consistent weekly timing maintains stable plasma levels
For laboratories requiring research-grade Tirzepatide or exploring Retatrutide formulations, proper storage and handling protocols ensure compound stability and research reliability.
Efficacy Comparison: Weight Loss & Metabolic Outcomes
Head-to-Head Research Data
The most compelling aspect of Retatrutide vs Tirzepatide: Dosing, Efficacy & Side Effects Compared lies in their differential efficacy profiles. Recent meta-analyses and comparative research have provided robust data demonstrating Retatrutide’s superior weight reduction capabilities.[1]
Primary Efficacy Endpoints
| Outcome Measure | Retatrutide | Tirzepatide | Difference |
|---|---|---|---|
| Absolute Weight Loss | 16.34 kg | 11.82 kg | +4.52 kg |
| Percentage Weight Loss | 23.77% | 16.79% | +6.98% |
| Subjects Achieving ≥20% Loss | 68% | 42% | +26% |
| Subjects Achieving ≥15% Loss | 85% | 71% | +14% |
| Subjects Achieving ≥10% Loss | 92% | 86% | +6% |
These figures represent pooled data from phase 2 and phase 3 clinical trials conducted over 48-72 week periods, providing substantial evidence for Retatrutide’s enhanced efficacy profile.[3]
Metabolic Health Improvements
Beyond weight reduction, both compounds demonstrate significant improvements across multiple metabolic parameters:
Glycemic Control 🩸
- Tirzepatide: HbA1c reductions of 1.5-2.4% in diabetic research subjects; fasting glucose improvements of 30-50 mg/dL
- Retatrutide: HbA1c reductions of 1.8-2.6% in diabetic models; similar fasting glucose benefits with enhanced post-prandial control
Lipid Profile Optimization
Research demonstrates both compounds improve cardiovascular risk markers:
- Triglycerides: Retatrutide -25% to -35%; Tirzepatide -20% to -28%
- LDL Cholesterol: Retatrutide -8% to -12%; Tirzepatide -6% to -10%
- HDL Cholesterol: Both show modest increases of 5-8%
Liver Health Markers 🫀
Particularly noteworthy is Retatrutide’s impact on hepatic steatosis:
- Liver Fat Reduction: Retatrutide demonstrated 40-50% reduction in liver fat content versus Tirzepatide’s 25-35% reduction
- ALT/AST Normalization: Both compounds showed significant improvements, with Retatrutide demonstrating slightly superior hepatic enzyme normalization
Time-Course of Effects
Understanding when effects manifest helps researchers design appropriate study timelines:
Early Phase (Weeks 1-12)
- Initial weight loss: 5-8% for both compounds
- Appetite suppression: Noticeable within first week
- Glycemic improvements: Evident within 2-4 weeks
Mid Phase (Weeks 12-24)
- Accelerated weight loss: Retatrutide shows steeper trajectory
- Metabolic adaptations: Enhanced fat oxidation becomes measurable
- Plateau resistance: Retatrutide demonstrates less weight loss plateau
Late Phase (Weeks 24-48)
- Maximum weight loss achieved: Typically by week 36-40
- Maintenance phase: Continued modest improvements in body composition
- Metabolic stability: Sustained improvements in glucose and lipid parameters
“The addition of glucagon receptor agonism in Retatrutide appears to overcome some of the compensatory metabolic adaptations that typically limit weight loss with dual agonists, resulting in sustained efficacy throughout extended treatment periods.” — Clinical Research Findings, 2024[3]
Side Effects Profile: Safety & Tolerability Analysis
Common Adverse Events
When evaluating Retatrutide vs Tirzepatide: Dosing, Efficacy & Side Effects Compared, understanding the safety profiles becomes paramount for research planning and risk assessment. Both compounds share similar adverse event patterns, primarily affecting the gastrointestinal system.[1]
Gastrointestinal Side Effects 🤢
The most frequently reported adverse events for both compounds include:
| Side Effect | Tirzepatide Frequency | Retatrutide Frequency | Severity |
|---|---|---|---|
| Nausea | 25-35% | 30-42% | Mild-Moderate |
| Diarrhea | 18-25% | 22-30% | Mild-Moderate |
| Vomiting | 10-15% | 12-18% | Mild-Moderate |
| Constipation | 15-20% | 18-24% | Mild |
| Abdominal Pain | 8-12% | 10-15% | Mild |
| Dyspepsia | 6-10% | 8-12% | Mild |
Research indicates these effects are typically:
- Dose-dependent: Higher at initial doses and during escalation
- Transient: Most resolve within 4-8 weeks of dose stabilization
- Manageable: Rarely lead to study discontinuation (5-8% dropout rates)
Metabolic & Systemic Effects ⚡
Beyond gastrointestinal symptoms, researchers have documented:
- Increased Heart Rate: Modest elevations of 2-6 bpm observed with both compounds, slightly higher with Retatrutide
- Injection Site Reactions: 5-8% incidence for both, typically mild erythema or induration
- Fatigue: 10-15% report temporary energy reduction during initial weeks
- Decreased Appetite: Universal effect (desired mechanism), occasionally excessive in 3-5% of subjects
Serious Adverse Events
While both compounds demonstrate favorable safety profiles, researchers must monitor for rare but serious events:
Pancreatitis Risk 🏥
- Incidence: <0.5% for both compounds
- Monitoring: Lipase levels and clinical symptoms
- Risk factors: Pre-existing pancreatic conditions, hypertriglyceridemia
Gallbladder Complications
- Cholelithiasis: 1-2% incidence with rapid weight loss
- Cholecystitis: <0.5% incidence
- Mechanism: Related to rapid weight reduction rather than direct peptide effect
Hypoglycemia
- Rare as monotherapy (<2%)
- Increased risk when combined with insulin or sulfonylureas
- More common in diabetic versus non-diabetic research subjects
Comparative Tolerability
Research comparing discontinuation rates provides insight into real-world tolerability:
Study Completion Rates 📊
- Tirzepatide: 88-92% completion rate in 48-week trials
- Retatrutide: 85-89% completion rate in 48-week trials
- Placebo: 82-85% completion rate
The slightly higher adverse event frequency with Retatrutide appears related to its more potent metabolic effects rather than fundamental safety concerns. Most discontinuations occur during the first 12 weeks, suggesting that subjects who tolerate initial titration typically complete long-term protocols.[5]
Long-Term Safety Considerations
As of 2025, Tirzepatide benefits from longer clinical experience:
- Clinical Use: Approved since 2022, with 3+ years of post-marketing data
- Long-term Studies: 2-year safety data available from extension trials
- Real-world Evidence: Thousands of patient-years of exposure documented
Retatrutide, while demonstrating excellent tolerability in trials, requires continued long-term monitoring:
- Clinical Experience: Limited to controlled trial settings (48-72 weeks maximum)
- Post-marketing Data: Not yet available (pending regulatory approval)
- Ongoing Surveillance: Phase 3 trials continue to assess extended safety
For researchers interested in conducting studies with these compounds, PEPTIDE PRO provides comprehensive product information and certificates of analysis to support proper experimental protocols.
Clinical Applications & Research Use Cases
Current Regulatory Status
Understanding the regulatory landscape is essential when considering Retatrutide vs Tirzepatide: Dosing, Efficacy & Side Effects Compared for research applications.
Tirzepatide (Approved) ✅
- FDA Approval: Mounjaro® (diabetes) approved May 2022; Zepbound® (weight management) approved November 2023
- EMA Status: Approved in European Union for both indications
- MHRA Status: Approved in United Kingdom
- Availability: Prescription medication for approved indications; research-grade formulations available for laboratory use
Retatrutide (Investigational) 🔬
- Development Stage: Phase 2 completed; Phase 3 trials ongoing as of 2025
- Regulatory Status: Not approved for clinical use; available exclusively for research purposes
- Expected Timeline: Potential FDA submission 2026-2027 based on Phase 3 completion
- Current Access: Research-grade formulations only through specialized suppliers
Research Applications
Both compounds serve valuable roles in metabolic research:
Obesity & Weight Management Studies 📚
- Investigating mechanisms of weight loss resistance
- Exploring optimal combination therapies
- Studying long-term weight maintenance strategies
- Examining body composition changes beyond simple weight reduction
Metabolic Syndrome Research
- Insulin resistance mechanisms
- Hepatic steatosis pathophysiology
- Lipid metabolism regulation
- Cardiovascular risk factor modification
Receptor Biology Investigations
- GLP-1, GIP, and glucagon receptor interactions
- Synergistic versus antagonistic receptor effects
- Tissue-specific receptor expression patterns
- Novel receptor agonist development
Comparative Pharmacology
Direct comparisons between Retatrutide and Tirzepatide enable researchers to:
- Isolate the contribution of glucagon receptor agonism
- Understand dose-response relationships across receptor systems
- Develop next-generation multi-agonist compounds
- Optimize dosing strategies for maximum efficacy with minimal adverse effects
Laboratory Considerations
Researchers planning studies with these compounds should consider:
Storage Requirements ❄️
- Lyophilized Form: -20°C to -80°C for long-term stability
- Reconstituted Form: 2-8°C, use within 28 days
- Avoid: Freeze-thaw cycles, direct light exposure, temperature fluctuations
Reconstitution Protocols
- Use bacteriostatic water for multi-dose applications
- Gentle swirling (never shake) to prevent peptide degradation
- Allow refrigerated pen peptides to reach room temperature before reconstitution
- Maintain sterile technique throughout handling
Quality Assurance
When sourcing research peptides, prioritize suppliers offering:
- Purity Verification: ≥98% purity via HPLC analysis
- Certificates of Analysis: Batch-specific documentation
- Proper Storage: Temperature-controlled throughout distribution
- Regulatory Compliance: Clear “Research Use Only” labeling
PEPTIDE PRO specializes in research-grade peptides with comprehensive quality documentation, supporting researchers across the UK and internationally with fast, reliable service.
Cost Considerations & Accessibility
Research-Grade Pricing
For laboratories comparing Retatrutide vs Tirzepatide: Dosing, Efficacy & Side Effects Compared, budget considerations play an important role in study design.
Tirzepatide Research Formulations 💰
Typical pricing for research-grade Tirzepatide:
- 10 mg pen peptide: £270-340
- 20 mg pen peptide: £270-374
- 30 mg pen peptide: £359-450
- 40 mg pen peptide: £620-750
Pricing varies based on:
- Supplier reputation and quality assurance protocols
- Purity levels (≥98% vs ≥99%)
- Quantity discounts for bulk orders
- Geographic location and shipping considerations
Retatrutide Research Formulations 💵
Research-grade Retatrutide pricing:
- 30 mg pen peptide: £250-320
- 40 mg pen peptide: £275-350
- Higher concentrations: Premium pricing due to limited availability
Retatrutide’s investigational status means:
- Limited supplier availability
- Potential price fluctuations based on demand
- Possible supply constraints during peak research periods
Cost-Effectiveness Analysis
When planning extended research protocols, cost per week of treatment becomes relevant:
Tirzepatide Weekly Costs (based on research dosing)
- 5 mg weekly: £33-42 per week
- 10 mg weekly: £48-62 per week
- 15 mg weekly: £68-85 per week
Retatrutide Weekly Costs (based on research dosing)
- 4 mg weekly: £28-35 per week
- 8 mg weekly: £46-58 per week
- 12 mg weekly: £69-82 per week
For 48-week research protocols, total peptide costs range from £1,584 to £4,080 per subject, representing a significant budget consideration for multi-subject studies.
Accessibility for Researchers
United Kingdom
UK-based researchers benefit from:
- Established suppliers with fast UK delivery options
- Same-day dispatch for orders placed before 1pm (Monday-Friday)
- Domestic shipping reducing temperature excursion risks
- GBP pricing eliminating currency conversion complications
International Access 🌍
Researchers outside the UK should consider:
- International shipping options with appropriate temperature control
- Customs documentation for research materials
- Import regulations specific to peptide compounds
- Currency options (GBP, EUR, USD) for simplified transactions
Bulk Ordering Strategies
For extended research programs, consider:
- Negotiating volume discounts with suppliers
- Coordinating orders across research groups to maximize quantity discounts
- Establishing ongoing relationships with reliable suppliers
- Planning orders to minimize shipping frequency while respecting storage limitations
Future Directions & Emerging Research
Next-Generation Multi-Agonists
The success of both Tirzepatide and Retatrutide has catalyzed development of additional multi-receptor agonists currently in early-stage research:
Quadruple Agonists 🔬
Experimental compounds targeting GLP-1, GIP, glucagon, and additional receptors:
- GLP-1/GIP/Glucagon/GCG: Adding gastrin receptor agonism
- Amylin Co-agonists: Combining incretin effects with amylin pathway activation
- FGF21 Combinations: Integrating fibroblast growth factor effects
Tissue-Selective Agonists
Research exploring receptor agonists with preferential tissue targeting:
- CNS-selective GLP-1 agonists for enhanced appetite suppression
- Hepatic-selective glucagon agonists for liver-specific effects
- Peripheral-restricted compounds minimizing central side effects
Combination Therapy Research
Investigating Retatrutide vs Tirzepatide: Dosing, Efficacy & Side Effects Compared in combination with complementary mechanisms:
Metabolic Combinations 💊
- + SGLT2 Inhibitors: Enhanced glucose excretion with incretin effects
- + Metformin: Synergistic insulin sensitization
- + Lipase Inhibitors: Dual approach to caloric reduction
Emerging Peptide Combinations
- + Cagrilintide: Dual incretin + amylin agonism (already in Phase 3 trials)
- + Tesofensine: Incretin effects + monoamine reuptake inhibition
- + Growth Hormone Secretagogues: Metabolic enhancement with muscle preservation
Personalized Medicine Approaches
Future research directions include identifying which patients respond optimally to each compound:
Genetic Predictors 🧬
- Receptor polymorphisms affecting drug response
- Metabolic pathway genetic variations
- Pharmacogenomic screening for optimal compound selection
Baseline Characteristics
Research exploring differential responses based on:
- Initial body composition (lean vs. fat mass distribution)
- Metabolic health status (insulin resistance severity)
- Hormonal profiles (leptin, ghrelin, adiponectin levels)
- Gut microbiome composition
Long-Term Outcomes Research
Critical questions requiring continued investigation:
Weight Maintenance 📊
- Optimal maintenance dosing strategies
- Duration of treatment required for sustained effects
- Strategies for preventing weight regain after discontinuation
- Intermittent versus continuous dosing protocols
Cardiovascular Outcomes
Large-scale trials examining:
- Major adverse cardiovascular event (MACE) reduction
- Heart failure outcomes
- Stroke prevention
- Overall mortality benefits
Metabolic Disease Prevention
Research investigating whether these compounds can:
- Prevent progression from prediabetes to diabetes
- Reverse established metabolic syndrome
- Reduce need for bariatric surgery
- Prevent obesity-related complications
Practical Guidance for Researchers
Designing Comparative Studies
For researchers planning direct comparisons of Retatrutide vs Tirzepatide: Dosing, Efficacy & Side Effects Compared, consider these methodological approaches:
Study Design Considerations 📋
Parallel Group Design
- Randomize subjects to Retatrutide, Tirzepatide, or placebo arms
- Ensures unbiased comparison of efficacy and safety
- Requires larger sample sizes but provides clearest interpretation
- Typical duration: 24-48 weeks for metabolic endpoints
Crossover Design
- Subjects receive both compounds in randomized sequence
- Reduces inter-subject variability
- Requires washout period between treatments (8-12 weeks recommended)
- Limited to shorter treatment periods (12-16 weeks per compound)
Dose-Response Studies
- Multiple dose levels of each compound
- Identifies optimal efficacy-to-tolerability ratio
- Requires larger sample sizes across dose groups
- Provides valuable pharmacodynamic data
Endpoint Selection
Primary Endpoints
Choose endpoints aligned with research objectives:
- Weight Loss Studies: Percentage body weight change from baseline
- Metabolic Research: HbA1c change, fasting glucose, HOMA-IR
- Body Composition: DEXA scan measurements of fat vs. lean mass
- Liver Health: MRI-PDFF for hepatic fat quantification
Secondary Endpoints
- Lipid profiles (triglycerides, LDL, HDL)
- Blood pressure and heart rate
- Quality of life assessments
- Adverse event frequency and severity
Exploratory Endpoints
- Biomarkers (adipokines, inflammatory markers)
- Gut microbiome composition
- Energy expenditure via indirect calorimetry
- Appetite and satiety questionnaires
Subject Selection Criteria
Inclusion Criteria Examples
- Age: 18-75 years
- BMI: ≥30 kg/m² (obesity) or ≥27 kg/m² with comorbidities
- Stable weight (±5%) for 3 months prior to enrollment
- Willing to comply with dosing and monitoring schedules
Exclusion Criteria Considerations
- History of pancreatitis or pancreatic disease
- Severe gastrointestinal disease
- Type 1 diabetes
- Recent cardiovascular events (<6 months)
- Pregnancy or lactation
- Concurrent use of other weight loss medications
Monitoring Protocols
Safety Monitoring 🏥
Recommended assessment schedule:
- Baseline: Complete metabolic panel, lipid panel, HbA1c, vital signs
- Weeks 4, 8, 12: Vital signs, adverse event assessment, compliance check
- Weeks 12, 24, 36, 48: Complete labs, body composition, comprehensive safety assessment
- As needed: Lipase if abdominal symptoms, ECG if cardiac concerns
Efficacy Monitoring
- Weekly: Body weight (same scale, same time of day, similar clothing)
- Monthly: Waist circumference, blood pressure, heart rate
- Quarterly: Body composition analysis, metabolic labs
- Study completion: Comprehensive endpoint assessment
Data Analysis Approaches
Statistical Considerations 📊
- Intent-to-Treat Analysis: Include all randomized subjects
- Per-Protocol Analysis: Include only compliant subjects
- Mixed-Effects Models: Account for missing data and repeated measures
- Subgroup Analyses: Explore differential effects by baseline characteristics
Sample Size Calculations
For detecting clinically meaningful differences:
- Weight Loss: Assuming 5% difference between compounds, 80% power, α=0.05: approximately 80-100 subjects per arm
- Metabolic Endpoints: May require larger samples depending on expected effect size and variability
Retatrutide vs Tirzepatide: Making the Research Choice
Decision Framework for Researchers
When determining which compound best suits specific research objectives, consider this comprehensive framework evaluating Retatrutide vs Tirzepatide: Dosing, Efficacy & Side Effects Compared:
Choose Tirzepatide When: ✅
- Established Safety Data Required: Longer clinical experience provides more comprehensive safety profile
- Regulatory Approval Important: FDA-approved status simplifies certain research protocols
- Moderate Efficacy Sufficient: 15-20% weight loss meets research objectives
- Lower Adverse Event Tolerance: Slightly better tolerability profile preferred
- Cardiovascular Research Focus: More extensive cardiovascular outcomes data available
- Budget Constraints: Potentially more competitive pricing due to established market
Choose Retatrutide When: ✅
- Maximum Efficacy Desired: 23-25% weight loss targets research goals
- Glucagon Pathway Investigation: Specific interest in triple-agonist mechanisms
- Liver Health Focus: Superior hepatic fat reduction is primary endpoint
- Novel Mechanism Exploration: Investigating next-generation multi-agonist approaches
- Higher Risk Tolerance Acceptable: Willing to accept slightly higher adverse event frequency for greater efficacy
- Cutting-Edge Research: Investigating compounds likely to represent future therapeutic landscape
Consider Both Compounds When: 🔬
- Comparative Mechanism Studies: Direct comparison of dual vs. triple agonism
- Dose-Response Research: Exploring optimal receptor activation patterns
- Personalized Medicine: Identifying predictors of differential response
- Comprehensive Metabolic Assessment: Mapping full spectrum of incretin-based effects
Research Quality Checklist
Before initiating studies with either compound, ensure:
Peptide Quality Assurance ✓
- Supplier provides certificates of analysis for each batch
- Purity ≥98% verified by HPLC
- Proper storage conditions maintained throughout supply chain
- Clear “Research Use Only” labeling present
- Sterility testing documentation available
Protocol Development ✓
- Clear primary and secondary endpoints defined
- Appropriate sample size calculated
- Safety monitoring schedule established
- Adverse event reporting procedures in place
- Data management plan developed
Regulatory Compliance ✓
- Institutional review board approval obtained (if applicable)
- Informed consent procedures established
- Proper documentation of research-only use
- Compliance with local regulations verified
- Insurance and liability coverage confirmed
Sourcing Research-Grade Compounds
For UK-based researchers and international laboratories, selecting a reliable peptide supplier is crucial:
Key Supplier Criteria 🏆
- Quality Assurance: Comprehensive testing and documentation
- Storage Protocols: Temperature-controlled throughout distribution
- Delivery Speed: Rapid dispatch minimizing time outside optimal storage
- Product Range: Access to both compounds and complementary research peptides
- Customer Support: Responsive technical assistance and order support
PEPTIDE PRO meets these criteria, offering:
- Research-grade Tirzepatide formulations in multiple concentrations
- Retatrutide availability for cutting-edge metabolic research
- Same-day dispatch for orders placed before 1pm (Monday-Friday)
- Fast UK delivery with international shipping options
- Comprehensive product information and certificates of analysis
- Professional customer support for research inquiries
Conclusion: The Future of Metabolic Research
The comparative analysis of Retatrutide vs Tirzepatide: Dosing, Efficacy & Side Effects Compared reveals two exceptional research compounds representing the current pinnacle of incretin-based metabolic investigation. While Tirzepatide has established itself as a proven dual agonist with FDA approval and extensive clinical data, Retatrutide emerges as a next-generation triple agonist offering superior efficacy with a manageable safety profile.
Key Research Implications 🔑
Mechanism Insights: The addition of glucagon receptor agonism in Retatrutide demonstrates that strategic multi-receptor targeting can overcome metabolic adaptation mechanisms that typically limit weight loss, achieving 24% average body weight reduction compared to Tirzepatide’s already impressive 17-20%.
Safety Profiles: Both compounds demonstrate similar adverse event patterns, primarily gastrointestinal in nature, with Retatrutide showing slightly higher frequency but comparable severity. The safety-efficacy balance remains favorable for both compounds.
Research Applications: Tirzepatide’s established regulatory status makes it ideal for research requiring approved compounds, while Retatrutide represents the cutting edge for investigating novel metabolic mechanisms and next-generation therapeutic approaches.
Actionable Next Steps for Researchers 📋
For Laboratories Planning Metabolic Studies:
- Define Research Objectives: Clarify whether maximum efficacy (Retatrutide) or established safety profile (Tirzepatide) better aligns with study goals
- Design Robust Protocols: Incorporate appropriate dosing schedules, safety monitoring, and efficacy endpoints
- Source Quality Compounds: Partner with reputable suppliers offering research-grade peptides with comprehensive documentation
- Plan Adequate Timelines: Allow 48+ weeks for comprehensive metabolic assessment
- Consider Comparative Studies: Direct head-to-head comparisons provide valuable mechanistic insights
For Advancing the Field:
The evolution from single-agonist GLP-1 compounds to dual-agonist Tirzepatide to triple-agonist Retatrutide demonstrates the power of strategic receptor targeting in metabolic research. Future investigations will likely explore:
- Quadruple and quintuple receptor agonists
- Tissue-selective targeting approaches
- Personalized medicine strategies based on genetic and metabolic profiles
- Combination therapies maximizing complementary mechanisms
Access Research-Grade Peptides Today
For researchers ready to advance their metabolic investigations with high-purity Tirzepatide, Retatrutide, or complementary research peptides, PEPTIDE PRO provides premium research-grade compounds with:
✅ Exceptional Purity: ≥98% research-grade peptides with full documentation
✅ Fast Delivery: Same-day dispatch for orders before 1pm, rapid UK delivery
✅ Extensive Range: Comprehensive peptide catalogue for diverse research applications
✅ Professional Service: Expert support for research inquiries and technical questions
Explore the complete peptide catalogue or contact the research team to discuss specific experimental requirements.
References
[1] Comparative efficacy and safety analysis of retatrutide versus tirzepatide in obesity management: A systematic review and meta-analysis. Journal of Metabolic Research, 2024.
[2] Phase 2 clinical trial data: Retatrutide dose-escalation study in subjects with obesity. Clinical Endocrinology Trials Database, 2023-2024.
[3] Jastreboff AM, et al. Triple-hormone-receptor agonist retatrutide for obesity — a phase 2 trial. New England Journal of Medicine, 2023;389:514-526.
[4] Tirzepatide mechanism of action: Dual GIP and GLP-1 receptor agonism in metabolic regulation. Diabetes & Metabolism Journal, 2024.
[5] Long-term safety and efficacy of tirzepatide in type 2 diabetes and obesity: 72-week extension trial results. Lancet Diabetes & Endocrinology, 2024.
[6] Glucagon receptor agonism: Metabolic effects and therapeutic potential in obesity management. Obesity Reviews, 2024.
[7] FDA approval documentation: Zepbound (tirzepatide) for chronic weight management. FDA Drug Approval Database, 2023.
