
TB-500 and the Future of Soft Tissue Recovery
In the complex field of regenerative medicine, the greatest challenge has always been the disparity in healing rates between different tissue types. While skin and liver possess remarkable regenerative capabilities, the musculoskeletal system—specifically tendons, ligaments, and cardiac tissue—often struggles to repair itself effectively. The most common application of Thymosin Beta-4 in the laboratory today is investigating this exact problem: soft tissue repair.
TB-500 for injury recovery research has exploded in popularity, driven by the peptide’s unique ability to address the fundamental biological bottlenecks of healing. It is no longer just about speeding up a process; it is about changing the quality of the repair. For tissues that notoriously heal poorly, often resulting in chronic weakness or dysfunction, TB-500 represents a shift from “patching” the damage to “regenerating” the structure. This article examines how TB-500 is changing the landscape of recovery research, moving from theoretical biochemistry to tangible structural restoration.
2. Tendinopathy and Ligament Research: Solving the Vascular Deficit
To understand why TB-500 is so critical for tendon and ligament research, one must first understand the anatomy of these tissues. Tendons (connecting muscle to bone) and ligaments (connecting bone to bone) are dense connective tissues composed primarily of Type I collagen. However, they share a critical flaw: they are avascular or hypovascular.
The Avascular Challenge
Unlike muscle, which is red and rich in blood vessels, tendons are white and fibrous. They rely largely on synopen peptide fluid diffusion for nutrients rather than direct blood flow. When a tendon is injured (tendinopathy), the body struggles to deliver the necessary immune cells, oxygen, and building blocks to the site. Consequently, the body resorts to a “quick fix”: it lays down scar tissue (Type III collagen) instead of the original, robust Type I collagen.
-
The Result: Scar tissue is disorganized, weaker, and less elastic. This leads to a high rate of re-injury and chronic pain.
The Angiogenic Solution
Research into TB-500 for injury focuses heavily on its angiogenic properties—the ability to stimulate the growth of new blood vessels. In animal models of Achilles tendon injury, TB-500 has been observed to stimulate the migration of endothelial cells into the injured tendon matrix.
-
Mechanism: By upregulating factors like VEGF (Vascular Endothelial Growth Factor) and stabilizing Hypoxia-Inducible Factor 1-alpha (HIF-1α), TB-500 essentially forces the body to build a new vascular network around the injury.
-
Outcome: This increased perfusion brings a surge of oxygen and nutrients. Studies suggest that tendons treated with TB-500 show a more organized collagen deposition, with fibers aligning in parallel (like healthy tendon) rather than the “basket-weave” pattern of scar tissue. This restoration of structure correlates with a restoration of tensile strength, potentially returning the tissue to pre-injury performance levels.
3. Muscle Tear and Strain Models: Awakening the Satellite Cell
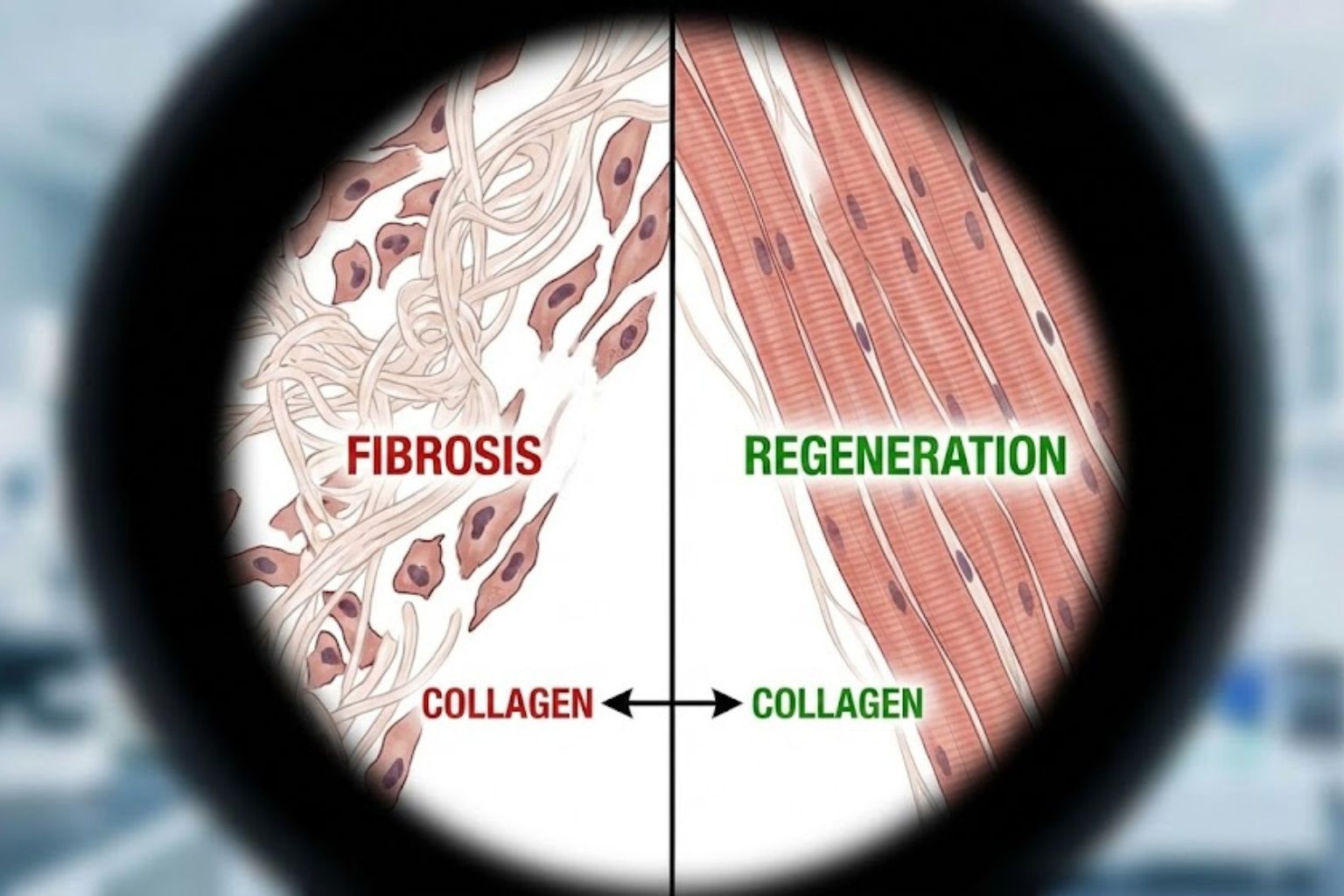
Muscle tissue heals differently than tendon, but it faces its own challenges, primarily fibrosis. A severe muscle tear often heals with a fibrotic scar that interrupts the contractile fibers, leaving a “dead zone” in the muscle that is prone to re-tearing.
The Satellite Cell: The Hero of Regeneration
The key to true muscle regeneration lies in a specific type of stem cell called the satellite cell. These cells sit dormant on the periphery of muscle fibers. When a muscle is strained or torn, these cells must wake up, divide, and fuse with the damaged fibers to repair them.
TB-500 and Myogenesis
Studies suggest that TB-500 for injury protocols acts as a potent activator of these satellite cells.
-
Activation: TB-500 signaling pathways (likely involving the AKT/mTOR pathway) trigger satellite cells to exit their quiescent state and enter the cell cycle.
-
Differentiation: Once active, these cells differentiate into myoblasts (muscle precursors) and fuse to form new myofibers.
-
Actin Regulation: As discussed in cytoskeletal studies, TB-500’s regulation of G-actin is crucial here. Forming new muscle fibers requires massive cytoskeletal remodeling. TB-500 provides the “raw materials” for this construction.
Preventing Fibrosis
Crucially, animal models with induced muscle trauma treated with TB-500 showed a significant reduction in fibrosis. By accelerating the myogenic (muscle-building) phase, TB-500 effectively “outpaces” the fibrotic (scar-building) phase. The result is a healed muscle with better fiber alignment, less stiffness, and greater contractile force compared to untreated controls.
4. Cardiac Repair Studies: The “Holy Grail” of Recovery
Perhaps the most groundbreaking—and high-stakes—research regarding TB-500 is in the heart. The heart is unique because adult cardiomyocytes (heart muscle cells) have almost zero capacity to regenerate.
The Problem: Myocardial Infarction
Following a myocardial infarction (heart attack), a portion of the heart muscle is starved of oxygen and dies. Because the heart cannot grow new muscle, it replaces the dead tissue with a non-contractile collagen scar. This scar maintains the structural integrity of the heart wall so it doesn’t burst, but it cannot pump. This loss of pumping power leads to progressive heart failure.
The Epicardial Activation
Thymosin Beta-4 has been shown in murine (mouse) models to stimulate the epicardium—the outer layer of the heart. In embryonic development, the epicardium is a source of cells that migrate inward to become heart muscle and vessels. In adults, this layer is dormant.
-
Re-awakening Development: TB-500 appears to “remind” the adult epicardium of its embryonic role. Treated subjects showed epicardial cells migrating into the damaged heart wall and differentiating into new cardiomyocytes and new blood vessels (neovascularization).
-
Cell Survival: Additionally, TB-500 has a potent anti-apoptotic effect. It prevents “stunned” heart cells in the border zone of the attack from dying, salvaging tissue that would otherwise be lost.
While still in the experimental phase, this suggests TB-500 could transform a heart attack from a permanent disability into a recoverable injury.
5. Flexibility and Inflammation: The Chronic Injury Loop
Chronic injuries, such as “tennis elbow” (lateral epicondylitis) or rotator cuff tendinopathy, are often driven by a cycle of chronic inflammation. This inflammation leads to edema (swelling), which restricts blood flow further, leading to tissue hypoxia and degradation.
Modulating the Microenvironment
TB-500 acts as a modulator of this inflammatory response. It does not just suppress the immune system (which would hinder healing); it alters the profile of cytokines.
-
Downregulation: It reduces the expression of pro-inflammatory cytokines like TNF-alpha and IL-1beta.
-
Upregulation: It promotes anti-inflammatory pathways that encourage tissue remodeling.
Maintaining Flexibility
The reduction in swelling and edema has a mechanical benefit: flexibility. Chronic inflammation makes tissues stiff and brittle. By resolving the fluid accumulation and preventing the formation of rigid scar tissue adhesions, TB-500 helps maintain the viscoelastic properties of the soft tissue. This is vital for athletes and aging populations alike, as flexible tissues are far less likely to suffer catastrophic ruptures under load.
The body of evidence supporting TB-500 for injury research is compelling because it addresses the universal failures of mammalian healing: poor blood flow, scar formation, and lack of stem cell activity.
From encouraging blood flow in avascular tendons to preventing fibrosis in skeletal muscle and potentially regenerating heart tissue, this peptide offers a multi-faceted approach to tissue regeneration. It suggests a future where medical treatment for injuries involves not just immobilizing the limb and waiting, but actively stimulating the body’s latent architectural plans to rebuild what was lost. As research moves from animal models to controlled clinical settings, TB-500 remains one of the most promising candidates in the pharmacopoeia of recovery.
