
1.The New Threshold of Metabolic Efficacy
The global obesity crisis has historically been met with a pharmaceutical shrug. For decades, the available tools—phentermine, orlistat, lorcaserin—offered modest weight loss, rarely exceeding 5–8% of total body weight. This was sufficient for cosmetic tweaking but insufficient for reversing the deep metabolic damage of Type 2 Diabetes, hypertension, and non-alcoholic steatohepatitis (NASH).
The arrival of Semaglutide (a GLP-1 mono-agonist) changed the conversation by proving that pharmacological agents could achieve ~15% weight loss. But as the research community digs deeper into the pathophysiology of obesity, we are realizing that even 15% is a floor, not a ceiling. The goalposts have moved. The new gold standard is “bariatric-mimetic” efficacy—weight loss exceeding 20% or 25%.
Enter Survodutide (BI 456906).
Co-developed by Boehringer Ingelheim and Zealand Pharma, Survodutide for weight loss represents the next evolutionary step: the Dual Agonist. By combining the proven appetite suppression of GLP-1 with the energy-expending power of Glucagon, Survodutide aims to break the “metabolic adaptation” that limits mono-agonist therapy.
This deep-dive review analyzes the pivotal Phase 2 clinical data published in the New England Journal of Medicine and presented at the European Association for the Study of Diabetes (EASD). We will dissect the numbers, scrutinize the safety profile, and evaluate whether Survodutide is merely another GLP-1 or a true paradigm shift in obesity medicine.
2. The Phase 2 Data: Deconstructing the 46-Week Findings
The primary source of our current understanding comes from the Phase 2, randomized, double-blind, placebo-controlled dose-finding trial (NCT04667377).
Study Design
-
Population: 387 adults with a BMI of ≥27 kg/m².
-
Duration: 46 weeks (20 weeks of dose escalation + 26 weeks of maintenance).
-
Arms: Patients were randomized to receive subcutaneous Survodutide at doses of 0.6mg, 2.4mg, 3.6mg, 4.8mg, or Placebo.
-
Titration: A rapid dose-escalation protocol (every 2 weeks) versus a slower protocol (every 4 weeks) was also tested to assess tolerability.
The Headline Result: 18.7% Weight Loss
The data was analyzed using two statistical estimands, a crucial distinction for researchers interpreting the “real world” versus “ideal world” efficacy.
-
Trial Product Estimand (Ideal): This analysis assumes all participants stayed on the drug for the full 46 weeks. In this scenario, the 4.8mg dose group achieved a mean weight loss of 18.7%.
-
Treatment Policy Estimand (Real World): This analysis includes everyone, even those who quit the study due to side effects. In this scenario, the 4.8mg group still achieved a mean weight loss of 14.9%.
Key Insight: The gap between 18.7% and 14.9% highlights the potency of the drug but also the challenge of tolerability. If patients can tolerate the drug (staying on it), the efficacy approaches 20% in less than a year.
The Comparison to Placebo
The placebo group lost approximately 2.8% of body weight (likely due to the “Hawthorne Effect” of being in a trial and receiving lifestyle counseling).
-
The Delta: Survodutide 4.8mg offered a treatment benefit of ~16% over placebo. This is a statistically massive margin (P<0.001), confirming that the weight loss is pharmacologically driven, not behavioral.
3. Dose-Response Relationship: The “Linear” Efficacy
One of the most compelling aspects of Survodutide for weight loss is the linear dose-response curve observed in Phase 2. Unlike some drugs that plateau quickly, Survodutide showed that “more is more.”
The Dose Ladder
-
0.6 mg: ~6% weight loss. (Sub-therapeutic for obesity, but distinct separation from placebo).
-
2.4 mg: ~12% weight loss. (Comparable to standard Semaglutide doses).
-
3.6 mg: ~16% weight loss.
-
4.8 mg: ~19% weight loss.
No Plateau Observed
Crucially, at week 46, the weight loss curves for the 3.6mg and 4.8mg doses had not flattened. They were still trending downwards.
-
Extrapolation: This suggests that if the trial had continued to 68 or 72 weeks (standard for Phase 3), the total weight loss could easily exceed 20–22%. The “Glucagon effect” (increased metabolic rate) appears to prevent the dreaded weight loss plateau often seen at month 8 with GLP-1 mono-agonists.
The Tolerability Trade-Off
However, the dose-response for adverse events (AEs) was also linear.
-
GI Issues: Nausea and vomiting were dose-dependent. In the rapid titration groups, discontinuation rates were high.
-
The Fix: The study found that a slower titration (escalating every 4 weeks instead of 2) significantly reduced dropout rates while maintaining the same final weight loss efficacy. This finding has dictated the protocol design for the ongoing Phase 3 trials.
4. Comparison to Semaglutide: The Battle of the Titans
How does Survodutide for weight loss stack up against the current market leader, Semaglutide 2.4mg (Wegovy)?
| Metric | Semaglutide 2.4mg (STEP 1) | Survodutide 4.8mg (Phase 2) |
| Duration | 68 Weeks | 46 Weeks |
| Mean Weight Loss | ~14.9% | ~18.7% |
| Mechanism | Mono-Agonist (Appetite) | Dual Agonist (Appetite + Burn) |
| Plateau | Observed at ~60 weeks | Not observed at 46 weeks |
| Discontinuation | ~4-7% | ~20% (Rapid) / Lower (Slow) |
Analysis of Superiority
-
Speed: Survodutide achieved nearly 19% loss in just 46 weeks, whereas Semaglutide took 68 weeks to reach 15%. Survodutide is chemically faster at stripping fat.
-
Potency: The addition of the Glucagon receptor agonist (GCGR) creates a caloric deficit from two sides. Semaglutide only reduces “Calories In.” Survodutide reduces “Calories In” AND increases “Calories Out.”
-
The Caveat: The side effect profile of Survodutide is “spicier.” The Glucagon component adds a layer of complexity (heart rate, more intense nausea) that Semaglutide lacks. Survodutide is a Ferrari; Semaglutide is a Lexus. One is faster, but the other is a smoother ride.
5. Beyond Weight: Secondary Endpoints & The “Glucagon Bonus”
Researching Survodutide for weight loss solely based on the scale misses half the story. The Phase 2 data revealed profound improvements in cardiometabolic markers that suggest systemic healing.
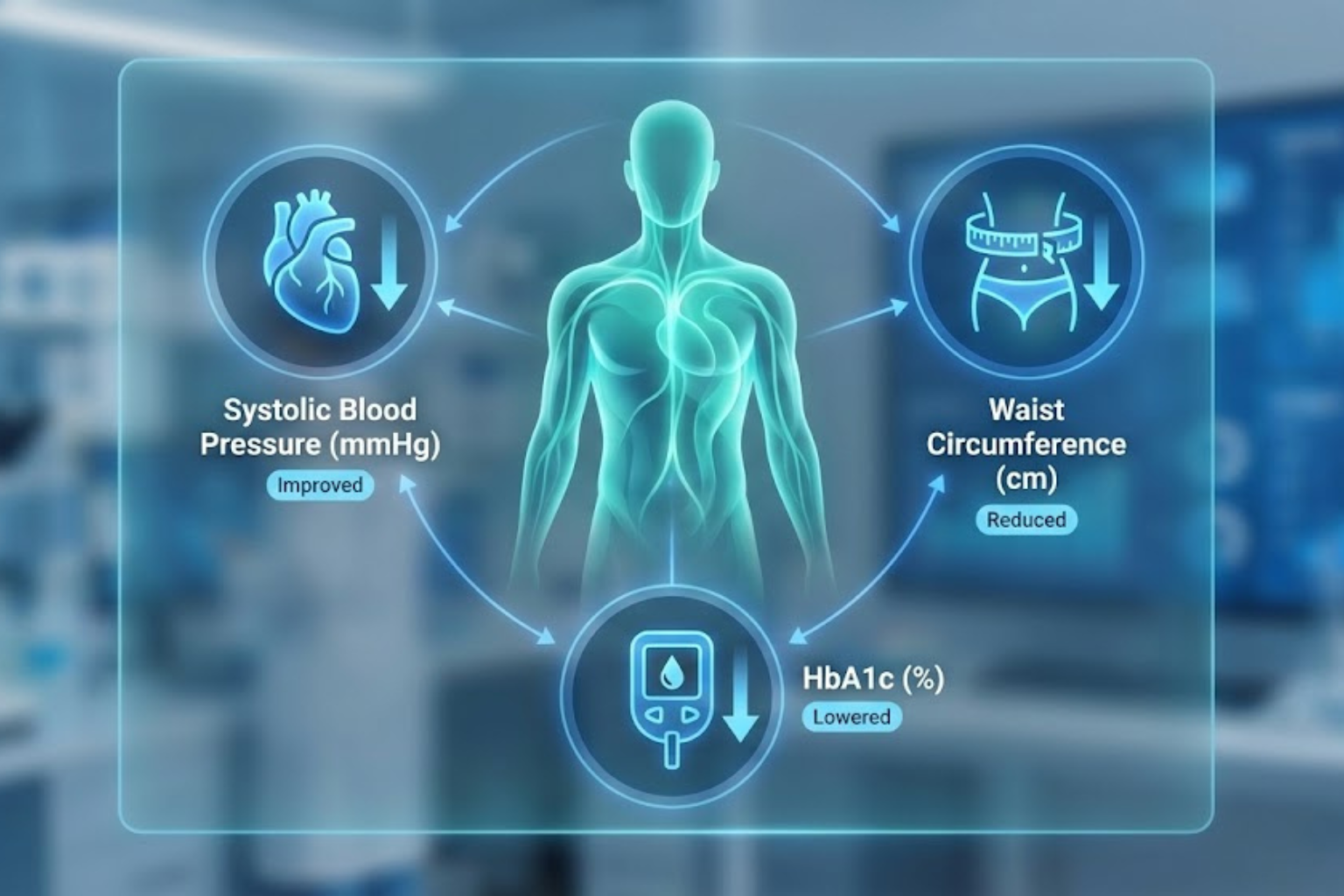
Blood Pressure
-
Result: A mean reduction in systolic blood pressure of ~6-8 mmHg.
-
Mechanism: Weight loss is the primary driver, but Glucagon also has a natriuretic effect (promoting sodium excretion by the kidneys), which may contribute to the BP lowering independent of weight.
Waist Circumference
-
Result: Reductions of 13–16 cm.
-
Significance: This confirms the loss of visceral fat. Visceral adipose tissue (VAT) is the metabolically dangerous fat wrapped around organs. Glucagon specifically targets hepatic and visceral lipid stores for mobilization.
HbA1c (Glycemic Control)
-
Result: Despite Glucagon’s theoretical risk of raising blood sugar, Survodutide lowered HbA1c significantly.
-
The Safety Check: This confirms that the GLP-1 component (which stimulates insulin) is strong enough to completely override the glucagon-induced hepatic glucose output. Patients got the fat-burning benefits of Glucagon without the hyperglycemic penalty.
The Liver (MASH) Data
While discussed in other blogs, it is relevant here. A subset of patients showed rapid normalization of liver enzymes (ALT/AST).
-
The Implication: For obese patients with fatty liver (which is ~70% of the obese population), Survodutide treats the liver disease directly, whereas Semaglutide treats it indirectly via weight loss.
6. The Future: What to Expect from Phase 3 (SYNCHRONIZE)
Boehringer Ingelheim has launched the massive SYNCHRONIZE Phase 3 program. Based on the Phase 2 learnings, what can we project?
Protocol Optimization
Phase 3 uses the slower titration schedule.
-
Prediction: This will lower the discontinuation rate. If more people stay on the drug, the “Treatment Policy” (Real World) efficacy number will rise to meet the “Trial Product” (Ideal) number. We expect to see intention-to-treat results crossing the 20% barrier.
Longer Duration (76 Weeks)
Phase 2 stopped at 46 weeks while the curve was still dropping.
-
Prediction: With an additional 30 weeks of treatment, Survodutide 4.8mg (or potentially higher doses if tested) could approach 22–24% total weight loss. This would put it in direct competition with Retatrutide (Triple Agonist) and perilously close to the results of a gastric sleeve.
Head-to-Head Comparisons
While not yet fully disclosed, future trials will likely pit Survodutide directly against Semaglutide or Tirzepatide.
-
The Battleground: The fight will not just be about “who lost more weight,” but “who improved liver fibrosis more.” Survodutide is betting the house on the Liver/MASH endpoint as its unique selling proposition.
7. Safety Profile Deep Dive: The Heart Rate Question
No clinical review is honest without addressing the safety signals found in the data.
Heart Rate Elevation
-
Data: Survodutide treatment was associated with a mean increase in resting pulse rate of +3 to +6 beats per minute (bpm).
-
Context: This is a known effect of Glucagon receptor agonism (chronotropy).
-
Risk: For a healthy 30-year-old, this is negligible. For a 65-year-old with ischemic heart disease, it is a variable that requires monitoring. The Phase 3 trials will have stringent cardiovascular safety monitoring to ensure this doesn’t translate into adverse cardiac events (MACE).
Gastrointestinal Tolerance
-
Data: ~90% of participants reported at least one adverse event, primarily GI (nausea, diarrhea, vomiting).
-
Analysis: This sounds high, but it is consistent with the incretin class. The key metric is severity. Most cases were mild-to-moderate and occurred during the dose-escalation phase. Once patients reached the maintenance dose, the incidence of nausea dropped significantly (“tachyphylaxis”).
8.Survodutide as a Potential Market Leader
Reviewing the clinical data reveals that Survodutide for weight loss is a highly potent therapeutic candidate. It offers a “one-two punch” that Semaglutide cannot match:
-
Potency: ~19% weight loss in <1 year, with a trajectory toward >20%.
-
Specific Utility: A unique mechanism (Glucagon backbone) that specifically torches liver fat and preserves metabolic rate.
For the researcher, the data suggests that Survodutide is the ideal candidate for studying severe obesity complicated by metabolic liver disease. While Tirzepatide (Mounjaro) currently holds the crown, Survodutide is carving out a massive niche as the “Hepatologist’s Choice” for weight loss.
As Phase 3 data matures in 2025/2026, we expect Survodutide to be approved not just as an anti-obesity medication, but as the first true pharmacological cure for MASH-driven obesity.
9. Statistical Appendix (For Researchers)
| Metric | Survodutide 4.8mg (46 Weeks) | Placebo (46 Weeks) | P-Value |
| Mean % Weight Change | -18.7% | -2.8% | <0.001 |
| % Achieving >5% Loss | ~90% | ~25% | <0.001 |
| % Achieving >10% Loss | ~75% | ~12% | <0.001 |
| % Achieving >15% Loss | ~55% | ~5% | <0.001 |
| Discontinuation (AEs) | ~24% | ~2% | N/A |
Data Source: Le Roux, C. W., et al. (2023). Dose-response efficacy of the dual glucagon receptor/GLP-1 receptor agonist survodutide in people with overweight or obesity (NCT04667377).
10. References
-
Le Roux, C. W., et al. (2024). Glucagon and GLP-1 Receptor Dual Agonist Survodutide for Obesity: A Phase 2 Randomized Clinical Trial. The Lancet.
-
Wilding, J. P. H., et al. (2021). Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP 1). NEJM.
-
Garvey, W. T., et al. (2023). Tirzepatide once weekly for the treatment of obesity in people with type 2 diabetes (SURMOUNT-2). The Lancet.
