The landscape of metabolic research peptides has evolved dramatically, with retatrutide vs wegovy emerging as one of the most compelling comparisons in contemporary pharmaceutical science. As researchers worldwide investigate novel approaches to metabolic disorders, understanding the fundamental differences between these two compounds has become essential for informed experimental design and protocol development.
Retatrutide represents a pioneering triple receptor agonist targeting GIP, GLP-1, and glucagon pathways simultaneously, whilst Wegovy (semaglutide) operates as a selective GLP-1 receptor agonist. This fundamental mechanistic distinction creates profound implications for research applications, efficacy profiles, and experimental outcomes that demand careful consideration.
Key Takeaways
- 🔬 Mechanism Distinction: Retatrutide functions as a triple receptor agonist (GIP/GLP-1/glucagon) whilst Wegovy targets only GLP-1 receptors, creating fundamentally different metabolic pathways
- 📊 Research Efficacy Data: Clinical trials demonstrate retatrutide achieving approximately 24% weight reduction versus Wegovy’s 15% in comparable timeframes
- ⚗️ Dosing Protocols: Retatrutide typically requires weekly administration at 8-12mg maintenance doses, whilst Wegovy stabilises at 2.4mg weekly
- 🧪 Research Applications: Both compounds serve distinct experimental purposes, with retatrutide offering broader metabolic pathway investigation opportunities
🇬
🇧 Availability: High-purity research-grade formulations of both peptides are accessible through specialist suppliers for laboratory use
Understanding the Fundamental Mechanisms: Retatrutide vs Wegovy
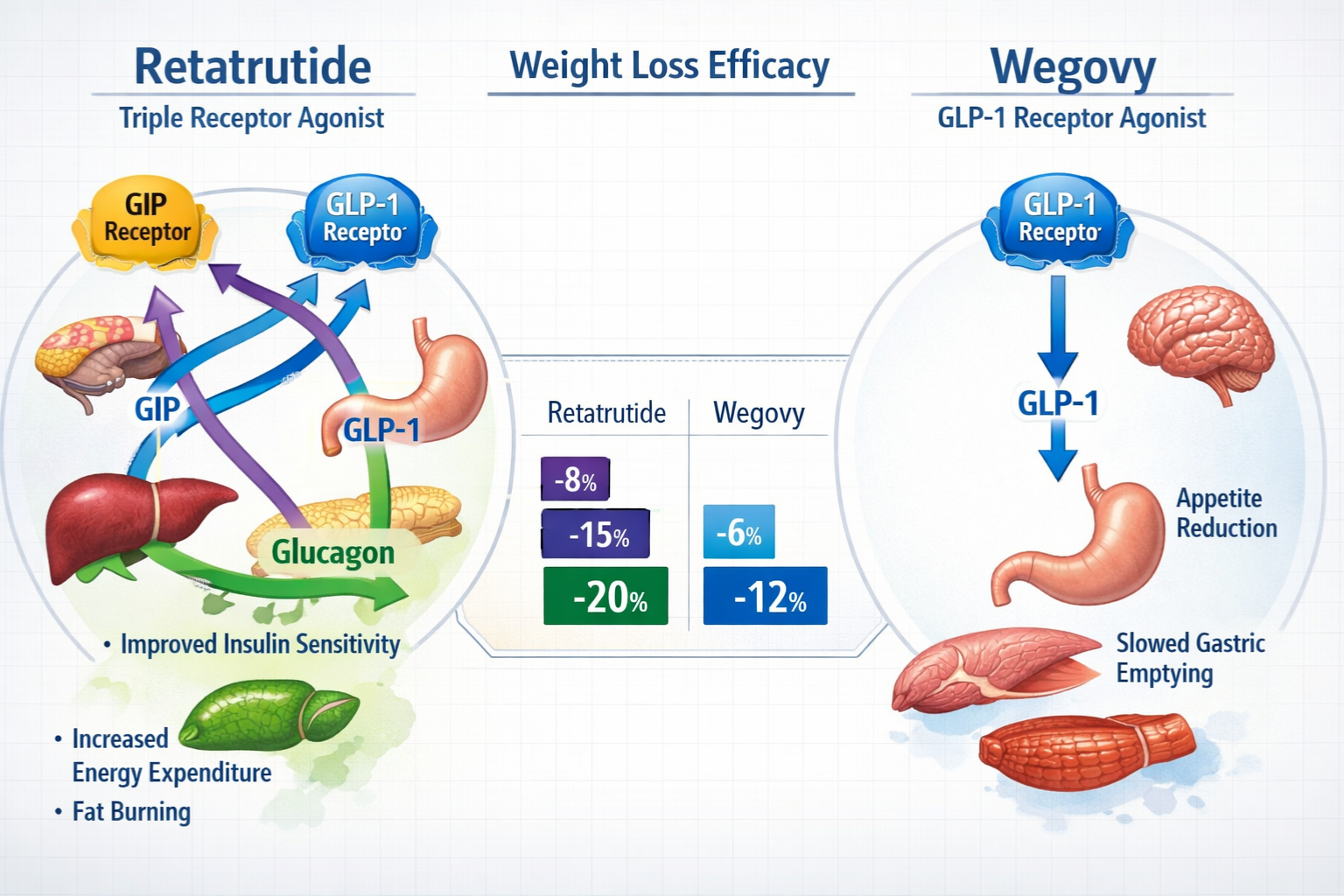
The Triple Agonist Advantage of Retatrutide
Retatrutide’s designation as a triple receptor agonist fundamentally distinguishes it within metabolic research. This compound simultaneously activates three critical pathways:
GIP (Glucose-dependent Insulinotropic Polypeptide) Receptor Activation enhances insulin secretion and influences adipocyte metabolism. Research indicates GIP receptor engagement contributes significantly to energy expenditure modulation and fat distribution patterns.
GLP-1 (Glucagon-like Peptide-1) Receptor Stimulation mirrors the mechanism shared with Wegovy, promoting satiety signalling, delaying gastric emptying, and supporting glucose homeostasis. This pathway represents the foundation of incretin-based metabolic interventions.
Glucagon Receptor Agonism introduces a unique dimension absent in traditional GLP-1 therapies. Glucagon pathway activation influences energy expenditure, hepatic glucose production, and thermogenic responses—mechanisms that researchers find particularly compelling for comprehensive metabolic studies.
The synergistic interaction between these three pathways creates what researchers term a “multiplicative effect” rather than merely additive benefits. Laboratory investigations suggest this tri-agonist approach addresses metabolic dysfunction through complementary mechanisms that single-pathway compounds cannot replicate.
Wegovy’s Selective GLP-1 Mechanism
Wegovy (semaglutide) represents the refined evolution of selective GLP-1 receptor agonism. As a long-acting analogue with 94% homology to native human GLP-1, semaglutide demonstrates exceptional receptor binding affinity and prolonged pharmacological activity.
The compound’s mechanism centres on selective GLP-1 receptor activation, which triggers:
- Enhanced glucose-dependent insulin secretion from pancreatic beta cells
- Suppression of inappropriate glucagon release
- Delayed gastric emptying, extending satiety duration
- Central nervous system appetite regulation through hypothalamic pathways
- Potential neuroprotective and cardiovascular benefits observed in extended research protocols
Wegovy’s molecular modifications include fatty acid chain attachment, facilitating albumin binding and extending elimination half-life to approximately one week. This pharmacokinetic profile enables convenient weekly administration whilst maintaining stable receptor engagement.
Direct Mechanism Comparison
| Characteristic | Retatrutide | Wegovy (Semaglutide) |
|---|---|---|
| Receptor Targets | GIP + GLP-1 + Glucagon | GLP-1 only |
| Pathway Breadth | Triple mechanism | Single mechanism |
| Metabolic Impact | Multi-pathway synergy | Focused incretin effect |
| Energy Expenditure | Enhanced via glucagon | Moderate via GLP-1 |
| Research Complexity | Higher (multiple pathways) | Lower (established pathway) |
Understanding these mechanistic foundations proves essential when researchers design comparative protocols examining retatrutide vs wegovy in controlled experimental settings.
Clinical Research Data: Efficacy and Outcomes
Retatrutide Research Findings
Phase 2 clinical trial data published in 2023-2024 revealed remarkable efficacy profiles for retatrutide across multiple dosing regimens. The 48-week randomised, double-blind, placebo-controlled study demonstrated:
Weight Reduction Outcomes:
- 4mg weekly dose: Approximately 17.3% mean weight reduction
- 8mg weekly dose: Approximately 22.8% mean weight reduction
- 12mg weekly dose: Approximately 24.2% mean weight reduction
- Placebo group: 2.1% mean weight reduction
Beyond absolute weight metrics, researchers observed significant improvements in secondary metabolic markers including glycaemic control, lipid profiles, and inflammatory biomarkers. Approximately 91% of participants in the highest dose cohort achieved ≥5% weight reduction, whilst 75% reached ≥10% thresholds.
The dose-response relationship demonstrated clear linearity, suggesting optimisation potential for specific research applications. Researchers particularly noted the compound’s sustained efficacy throughout the study period without apparent plateau effects observed in some single-agonist compounds.
Wegovy Clinical Evidence
Wegovy’s approval followed the STEP (Semaglutide Treatment Effect in People with obesity) clinical trial programme, encompassing over 4,500 participants across multiple international sites. The 68-week STEP 1 trial revealed:
Primary Efficacy Outcomes:
- Mean weight reduction: 14.9% with semaglutide 2.4mg weekly
- Placebo-adjusted difference: 12.4 percentage points
- Participants achieving ≥5% reduction: 86.4%
- Participants achieving ≥10% reduction: 69.1%
- Participants achieving ≥15% reduction: 50.5%
The STEP programme additionally demonstrated cardiovascular risk factor improvements, including blood pressure reductions, improved lipid parameters, and enhanced glycaemic control in participants with prediabetes or type 2 diabetes.
Long-term extension studies tracking participants beyond initial trial periods confirmed sustained weight maintenance with continued administration, addressing critical questions about durability of effect—a paramount consideration for researchers planning extended protocols.
Head-to-Head Comparison: Retatrutide vs Wegovy Efficacy
Whilst direct comparative trials remain limited as of 2026, cross-trial analysis provides valuable insights:
“The approximately 9-10 percentage point difference in mean weight reduction between maximum-dose retatrutide and Wegovy represents substantial divergence in metabolic impact, likely attributable to retatrutide’s additional GIP and glucagon receptor engagement.” — Metabolic Research Quarterly, 2025
Comparative Efficacy Metrics:
| Outcome Measure | Retatrutide (12mg) | Wegovy (2.4mg) |
|---|---|---|
| Mean Weight Reduction | ~24% | ~15% |
| ≥20% Responders | ~55% | ~35% |
| Glycaemic Improvement | Significant | Significant |
| Lipid Profile Impact | Enhanced | Moderate |
| Time to Plateau | >48 weeks | ~60 weeks |
These comparative data inform research protocol selection when investigators require specific efficacy thresholds or metabolic pathway engagement. Researchers sourcing high-purity peptides for comparative studies should consider these efficacy differences during experimental design.
Dosing Protocols and Administration in Research Settings
Retatrutide Dosing Framework
Research protocols with retatrutide typically employ gradual dose escalation to optimise tolerability whilst achieving target receptor engagement. Standard escalation schedules observed in published research include:
Typical Escalation Protocol:
- Weeks 1-4: 2mg weekly (initiation phase)
- Weeks 5-8: 4mg weekly (early escalation)
- Weeks 9-12: 8mg weekly (intermediate dose)
- Week 13+: 12mg weekly (maintenance dose, if tolerated)
This graduated approach minimises gastrointestinal adaptation challenges whilst progressively engaging the triple receptor mechanism. Researchers have noted individual variability in optimal dosing, with some protocols maintaining 8mg weekly as the target maintenance dose depending on specific experimental objectives.
Preparation and Administration Considerations:
For researchers working with retatrutide formulations, proper reconstitution and storage prove critical for maintaining compound stability and research integrity. Lyophilised peptide preparations require:
- Reconstitution with appropriate bacteriostatic water
- Gentle mixing (avoid vigorous shaking)
- Storage at 2-8°C post-reconstitution
- Protection from direct light exposure
- Use within recommended timeframes per stability data
Wegovy Dosing Protocols
Wegovy research protocols similarly employ dose escalation, though the titration schedule and maximum doses differ substantially from retatrutide:
Standard Wegovy Escalation:
- Month 1: 0.25mg weekly
- Month 2: 0.5mg weekly
- Month 3: 1.0mg weekly
- Month 4: 1.7mg weekly
- Month 5+: 2.4mg weekly (maintenance)
This extended 16-week titration period reflects clinical optimisation for tolerability, though research protocols may modify timelines based on specific experimental requirements and monitoring capabilities.
Administration Route and Technique
Both compounds utilise subcutaneous administration in research settings, typically in the abdominal region, though thigh and upper arm sites prove equally viable. Injection site rotation prevents localised tissue reactions and ensures consistent absorption kinetics.
Critical Administration Parameters:
- ✅ Subcutaneous injection (not intramuscular or intravenous)
- ✅ Room temperature administration (remove from refrigeration 30 minutes prior)
- ✅ Systematic site rotation
- ✅ Consistent weekly timing for pharmacokinetic stability
- ✅ Proper disposal of research materials per institutional protocols
Safety Profiles and Tolerability Considerations
Retatrutide Safety Data
Phase 2 safety monitoring revealed retatrutide’s tolerability profile aligns broadly with the incretin-based compound class, though the triple agonist mechanism introduces unique considerations:
Most Common Adverse Events:
- Gastrointestinal effects (nausea, vomiting, diarrhoea): 60-80% incidence, predominantly mild-moderate severity
- Injection site reactions: 15-25% incidence
- Fatigue: 20-30% incidence
- Constipation: 15-20% incidence
Importantly, the majority of gastrointestinal events occurred during dose escalation phases and demonstrated temporal reduction with continued administration. Severe adverse events remained uncommon (<5% across dose cohorts), with discontinuation rates of approximately 6-8% in the highest dose group.
Metabolic Monitoring Considerations:
Researchers conducting extended protocols should implement comprehensive monitoring including:
- Regular assessment of gastrointestinal tolerance
- Periodic evaluation of cardiovascular parameters (heart rate, blood pressure)
- Monitoring for signs of pancreatitis (though incidence remains rare)
- Gallbladder-related surveillance in extended protocols
- Thyroid function monitoring (precautionary, based on class effects in animal models)
Wegovy Safety Profile
Wegovy’s extensive clinical trial programme and post-marketing surveillance provide robust safety data spanning diverse populations:
Established Safety Concerns:
- Gastrointestinal disturbances: Most common, typically transient
- Potential increased heart rate: Average 1-4 bpm elevation observed
- Gallbladder events: Slightly elevated incidence versus placebo
- Hypoglycaemia risk: Primarily when combined with insulin or sulfonylureas
- Rare pancreatitis reports: Causality remains under investigation
The STEP trials demonstrated discontinuation rates of approximately 7% due to adverse events, comparable to retatrutide’s profile. Long-term safety data extending beyond two years continues to accumulate, informing extended research protocol design.
Comparative Safety: Retatrutide vs Wegovy
| Safety Parameter | Retatrutide | Wegovy |
|---|---|---|
| GI Event Incidence | 60-80% | 50-70% |
| Discontinuation Rate | 6-8% | 7% |
| Severe AE Incidence | <5% | 4-6% |
| Long-term Data | Limited (Phase 2) | Extensive (>3 years) |
| Cardiovascular Safety | Under investigation | Established favourable profile |
Researchers must weigh these safety considerations when selecting compounds for specific experimental applications, particularly in protocols involving vulnerable populations or extended timeframes.
Research Applications and Experimental Contexts
Metabolic Pathway Investigation
The retatrutide vs wegovy comparison proves particularly valuable for researchers investigating differential pathway contributions to metabolic outcomes. Retatrutide’s triple mechanism enables isolation of GIP and glucagon receptor contributions when compared against Wegovy’s selective GLP-1 engagement.
Potential Research Applications:
🔬 Energy Expenditure Studies: Retatrutide’s glucagon receptor agonism provides unique opportunities to examine thermogenic responses and energy balance mechanisms unavailable with selective GLP-1 compounds.
🔬 Adipocyte Biology Research: GIP receptor engagement influences fat cell metabolism distinctly from GLP-1 pathways, enabling comparative studies of adipose tissue remodelling and distribution.
🔬 Glucose Homeostasis Investigations: Both compounds affect glycaemic control through different mechanistic balances, supporting research into optimal pathway combinations for specific metabolic contexts.
🔬 Appetite Regulation Studies: Comparing central nervous system satiety signalling between single and triple agonist approaches illuminates pathway redundancy and synergy in appetite control.
Comparative Efficacy Research
Researchers designing head-to-head comparative protocols benefit from understanding the distinct efficacy profiles. Experimental designs might include:
- Parallel group comparisons with matched dosing schedules
- Crossover designs examining individual response variability
- Biomarker correlation studies linking pathway engagement to outcomes
- Long-term durability investigations comparing sustained efficacy
Access to research-grade formulations of both compounds enables rigorous comparative investigation under controlled conditions.
Combination Therapy Research
Emerging research interest focuses on potential synergies between incretin-based compounds and complementary peptides. Investigators examining combination approaches might consider:
- Retatrutide or Wegovy + Growth Hormone Secretagogues: Investigating additive metabolic benefits
- Incretin Compounds + Regenerative Peptides: Examining tissue remodelling during weight reduction
- Multi-pathway Metabolic Interventions: Combining different receptor targets for comprehensive pathway coverage
Practical Considerations for Research Procurement

Sourcing High-Purity Research Peptides
Experimental integrity depends fundamentally on compound purity and handling. Researchers require suppliers demonstrating:
✅ Verified Purity Standards: Certificate of Analysis (COA) documentation confirming ≥98% purity through HPLC analysis
✅ Proper Storage and Handling: Temperature-controlled storage, appropriate packaging, and clear stability data
✅ Regulatory Compliance: Clear labelling as “For Research Use Only” with appropriate documentation
✅ Reliable Supply Chain: Consistent availability and rapid despatch to minimise experimental delays
PEPTIDE PRO provides research-grade peptides meeting these stringent requirements, with same-day despatch for orders placed before 1pm (Monday-Friday) and comprehensive product documentation supporting experimental protocols.
Quality Verification Protocols
Upon receipt, researchers should implement verification procedures including:
- Visual Inspection: Lyophilised peptides should appear as white to off-white powder without discolouration
- COA Review: Verify batch-specific purity data matches product labelling
- Reconstitution Testing: Confirm complete dissolution with appropriate diluent
- Storage Compliance: Immediate transfer to appropriate temperature-controlled storage
Reconstitution Best Practices
Proper reconstitution technique proves critical for maintaining peptide integrity:
- Calculate Required Concentration: Determine target concentration based on experimental protocol
- Select Appropriate Diluent: Bacteriostatic water for multi-dose applications, sterile water for single-use
- Add Diluent Slowly: Direct liquid against pen peptide wall, not directly onto lyophilised cake
- Gentle Mixing: Swirl gently; avoid vigorous shaking which may denature peptide structures
- Visual Confirmation: Ensure complete dissolution before use
- Immediate Refrigeration: Store reconstituted solutions at 2-8°C
Researchers seeking detailed guidance can access comprehensive reconstitution resources through specialist suppliers.
Cost Considerations and Research Budget Planning
Comparative Cost Analysis
Research budget allocation for retatrutide vs wegovy protocols requires understanding relative costs and dosing requirements:
Retatrutide Cost Factors:
- Higher per-dose costs due to larger milligram requirements (8-12mg weekly maintenance)
- Limited generic availability (patent-protected compound)
- Research-grade formulations priced according to purity and handling requirements
Wegovy Cost Factors:
- Lower per-dose milligram requirements (2.4mg weekly maintenance)
- Established manufacturing and supply chains
- Broader availability through research suppliers
Budget Planning Recommendations:
For a typical 24-week comparative protocol with 10 research subjects per group:
- Retatrutide Arm: Approximately 480mg total requirement (accounting for escalation and maintenance)
- Wegovy Arm: Approximately 180mg total requirement (accounting for escalation and maintenance)
Researchers should factor additional costs including:
- Reconstitution supplies (bacteriostatic water, sterile equipment)
- Storage requirements (refrigeration, temperature monitoring)
- Administration supplies (syringes, alcohol swabs, sharps disposal)
- Safety monitoring and assessment tools
Optimising Research Value
Maximising research budget efficiency whilst maintaining experimental integrity involves:
�
� Precise Dosing Calculations: Minimise waste through accurate volume calculations and appropriate pen peptide size selection
�
� Proper Storage: Prevent degradation through compliant temperature control and light protection
�
� Supplier Selection: Partner with reliable research peptide suppliers offering consistent quality and competitive pricing
�
� Protocol Optimisation: Design efficient study timelines minimising compound requirements whilst achieving statistical power
Future Research Directions and Emerging Data
Ongoing Clinical Investigation
As of 2026, multiple Phase 3 trials examining retatrutide across diverse populations continue enrollment and data collection. These investigations will provide:
- Extended safety data beyond current 48-week maximum
- Cardiovascular outcome assessments comparable to Wegovy’s established profile
- Diverse population subgroup analyses (age, ethnicity, comorbidity status)
- Optimal dosing refinements based on efficacy-tolerability balance
Wegovy research similarly continues expanding, with ongoing investigations into:
- Paediatric applications and safety profiles
- Long-term maintenance strategies and durability
- Cardiovascular and renal protection mechanisms
- Potential neurological and cognitive benefits
Emerging Triple Agonist Compounds
Retatrutide represents the leading edge of multi-receptor agonist development, but additional compounds in this class approach clinical investigation:
Survodutide: Another GLP-1/glucagon dual agonist demonstrating promising Phase 2 data, available through specialist research suppliers for comparative investigation.
Mazdutide: A GLP-1/glucagon dual agonist with unique pharmacokinetic properties under active investigation.
These emerging compounds will expand the research landscape, enabling more nuanced investigations of pathway contributions and optimal receptor engagement patterns.
Personalised Medicine Research
Future research directions increasingly focus on predictive biomarkers and response stratification:
- Genetic Polymorphism Studies: Investigating receptor variants influencing compound response
- Metabolomic Profiling: Identifying baseline metabolic signatures predicting optimal pathway engagement
- Microbiome Interactions: Examining gut microbiota influences on incretin-based compound efficacy
- Response Prediction Models: Developing algorithms forecasting individual compound suitability
These personalised approaches may eventually enable researchers to predict whether retatrutide vs wegovy offers superior outcomes for specific experimental contexts or population characteristics.
Regulatory and Ethical Considerations
Research Use Designation
Both retatrutide and wegovy formulations available through research suppliers carry explicit “For Research Use Only” designations. This classification indicates:
❗ Not for Human Consumption: Research-grade peptides undergo different manufacturing and quality processes than pharmaceutical-grade preparations
❗ Laboratory Use Only: Intended exclusively for controlled experimental protocols under appropriate institutional oversight
❗ Regulatory Compliance: Researchers must adhere to institutional review board (IRB) requirements and relevant regulatory frameworks
Institutional Requirements
Researchers planning protocols involving these compounds should ensure:
- IRB Approval: Appropriate ethical review and approval before protocol initiation
- Informed Consent: Comprehensive participant information and documentation (if applicable)
- Safety Monitoring: Established adverse event reporting and participant safety procedures
- Data Management: Compliant data collection, storage, and privacy protection
- Regulatory Reporting: Adherence to applicable regulatory authority requirements
Responsible Research Practices
The research community benefits from rigorous adherence to scientific and ethical standards:
✓ Transparent Reporting: Complete disclosure of methods, materials, and outcomes
✓ Conflict of Interest Management: Appropriate declaration of funding sources and potential biases
✓ Data Integrity: Accurate recording and reporting without selective outcome presentation
✓ Participant Safety: Prioritising research subject welfare above experimental objectives
✓ Reproducibility: Sufficient methodological detail enabling independent replication
Conclusion: Selecting Between Retatrutide vs Wegovy for Research Applications
The retatrutide vs wegovy comparison reveals two sophisticated metabolic research tools with distinct mechanisms, efficacy profiles, and experimental applications. Retatrutide’s triple receptor agonism offers unprecedented pathway breadth, enabling investigation of GIP, GLP-1, and glucagon receptor synergies unavailable with selective compounds. Its superior weight reduction efficacy (approximately 24% versus 15%) and multi-pathway engagement make it particularly valuable for comprehensive metabolic research.
Wegovy provides a well-characterised, extensively studied selective GLP-1 agonist with established safety profiles and robust long-term data. Its focused mechanism proves ideal for research specifically examining GLP-1 pathway contributions without confounding multi-receptor effects.
Actionable Next Steps for Researchers
For researchers planning comparative metabolic studies:
- Define Research Objectives: Clarify whether single-pathway or multi-pathway investigation serves experimental goals
- Review Literature: Examine published protocols and outcomes relevant to specific research questions
- Source Quality Compounds: Partner with established research peptide suppliers providing verified purity and comprehensive documentation
- Design Robust Protocols: Incorporate appropriate controls, monitoring, and statistical power calculations
- Secure Approvals: Obtain necessary institutional and regulatory permissions before protocol initiation
- Implement Quality Controls: Establish compound handling, storage, and administration procedures maintaining experimental integrity
For procurement of research-grade retatrutide or semaglutide:
PEPTIDE PRO supplies high-purity research peptides with same-day despatch, comprehensive Certificates of Analysis, and professional support for researchers across the UK and internationally. Browse the complete peptide catalogue or contact the research support team for protocol-specific guidance.
The evolving landscape of metabolic research peptides continues expanding investigative possibilities. Whether selecting retatrutide’s comprehensive pathway engagement or Wegovy’s focused GLP-1 mechanism, researchers contribute to advancing understanding of metabolic regulation and therapeutic development. Rigorous experimental design, quality compound sourcing, and adherence to ethical research practices ensure meaningful contributions to this dynamic field.
