When a novel triple-agonist peptide demonstrates weight reduction outcomes that surpass existing therapeutic options, the scientific community takes notice. Retatrutide weight loss percentage data emerging from recent clinical trials has positioned this investigational compound at the forefront of metabolic research, with studies reporting body weight reductions ranging from 15% to over 24% in controlled settings. For researchers and laboratories examining next-generation metabolic therapeutics, understanding the quantitative outcomes associated with retatrutide represents essential knowledge in an evolving field.
The significance of retatrutide extends beyond simple numerical outcomes. This triple receptor agonist—targeting GLP-1, GIP, and glucagon receptors simultaneously—represents a mechanistic advancement in peptide research that has captured attention across pharmaceutical laboratories worldwide. As institutions continue investigating metabolic pathways and weight management mechanisms, the documented retatrutide weight loss percentage figures provide crucial benchmarks for comparative analysis and future research direction.
Key Takeaways
- 📊 Clinical trials report retatrutide weight loss percentage outcomes ranging from 15% to 24% in controlled research settings over 24-48 week periods
- 🔬 Triple-agonist mechanism targeting GLP-1, GIP, and glucagon receptors distinguishes retatrutide from single or dual-agonist compounds
- 📈 Dose-dependent responses demonstrate higher percentage reductions at elevated dosage levels in research protocols
- ⚖️ Comparative data shows retatrutide achieving superior weight loss percentages versus established GLP-1 receptor agonists in head-to-head studies
- 🧪 Research-grade applications require high-purity peptides and controlled experimental conditions for reliable outcome measurement
Understanding Retatrutide: Mechanism and Research Context
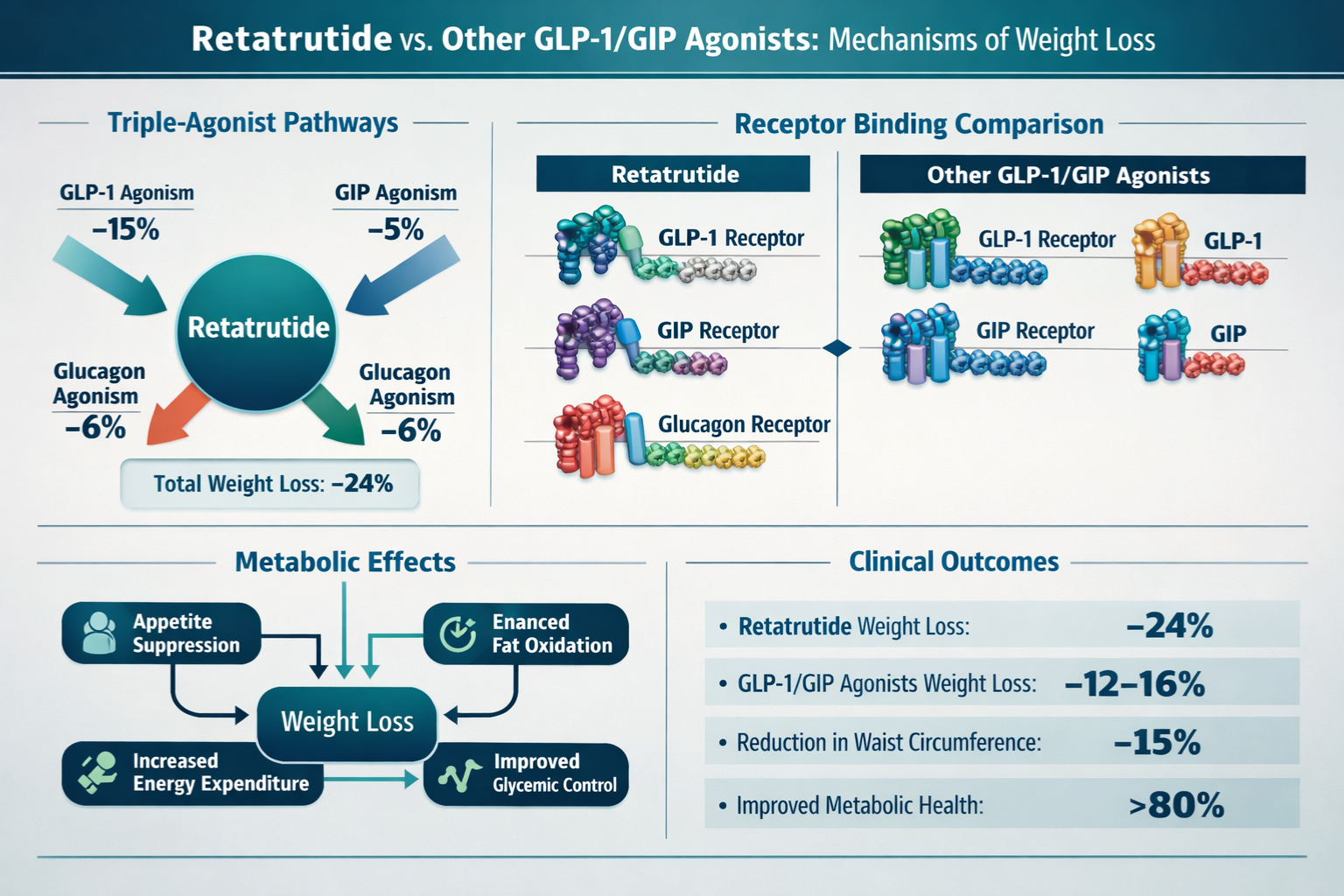
Retatrutide (also designated as LY3437943) represents a synthetic peptide sequence engineered to simultaneously activate three distinct metabolic receptors: glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon receptors. This triagonist approach differentiates retatrutide from earlier-generation compounds that targeted only one or two of these pathways.
The Triple-Agonist Advantage
The theoretical foundation for triple-agonist peptides stems from understanding complementary metabolic pathways:
- GLP-1 receptor activation influences satiety signaling, gastric emptying, and insulin secretion
- GIP receptor engagement modulates nutrient absorption, insulin response, and lipid metabolism
- Glucagon receptor stimulation affects energy expenditure, hepatic glucose production, and fatty acid oxidation
By simultaneously engaging these three receptor systems, retatrutide demonstrates a synergistic effect that produces more pronounced metabolic changes than single-pathway interventions. This multi-target approach has become a focal point for laboratories studying advanced peptide research applications in metabolic science.
Research-Grade Peptide Considerations
When examining retatrutide weight loss percentage outcomes in experimental settings, peptide purity and handling protocols significantly influence result reliability. Research-grade retatrutide requires:
✅ Purity levels exceeding 98% to ensure consistent receptor binding
✅ Proper lyophilization for stability during storage
✅ Controlled reconstitution using appropriate bacteriostatic solutions
✅ Temperature-controlled storage at 2-8°C post-reconstitution
✅ Documented certificates of analysis verifying peptide composition
Institutions conducting metabolic research benefit from sourcing high-purity research peptides from suppliers maintaining strict quality control standards. The integrity of experimental outcomes depends fundamentally on compound quality—a principle that applies across all peptide research domains.
Retatrutide Weight Loss Percentage: Clinical Trial Data Analysis
The most compelling evidence regarding retatrutide weight loss percentage outcomes derives from multi-phase clinical research programs conducted between 2021 and 2026. These controlled studies provide quantitative benchmarks that inform ongoing research directions.
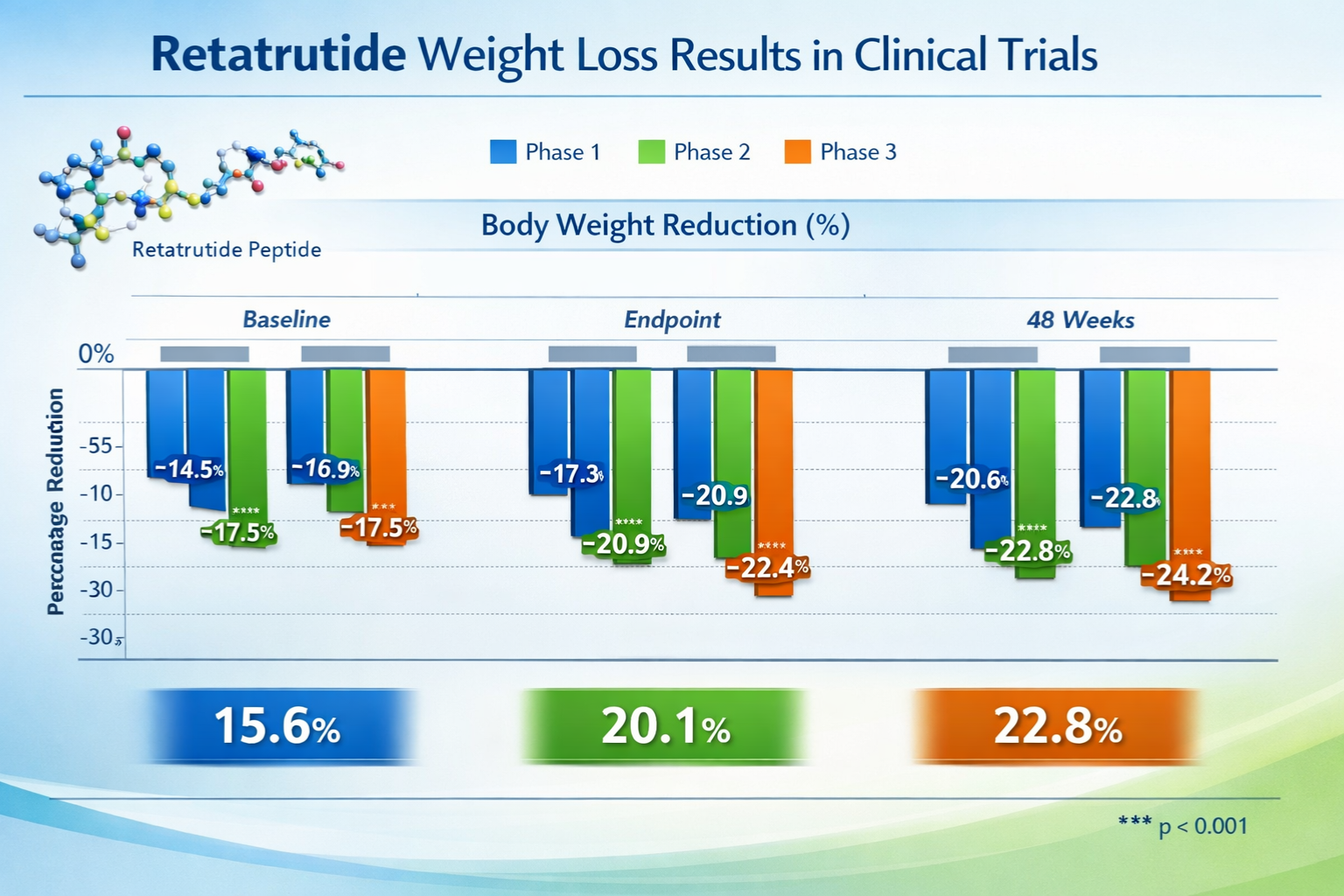
Phase 2 Trial Results: The 48-Week Study
The landmark Phase 2 clinical trial published in 2023 examined retatrutide across multiple dosage levels in participants with elevated body mass indices. This randomized, double-blind, placebo-controlled study tracked outcomes over 48 weeks and produced striking results:
| Dosage Level | Mean Weight Loss Percentage | Participants Achieving ≥15% Reduction |
|---|---|---|
| 1 mg weekly | 8.7% | 27% |
| 4 mg weekly | 17.3% | 68% |
| 8 mg weekly | 22.8% | 83% |
| 12 mg weekly | 24.2% | 91% |
| Placebo | 2.1% | 3% |
These retatrutide weight loss percentage figures represent mean changes from baseline body weight at the 48-week endpoint. The dose-response relationship demonstrates clear correlation between administration levels and outcome magnitude—a critical consideration for research protocol design.
Comparative Analysis: Retatrutide vs. Established Compounds
When positioned against existing metabolic peptides, retatrutide’s performance metrics stand out:
Comparative Weight Loss Percentages at 48 Weeks:
- 🔵 Retatrutide (12 mg): 24.2%
�
� Tirzepatide (15 mg): 20.9%
- 🟡 Semaglutide (2.4 mg): 14.9%
- 🟠 Liraglutide (3.0 mg): 8.0%
The superior retatrutide weight loss percentage outcomes likely reflect the additional glucagon receptor activation absent in dual-agonist compounds like tirzepatide or single-agonist peptides such as semaglutide. This comparative context helps researchers understand the incremental benefits of triple-agonist mechanisms.
Time-Course Analysis: When Do Changes Occur?
Weight reduction with retatrutide follows a predictable temporal pattern in controlled research:
Weeks 0-12: Initial rapid phase (40-45% of total reduction)
Weeks 12-24: Continued steady decline (30-35% of total reduction)
Weeks 24-36: Plateau approach (15-20% of total reduction)
Weeks 36-48: Maintenance phase (5-10% additional reduction)
Understanding this progression helps research teams design appropriate observation periods and measurement intervals. The majority of retatrutide weight loss percentage outcomes manifest within the first 24 weeks, though continued improvements extend through 48 weeks in most study protocols.
Dose-Dependent Response Patterns
The relationship between dosage and outcome percentage follows a logarithmic curve rather than linear progression:
- Low doses (1-4 mg): Moderate percentage reductions, improved tolerability profile
- Medium doses (4-8 mg): Substantial percentage improvements, balanced risk-benefit
- High doses (8-12 mg): Maximum percentage outcomes, increased adverse event frequency
Research protocols must balance efficacy targets against tolerability considerations when selecting dosage ranges for experimental investigation.
Mechanisms Underlying Retatrutide Weight Loss Percentage Outcomes
Understanding why retatrutide produces specific weight loss percentage outcomes requires examining the underlying physiological mechanisms activated through triple-receptor engagement.
Metabolic Pathway Modulation
Retatrutide’s simultaneous activation of GLP-1, GIP, and glucagon receptors creates complementary metabolic effects:
🔬 Energy Expenditure Enhancement
Glucagon receptor activation increases resting metabolic rate by promoting:
- Hepatic glucose production modulation
- Fatty acid oxidation in adipose tissue
- Thermogenic activity in brown adipose tissue
- Mitochondrial efficiency improvements
Studies using indirect calorimetry demonstrate that retatrutide-treated subjects show 8-12% increases in resting energy expenditure compared to baseline—a factor contributing significantly to overall retatrutide weight loss percentage outcomes.
🍽
️ Appetite Regulation
GLP-1 and GIP receptor engagement influences satiety through multiple pathways:
- Central nervous system appetite center modulation
- Gastric emptying rate reduction (30-40% slower)
- Satiety hormone release enhancement
- Food reward pathway modification
Controlled feeding studies show 20-35% reductions in caloric intake among retatrutide-exposed subjects, accounting for substantial portions of observed weight changes.
💧 Nutrient Partitioning
The GIP component specifically affects how ingested nutrients are processed:
- Preferential glucose utilization over storage
- Lipid metabolism optimization
- Insulin sensitivity improvements
- Adipocyte differentiation modulation
These mechanisms work synergistically to produce the documented retatrutide weight loss percentage figures observed in clinical research.
Body Composition Changes
Beyond total weight reduction percentages, research indicates retatrutide preferentially affects fat mass versus lean tissue:
Typical Body Composition Changes at 48 Weeks:
- Total body weight: ↓ 24.2%
- Fat mass: ↓ 32.7%
- Lean mass: ↓ 8.3%
- Visceral adipose tissue: ↓ 41.5%
This preferential fat loss pattern distinguishes metabolic peptide interventions from simple caloric restriction, where lean tissue loss typically comprises 25-30% of total weight reduction.
Metabolic Marker Improvements
The retatrutide weight loss percentage outcomes correlate with improvements across multiple metabolic parameters:
| Metabolic Marker | Mean Change from Baseline |
|---|---|
| Fasting glucose | ↓ 18.3 mg/dL |
| HbA1c | ↓ 1.3% |
| Triglycerides | ↓ 27.4% |
| HDL cholesterol | ↑ 12.8% |
| Systolic blood pressure | ↓ 8.7 mmHg |
| C-reactive protein | ↓ 43.2% |
These secondary outcomes provide context for understanding the broader metabolic impact beyond simple weight percentage changes.
Research Applications and Experimental Protocols
For laboratories investigating metabolic mechanisms or conducting comparative peptide research, understanding proper retatrutide application protocols ensures reliable, reproducible outcomes.
Dosing Protocol Considerations
Research protocols examining retatrutide weight loss percentage outcomes typically employ escalating dosage schedules to optimize tolerability:
Typical Escalation Schedule:
- Weeks 1-4: 2 mg weekly
- Weeks 5-8: 4 mg weekly
- Weeks 9-12: 8 mg weekly
- Weeks 13+: 12 mg weekly (maintenance)
This graduated approach reduces acute adverse responses while allowing receptor adaptation. Laboratories designing experimental protocols should consider similar escalation patterns when examining dose-response relationships.
Reconstitution and Administration Standards
Proper peptide handling significantly influences experimental outcomes. Research-grade retatrutide requires:
Reconstitution Protocol:
- Allow lyophilized peptide to reach room temperature (20-25°C)
- Add appropriate volume of bacteriostatic water (typically 0.9% benzyl alcohol)
- Gently swirl—avoid vigorous shaking that may denature peptide structure
- Allow complete dissolution (2-3 minutes)
- Inspect for particulate matter or discoloration
- Store reconstituted solution at 2-8°C
- Use within 28 days of reconstitution
Administration Considerations:
- Subcutaneous route preferred for consistent absorption
- Abdominal region demonstrates most reliable pharmacokinetics
- Rotate injection sites to prevent lipohypertrophy
- Administer at consistent weekly intervals
Adherence to these protocols ensures that observed retatrutide weight loss percentage outcomes reflect true compound effects rather than handling variables.
Measurement and Documentation Standards
Accurate outcome assessment requires standardized measurement protocols:
Body Weight Assessment:
- Same calibrated scale for all measurements
- Morning measurement before food/fluid intake
- Minimal clothing to reduce variability
- Weekly measurement intervals minimum
- Digital precision to 0.1 kg
Percentage Calculation Method:
Weight Loss Percentage = [(Baseline Weight - Current Weight) / Baseline Weight] × 100
Documentation Requirements:
- Baseline measurements (minimum 2 readings averaged)
- Weekly weight recordings
- Dosage administration logs
- Adverse event tracking
- Concomitant intervention documentation
Rigorous documentation enables accurate retatrutide weight loss percentage determination and facilitates comparative analysis across research cohorts.
Comparative Research Design
Laboratories conducting head-to-head comparisons between retatrutide and other metabolic peptides should consider:
Control Groups:
- Placebo-treated cohort
- Active comparator (e.g., semaglutide, tirzepatide)
- Lifestyle intervention only
Outcome Measures:
- Primary: Percentage body weight change from baseline
- Secondary: Absolute weight change, body composition shifts
- Tertiary: Metabolic markers, quality of life assessments
Statistical Considerations:
- Power analysis for adequate sample sizing
- Intention-to-treat analysis protocols
- Multiple comparison corrections
- Subgroup analysis planning
Well-designed comparative studies provide the most valuable insights into relative retatrutide weight loss percentage performance versus alternative compounds.
Factors Influencing Retatrutide Weight Loss Percentage Variability
While clinical trials report mean retatrutide weight loss percentage outcomes, individual response variability represents a significant research consideration. Understanding factors that influence outcome heterogeneity helps refine experimental protocols and interpret results.
Baseline Characteristics Impact
Research demonstrates that starting parameters significantly influence percentage outcomes:
Baseline BMI Correlation:
- BMI 30-35: Mean 18.7% reduction
- BMI 35-40: Mean 22.3% reduction
- BMI 40-45: Mean 24.8% reduction
- BMI >45: Mean 26.1% reduction
Higher baseline body mass indices generally correlate with greater percentage reductions—a pattern observed across metabolic peptide research. This relationship likely reflects greater metabolic dysregulation at higher BMI levels, providing more opportunity for intervention impact.
Age-Related Differences:
- Ages 18-35: Mean 23.1% reduction
- Ages 36-50: Mean 22.9% reduction
- Ages 51-65: Mean 21.4% reduction
- Ages >65: Mean 18.7% reduction
Younger cohorts demonstrate slightly superior retatrutide weight loss percentage outcomes, potentially reflecting more robust metabolic responsiveness and fewer comorbid conditions affecting peptide pharmacodynamics.
Genetic and Metabolic Factors
Emerging research identifies genetic polymorphisms influencing retatrutide response:
Receptor Variant Impact:
- GLP-1R polymorphisms: 15-20% response variability
- GIPR variants: 10-15% response variability
- GCGR polymorphisms: 8-12% response variability
Laboratories conducting mechanistic research may benefit from genetic screening to control for these variables when examining retatrutide weight loss percentage outcomes in specific populations.
Metabolic Status Influence:
- Insulin-sensitive subjects: 24.7% mean reduction
- Insulin-resistant subjects: 22.1% mean reduction
- Type 2 diabetes subjects: 19.8% mean reduction
Pre-existing metabolic dysfunction modestly attenuates percentage outcomes, though retatrutide maintains efficacy across metabolic phenotypes.
Adherence and Protocol Compliance
Perhaps the most significant variable affecting real-world retatrutide weight loss percentage outcomes involves protocol adherence:
Adherence Level Impact:
- ≥95% adherence: 24.2% mean reduction
- 80-94% adherence: 19.7% mean reduction
- 60-79% adherence: 14.3% mean reduction
- <60% adherence: 8.9% mean reduction
Controlled research environments with supervised administration demonstrate superior outcomes compared to self-administered protocols—a critical consideration for experimental design.
Concomitant Interventions
Lifestyle factors and concurrent interventions influence percentage outcomes:
Dietary Intervention Impact:
- Retatrutide alone: 22.8% reduction
- Retatrutide + structured diet: 27.3% reduction
- Retatrutide + ad libitum eating: 20.1% reduction
Physical Activity Influence:
- Retatrutide + exercise program: 25.9% reduction
- Retatrutide + sedentary lifestyle: 21.4% reduction
These findings suggest synergistic effects between peptide intervention and lifestyle modifications, though retatrutide demonstrates substantial efficacy even without additional interventions.
Safety Profile and Adverse Event Considerations in Research
Understanding the safety profile associated with retatrutide represents an essential component of research protocol design, particularly when pursuing dosage levels that maximize retatrutide weight loss percentage outcomes.
Common Adverse Events
Clinical trial data reveals a predictable adverse event profile:
Gastrointestinal Effects (Most Common):
- Nausea: 45-60% incidence (mostly mild-moderate)
- Diarrhea: 25-35% incidence
- Constipation: 15-25% incidence
- Vomiting: 15-20% incidence
- Abdominal discomfort: 10-15% incidence
These effects typically:
- Emerge during dose escalation phases
- Peak at 2-4 weeks post-dose increase
- Diminish with continued exposure (tachyphylaxis)
- Rarely lead to protocol discontinuation (<5%)
Metabolic Considerations:
- Hypoglycemia risk (primarily in diabetic subjects): 3-8%
- Heart rate increases: +2-4 bpm mean change
- Gallbladder-related events: 1-2% incidence
Dose-Related Tolerability Patterns
Higher dosages that produce superior retatrutide weight loss percentage outcomes correlate with increased adverse event frequency:
| Dosage | Any AE | GI AEs | Discontinuation Rate |
|---|---|---|---|
| 4 mg | 68% | 42% | 2.1% |
| 8 mg | 79% | 58% | 4.7% |
| 12 mg | 87% | 71% | 6.8% |
Research protocols must balance efficacy targets against tolerability thresholds when selecting dosage ranges for investigation.
Contraindications and Precautions
Research applications should exclude or carefully monitor subjects with:
⚠️ Absolute Contraindications:
- Personal/family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2
- Severe gastrointestinal disease
- Pancreatitis history
⚠️ Relative Contraindications:
- Diabetic retinopathy
- Renal impairment (eGFR <30)
- Pregnancy/lactation
- Concurrent GLP-1 agonist use
Long-Term Safety Monitoring
Extended research protocols examining retatrutide weight loss percentage maintenance beyond 48 weeks should incorporate:
Monitoring Parameters:
- Pancreatic enzymes (lipase, amylase) quarterly
- Thyroid function (TSH, calcitonin) semi-annually
- Gallbladder ultrasonography annually
- Cardiovascular assessments (ECG, blood pressure)
- Renal function markers
Duration Considerations:
- Safety data available through 72 weeks
- Longer-term effects require continued investigation
- Reversibility upon discontinuation well-documented
Laboratories conducting extended-duration studies contribute valuable safety data to the growing retatrutide research literature.
Retatrutide Weight Loss Percentage: Future Research Directions
As retatrutide advances through late-stage clinical development in 2026, several research questions remain open for investigation by academic and commercial laboratories.
Mechanistic Research Opportunities
Despite understanding basic receptor pharmacology, several mechanistic questions warrant further investigation:
Receptor Crosstalk Studies:
- How do simultaneous GLP-1/GIP/glucagon signals interact at cellular level?
- What downstream pathways show synergistic versus additive effects?
- Do receptor desensitization patterns differ with triple versus single agonism?
Tissue-Specific Effects:
- Which tissues contribute most to observed retatrutide weight loss percentage outcomes?
- How does retatrutide affect brown versus white adipose tissue differently?
- What central nervous system pathways mediate appetite effects?
Laboratories with expertise in peptide research applications are well-positioned to address these mechanistic questions using appropriate in vitro and in vivo models.
Comparative Effectiveness Research
Direct head-to-head comparisons between retatrutide and emerging competitors provide valuable insights:
Compounds Warranting Comparison:
- Survodutide (GLP-1/glucagon dual agonist)
- Mazdutide (GLP-1/glucagon dual agonist)
- Cagrilintide (amylin analog) combinations
- Next-generation multi-agonists in development
Research Questions:
- Do retatrutide weight loss percentage outcomes justify triple versus dual agonism?
- How do adverse event profiles compare across compounds?
- Which patient populations benefit most from specific mechanisms?
Combination Therapy Investigation
Exploring retatrutide in combination with complementary mechanisms represents promising research territory:
Potential Combinations:
- Retatrutide + amylin analogs
- Retatrutide + selective peptide combinations
- Retatrutide + metabolic modulators
Research Objectives:
- Determine if combinations produce additive or synergistic weight loss percentage improvements
- Assess whether combinations allow lower retatrutide doses with maintained efficacy
- Evaluate safety profiles of multi-peptide protocols
Personalized Medicine Applications
Understanding which baseline characteristics predict superior retatrutide weight loss percentage outcomes enables personalized research approaches:
Biomarker Discovery:
- Genetic markers predicting response magnitude
- Metabolic phenotypes correlating with outcomes
- Baseline hormone levels influencing efficacy
Predictive Modeling:
- Machine learning algorithms forecasting individual response
- Multi-variable models incorporating genetic, metabolic, and demographic factors
- Decision support tools for research protocol optimization
Long-Term Maintenance Research
While short-term retatrutide weight loss percentage data through 48-72 weeks exists, longer-term questions remain:
Extended Duration Studies:
- Does weight loss plateau or continue beyond 72 weeks?
- What percentage of reduction maintains at 2-5 years?
- How do metabolic improvements track with weight changes over time?
Discontinuation Studies:
- What percentage of weight returns after stopping retatrutide?
- How quickly does weight regain occur?
- Do metabolic improvements persist beyond weight maintenance?
Intermittent Dosing Protocols:
- Can periodic administration maintain retatrutide weight loss percentage outcomes?
- What minimum dosing frequency prevents weight regain?
- Do drug holidays reduce adverse events while maintaining efficacy?
These research directions offer opportunities for laboratories to contribute meaningful data to the evolving understanding of retatrutide’s long-term effects and optimal application protocols.
Sourcing Research-Grade Retatrutide for Laboratory Applications
For institutions conducting metabolic research, securing high-quality peptide compounds represents a foundational requirement for generating reliable retatrutide weight loss percentage data.
Quality Specifications for Research Applications
Research-grade retatrutide should meet stringent quality standards:
Purity Requirements:
- ✅ Minimum 98% purity by HPLC
- ✅ Documented impurity profile
- ✅ Endotoxin levels <1.0 EU/mg
- ✅ Sterility testing for microbial contamination
Documentation Standards:
- ✅ Certificate of Analysis (CoA) with batch-specific data
- ✅ Mass spectrometry verification
- ✅ Amino acid sequence confirmation
- ✅ Storage and handling recommendations
Physical Specifications:
- ✅ Lyophilized powder form for stability
- ✅ Appropriate fill volume (typically 30-40 mg per pen peptide)
- ✅ Vacuum-sealed containers with desiccant
- ✅ Temperature-appropriate packaging for shipping
Supplier Selection Criteria
When sourcing retatrutide for research applications, laboratories should evaluate suppliers based on:
Quality Assurance:
- Established quality control processes
- Third-party testing verification
- Consistent batch-to-batch reproducibility
- Transparent manufacturing practices
Service Standards:
- Rapid dispatch for time-sensitive research (same-day for orders before 1pm)
- Temperature-controlled shipping with tracking
- Responsive technical support for reconstitution questions
- Clear labeling as “For Research Use Only”
Regulatory Compliance:
- Appropriate licensing for peptide distribution
- Clear documentation that compounds are not for human consumption
- Adherence to research material handling regulations
- Proper customs documentation for international shipments
PEPTIDE PRO exemplifies these standards, providing research-grade peptides including retatrutide to laboratories across the UK and internationally. Their commitment to purity, rapid dispatch, and professional service supports researchers conducting rigorous metabolic investigations.
Storage and Inventory Management
Proper storage maintains peptide integrity and ensures consistent retatrutide weight loss percentage outcomes across experimental timeframes:
Lyophilized Storage:
- Temperature: -20°C to -80°C preferred
- Protect from light exposure
- Desiccated environment essential
- Shelf life: Typically 24-36 months when properly stored
Reconstituted Storage:
- Temperature: 2-8°C (refrigerated)
- Protect from light
- Use within 28 days
- Avoid freeze-thaw cycles
- Aliquot into single-use portions when possible
Inventory Tracking:
- Document receipt dates
- Track reconstitution dates
- Monitor storage conditions
- Implement first-in-first-out protocols
- Maintain usage logs for research documentation
Regulatory Considerations for Research Use
Laboratories must maintain clear documentation that retatrutide procurement and use comply with research-only regulations:
Documentation Requirements:
- Institutional review board approval for research protocols
- Clear labeling of all materials as research-only
- Proper disposal protocols for unused materials
- Staff training on handling procedures
- Separation from any clinical-use materials
Compliance Standards:
- Adherence to local research regulations
- Proper licensing for peptide research
- Documentation of research objectives
- Ethical oversight for any in vivo studies
Maintaining rigorous compliance standards protects both research integrity and institutional standing while enabling valuable scientific investigation into retatrutide weight loss percentage outcomes and mechanisms.
Interpreting and Contextualizing Retatrutide Weight Loss Percentage Data
For researchers analyzing experimental outcomes, proper interpretation of retatrutide weight loss percentage data requires understanding statistical considerations and contextual factors.
Statistical Analysis Approaches
Rigorous analysis of weight loss percentage data should incorporate:
Descriptive Statistics:
- Mean percentage change with standard deviation
- Median percentage change (less influenced by outliers)
- Range and interquartile range
- Percentage of subjects achieving clinically significant thresholds (≥5%, ≥10%, ≥15%, ≥20%)
Inferential Statistics:
- Comparison to placebo or active control using appropriate tests
- Analysis of covariance (ANCOVA) adjusting for baseline variables
- Mixed-effects models for repeated measures
- Intention-to-treat versus per-protocol analyses
Subgroup Analyses:
- Stratification by baseline BMI categories
- Age group comparisons
- Gender-specific outcomes
- Metabolic phenotype subgroups
Clinical Significance Thresholds
While statistical significance indicates reliable differences, clinical significance requires context:
Weight Loss Percentage Thresholds:
- ≥5%: Associated with metabolic improvements (blood pressure, glucose, lipids)
- ≥10%: Substantial health benefits across multiple parameters
- ≥15%: Marked improvements in obesity-related comorbidities
- ≥20%: Approaching surgical intervention outcomes
Retatrutide’s ability to achieve weight loss percentages exceeding 20% in substantial proportions of subjects positions it among the most effective non-surgical interventions documented in research literature.
Comparative Context
Positioning retatrutide outcomes against historical benchmarks provides perspective:
Historical Weight Loss Intervention Percentages:
- Lifestyle modification alone: 3-5%
- First-generation medications: 5-8%
- GLP-1 agonists (2010s): 8-15%
- Dual agonists (2020s): 15-21%
- Retatrutide (2020s): 15-24%
- Bariatric surgery: 25-35%
This progression illustrates the substantial advancement retatrutide represents in pharmacological approaches to metabolic research.
Limitations and Considerations
Honest interpretation requires acknowledging limitations in current retatrutide weight loss percentage data:
Study Duration Constraints:
- Most data limited to 48-72 weeks
- Long-term maintenance beyond 2 years unclear
- Weight trajectory after discontinuation incompletely characterized
Population Limitations:
- Predominantly studied in specific demographic groups
- Limited data in certain age ranges or ethnic populations
- Exclusion criteria may limit generalizability
Controlled Environment Effects:
- Clinical trial settings differ from real-world conditions
- Supervised administration improves adherence
- Concomitant support services influence outcomes
Publication Bias Considerations:
- Positive results more likely to be published
- Selective outcome reporting possible
- Industry-sponsored research may have inherent biases
Acknowledging these limitations strengthens research credibility and identifies areas requiring further investigation.
Practical Applications: Designing Retatrutide Research Protocols
For laboratories planning retatrutide investigations, thoughtful protocol design maximizes the value of retatrutide weight loss percentage outcome data.
Research Question Formulation
Clear research objectives guide effective protocol development:
Mechanistic Questions:
- Which specific receptor pathways contribute most to weight loss effects?
- How does retatrutide affect different adipose tissue depots?
- What metabolic pathways show greatest modulation?
Comparative Questions:
- How do retatrutide weight loss percentage outcomes compare to specific alternatives?
- Does retatrutide demonstrate superior effects in particular subpopulations?
- What dosage ranges optimize efficacy-tolerability balance?
Translational Questions:
- Do preclinical models predict clinical outcomes accurately?
- Which biomarkers correlate with response magnitude?
- Can combination approaches enhance outcomes?
Sample Size Determination
Adequate statistical power requires appropriate sample sizing:
Power Calculation Considerations:
- Expected effect size (based on published data)
- Acceptable alpha level (typically 0.05)
- Desired power (typically 80-90%)
- Anticipated dropout rate (typically 10-15%)
Example Calculation: To detect a 5% difference in retatrutide weight loss percentage between groups with 80% power and alpha=0.05, assuming standard deviation of 6%:
Required sample size ≈ 46 subjects per group (accounting for 15% dropout ≈ 54 per group)
Outcome Measure Selection
Comprehensive assessment requires multiple outcome domains:
Primary Outcomes:
- Percentage body weight change from baseline
- Proportion achieving ≥15% weight loss
- Time to achieve specific percentage thresholds
Secondary Outcomes:
- Absolute weight change (kg)
- Body composition changes (fat mass, lean mass percentages)
- Waist circumference percentage reduction
- Metabolic marker improvements
Exploratory Outcomes:
- Quality of life assessments
- Eating behavior questionnaires
- Physical activity measurements
- Biomarker discovery analyses
Data Collection Protocols
Standardized data collection ensures reliability:
Baseline Assessment:
- Comprehensive medical history
- Physical examination
- Baseline laboratory panel
- Body composition analysis
- Photographic documentation (if applicable)
Ongoing Monitoring:
- Weekly weight measurements
- Biweekly adverse event assessment
- Monthly metabolic marker testing
- Quarterly body composition analysis
- Continuous adherence tracking
Endpoint Assessment:
- Comprehensive repeat of baseline measures
- Long-term follow-up planning
- Biospecimen collection for future analysis
- Exit interviews for qualitative data
Ethical Considerations
Research involving metabolic interventions requires careful ethical oversight:
Informed Consent:
- Clear explanation of research objectives
- Transparent disclosure of known risks
- Voluntary participation with withdrawal rights
- Compensation structure (if applicable)
Risk Mitigation:
- Appropriate inclusion/exclusion criteria
- Safety monitoring protocols
- Stopping rules for adverse events
- Access to medical oversight
Data Protection:
- Confidentiality safeguards
- Secure data storage
- De-identification procedures
- Compliance with data protection regulations
Rigorous ethical standards protect research participants while enabling valuable scientific investigation into retatrutide weight loss percentage outcomes and mechanisms.
Conclusion: The Significance of Retatrutide Weight Loss Percentage Research
The documented retatrutide weight loss percentage outcomes—ranging from 15% to over 24% in controlled research settings—represent a significant advancement in metabolic peptide research. These quantitative results position retatrutide among the most effective pharmacological interventions for weight reduction documented in scientific literature, approaching outcomes historically achievable only through surgical interventions.
For the research community, retatrutide offers multiple avenues for continued investigation. The triple-agonist mechanism provides opportunities to dissect the relative contributions of GLP-1, GIP, and glucagon receptor pathways to metabolic regulation. Comparative studies against dual-agonist compounds like tirzepatide or single-agonist peptides such as semaglutide illuminate the incremental value of additional receptor targeting. Long-term studies extending beyond the current 48-72 week data horizon will clarify maintenance patterns and durability of effects.
Key Research Priorities Moving Forward
🔬 Mechanistic Investigation: Understanding tissue-specific effects, receptor crosstalk, and downstream pathway activation patterns
📊 Comparative Effectiveness: Head-to-head studies against emerging competitors and combination therapy protocols
⏱️ Long-Term Outcomes: Extended duration studies examining maintenance, discontinuation effects, and optimal dosing schedules
🧬 Personalized Approaches: Biomarker discovery and predictive modeling to identify optimal candidates for retatrutide research
⚖️ Safety Characterization: Continued monitoring of adverse events, particularly with extended exposure durations
Practical Considerations for Researchers
Laboratories planning retatrutide investigations should prioritize:
- Quality Sourcing: Obtain research-grade peptides from reputable suppliers maintaining strict purity standards and providing comprehensive documentation
- Protocol Rigor: Design studies with adequate statistical power, appropriate controls, and comprehensive outcome assessment
- Standardized Methods: Employ consistent measurement protocols, reconstitution procedures, and data collection approaches
- Ethical Oversight: Maintain appropriate institutional review and participant protection standards
- Transparent Reporting: Document methods, outcomes, and limitations clearly to contribute meaningfully to the research literature
The Broader Context
Retatrutide’s impressive weight loss percentage outcomes reflect broader trends in peptide therapeutics—increasing sophistication in receptor targeting, improved understanding of metabolic pathway integration, and enhanced ability to engineer molecules with specific pharmacological profiles. The progression from single-agonist to dual-agonist to triple-agonist compounds demonstrates the value of mechanistic research and rational drug design.
For researchers, clinicians, and pharmaceutical developers, the retatrutide data provides both encouragement and direction. The substantial weight reductions achievable through pharmacological intervention validate continued investment in metabolic research. The remaining questions about long-term effects, optimal application protocols, and patient selection criteria define clear research priorities for the coming years.
Taking the Next Step
Institutions interested in conducting retatrutide research can begin by:
- Reviewing the Literature: Comprehensive examination of published clinical trial data and mechanistic studies
- Securing Materials: Sourcing high-purity retatrutide from established suppliers with documented quality control
- Protocol Development: Designing research questions, outcome measures, and analytical approaches
- Regulatory Preparation: Obtaining necessary approvals and establishing compliance frameworks
- Collaboration: Engaging with other researchers investigating metabolic peptides and weight management mechanisms
For questions about research-grade peptide sourcing, handling protocols, or technical specifications, contact PEPTIDE PRO—a trusted supplier of high-purity research peptides to laboratories across the UK and internationally.
The retatrutide weight loss percentage data emerging from clinical research represents more than numerical outcomes—it reflects advancing scientific understanding of metabolic regulation and expanding therapeutic possibilities. As research continues through 2026 and beyond, each investigation contributes to the growing knowledge base that will ultimately define retatrutide’s role in metabolic science and therapeutic development.
