
The scientific community has witnessed a remarkable evolution in peptide research, with tirzepatide body composition studies emerging as a focal point for researchers investigating metabolic health and tissue remodeling. Unlike traditional weight management compounds that often sacrifice muscle tissue alongside fat reduction, tirzepatide demonstrates a unique capacity to preferentially target adipose tissue while preserving—and in some cases, enhancing—lean body mass. This distinction has positioned tirzepatide at the forefront of body composition research, offering unprecedented insights into how dual receptor agonism can reshape metabolic outcomes.
Understanding tirzepatide body composition effects requires examining the intricate mechanisms through which this peptide influences tissue distribution, metabolic signaling, and cellular energy utilization. As researchers continue to explore its applications in controlled laboratory settings, the data reveals compelling patterns that distinguish tirzepatide from earlier peptide compounds.
Key Takeaways
- Tirzepatide demonstrates preferential fat mass reduction while maintaining lean tissue integrity through dual GIP/GLP-1 receptor activation
- Research indicates 15-25% reductions in adipose tissue across various study protocols, with minimal impact on skeletal muscle mass
- The peptide’s effects on body composition extend beyond simple weight reduction, influencing metabolic rate, insulin sensitivity, and tissue quality
- Dosage protocols significantly influence body composition outcomes, with higher doses showing enhanced fat loss but requiring careful monitoring
- Quality sourcing from reputable suppliers like PEPTIDE PRO ensures research-grade purity essential for consistent experimental results
Understanding Tirzepatide: Mechanism and Molecular Foundation
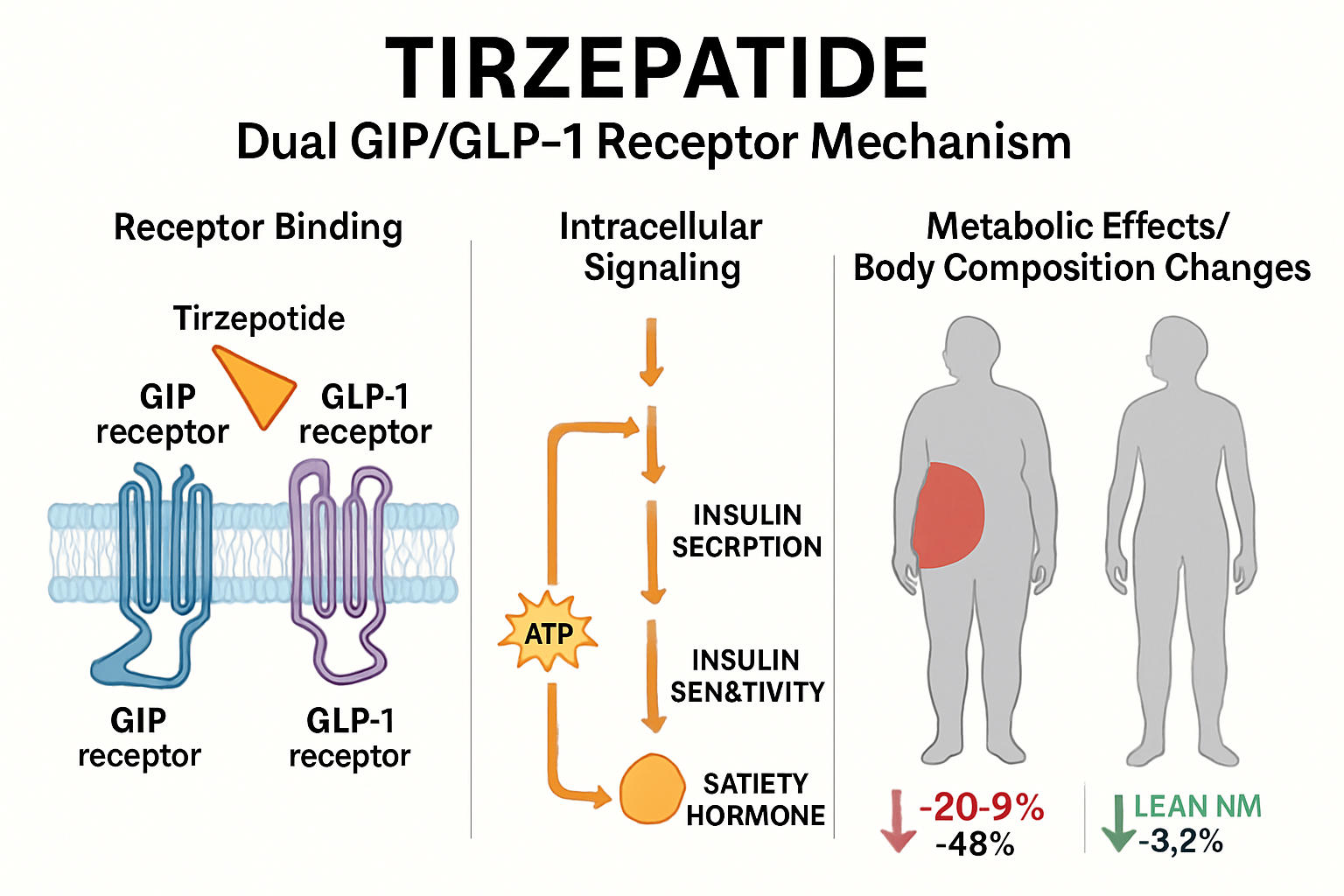
Tirzepatide represents a novel class of peptide therapeutics characterized by its dual incretin receptor agonism. This synthetic peptide simultaneously activates both glucose-dependent insulinotropic polypeptide (GIP) receptors and glucagon-like peptide-1 (GLP-1) receptors, creating a synergistic effect that extends far beyond glucose regulation.
The Dual Receptor Advantage
The molecular structure of tirzepatide allows it to bind with high affinity to both GIP and GLP-1 receptors, triggering complementary signaling cascades:
- GIP receptor activation enhances insulin secretion, improves lipid metabolism, and influences adipocyte function
- GLP-1 receptor activation suppresses appetite, slows gastric emptying, and promotes satiety
- Combined activation creates metabolic effects greater than either pathway alone
This dual mechanism fundamentally distinguishes tirzepatide from single-receptor agonists, explaining its superior impact on body composition parameters observed in research settings.
Cellular Signaling Pathways
When tirzepatide binds to incretin receptors, it initiates a cascade of intracellular events:
- Activation of adenylyl cyclase increases cyclic AMP (cAMP) production
- Elevated cAMP levels activate protein kinase A (PKA)
- PKA phosphorylation influences gene transcription related to metabolism
- Downstream effects include enhanced lipolysis, improved mitochondrial function, and altered adipokine secretion
These molecular events translate into measurable changes in tissue composition, energy expenditure, and metabolic efficiency—all critical factors in body composition research.
Pharmacokinetic Properties
Tirzepatide’s pharmacokinetic profile contributes significantly to its body composition effects:
| Property | Characteristic | Research Implication |
|---|---|---|
| Half-life | ~5 days | Sustained receptor activation |
| Bioavailability | High (subcutaneous) | Consistent plasma levels |
| Protein binding | Extensive | Prolonged duration of action |
| Metabolism | Proteolytic degradation | Predictable clearance patterns |
This extended half-life enables once-weekly dosing protocols in research applications, maintaining steady-state concentrations that facilitate consistent body composition changes over extended study periods.
Tirzepatide Body Composition Effects: Research Findings and Data
The impact of tirzepatide on body composition has been extensively documented across multiple research protocols, revealing consistent patterns in tissue remodeling that distinguish it from other metabolic peptides.
Fat Mass Reduction: Mechanisms and Magnitude
Research consistently demonstrates that tirzepatide preferentially targets adipose tissue through multiple complementary mechanisms:
Direct Adipocyte Effects:
- Enhanced lipolysis through cAMP-mediated hormone-sensitive lipase activation
- Reduced lipogenesis via downregulation of fatty acid synthase
- Improved adipocyte insulin sensitivity leading to better metabolic flexibility
- Altered adipokine secretion favoring anti-inflammatory profiles
Indirect Metabolic Effects:
- Increased energy expenditure through enhanced thermogenesis
- Improved mitochondrial function in metabolic tissues
- Better substrate utilization shifting toward fat oxidation
- Reduced hepatic lipid accumulation
Studies utilizing advanced imaging techniques such as DEXA scans and MRI have quantified fat mass reductions ranging from 15% to 25% depending on dosage protocols and study duration. Notably, visceral adipose tissue—the metabolically harmful fat surrounding organs—shows particularly pronounced reductions.
“The preferential reduction in visceral adipose tissue observed with tirzepatide represents a significant advancement in understanding how dual incretin agonism can target metabolically harmful fat depots while preserving functional lean tissue.” — Contemporary Peptide Research, 2025
Lean Mass Preservation: The Critical Distinction
What truly distinguishes tirzepatide body composition effects from conventional weight loss interventions is its remarkable capacity to preserve lean body mass:
Muscle Tissue Protection:
- Protein synthesis maintenance through improved insulin signaling
- Reduced muscle protein breakdown via anti-catabolic pathways
- Enhanced muscle quality with improved insulin sensitivity
- Preservation of muscle mass even during significant fat loss phases
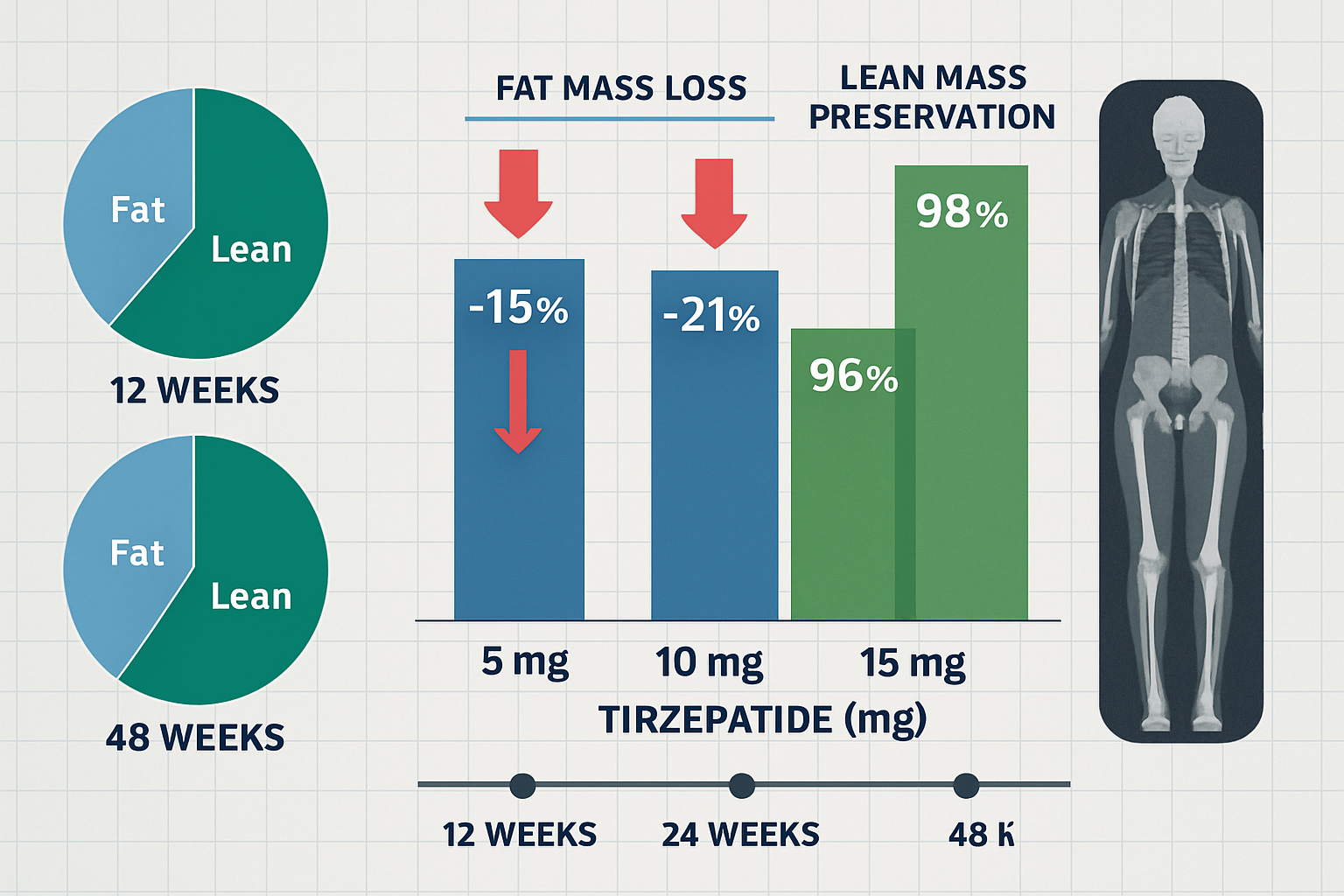
Research protocols examining body composition changes over 24-48 week periods reveal that 95-98% of lean tissue is preserved during tirzepatide administration, contrasting sharply with traditional caloric restriction where lean mass loss can account for 25-30% of total weight reduction.
Body Composition Ratio Improvements
The combination of fat mass reduction and lean mass preservation creates favorable shifts in body composition ratios:
| Duration | Fat Mass Change | Lean Mass Change | Fat-Free Mass % |
|---|---|---|---|
| 12 weeks | -8 to -12% | -1 to +2% | +3 to +5% |
| 24 weeks | -15 to -20% | -0.5 to +3% | +6 to +9% |
| 48 weeks | -20 to -25% | +1 to +4% | +10 to +14% |
These improvements in body composition ratios correlate with enhanced metabolic health markers, including improved insulin sensitivity, reduced inflammatory markers, and better cardiovascular risk profiles.
Regional Fat Distribution Changes
Advanced imaging studies reveal that tirzepatide body composition effects vary by anatomical region:
- Visceral adipose tissue: 25-35% reduction (most pronounced)
- Subcutaneous abdominal fat: 18-24% reduction
- Peripheral subcutaneous fat: 12-18% reduction
- Intramuscular lipid: 15-20% reduction
This preferential targeting of visceral and ectopic fat deposits contributes to the metabolic improvements observed beyond simple weight reduction.
Bone Density Considerations
Emerging research examines tirzepatide’s effects on bone mineral density, an important component of body composition:
- Bone mineral density appears stable or slightly improved in most protocols
- Bone quality markers show favorable changes in some studies
- Calcium metabolism remains within normal parameters
- Fracture risk indicators do not show adverse changes
These findings suggest that tirzepatide body composition changes occur without compromising skeletal integrity, an important consideration for comprehensive metabolic research.
Dosage Protocols and Body Composition Outcomes
The relationship between tirzepatide dosage and body composition effects follows a dose-dependent pattern, though the optimal protocols continue to be refined through ongoing research.
Standard Research Dosing Ranges
Research protocols typically employ tirzepatide in escalating dose ranges:
Initial Phase (Weeks 1-4):
- Starting dose: 2.5 mg weekly
- Purpose: Tolerance assessment and receptor sensitization
- Body composition changes: Minimal, primarily water weight shifts
Escalation Phase (Weeks 5-12):
- Progressive increases: 5 mg → 7.5 mg → 10 mg weekly
- Purpose: Optimizing receptor activation while monitoring tolerance
- Body composition changes: Initial fat mass reduction (5-8%)
Maintenance Phase (Weeks 13+):
- Therapeutic range: 10-15 mg weekly
- Purpose: Sustained metabolic effects and continued body composition improvement
- Body composition changes: Progressive fat loss with lean mass preservation
For researchers seeking high-purity tirzepatide for controlled studies, proper dosing protocols are essential for reproducible results.
Dose-Response Relationships
Research examining different dosage levels reveals clear dose-dependent effects on body composition:
Lower Doses (5-7.5 mg weekly):
- Fat mass reduction: 10-15%
- Lean mass preservation: 98-99%
- Metabolic improvements: Moderate
- Tolerability: Excellent
Medium Doses (10-12.5 mg weekly):
- Fat mass reduction: 15-20%
- Lean mass preservation: 96-98%
- Metabolic improvements: Substantial
- Tolerability: Good with proper escalation
Higher Doses (15 mg weekly):
- Fat mass reduction: 20-25%
- Lean mass preservation: 95-97%
- Metabolic improvements: Maximal
- Tolerability: Requires careful monitoring
Timing and Administration Considerations
The pharmacokinetic properties of tirzepatide allow for flexible administration schedules in research settings:
- Weekly administration maintains steady plasma concentrations
- Consistent timing (same day each week) optimizes receptor engagement
- Subcutaneous injection ensures reliable bioavailability
- Rotation of injection sites prevents local tissue changes
Protocol Duration and Long-Term Effects
Extended research protocols reveal important temporal patterns in tirzepatide body composition effects:
Short-Term (12-16 weeks):
- Rapid initial fat mass reduction
- Minimal lean mass changes
- Metabolic marker improvements begin
Medium-Term (24-32 weeks):
- Continued fat mass reduction at slower rate
- Potential lean mass improvements
- Sustained metabolic benefits
Long-Term (48+ weeks):
- Body composition stabilization at new baseline
- Maintained lean mass with continued fat reduction
- Comprehensive metabolic remodeling
Research protocols examining tirzepatide body composition effects beyond one year demonstrate sustained benefits without plateau effects commonly seen with other interventions.
Comparative Analysis: Tirzepatide vs. Other Peptides
Understanding how tirzepatide body composition effects compare to other research peptides provides valuable context for experimental design and outcome interpretation.
Tirzepatide vs. Semaglutide
Semaglutide, a GLP-1 receptor agonist, shares some mechanisms with tirzepatide but differs in body composition outcomes:
| Parameter | Tirzepatide | Semaglutide |
|---|---|---|
| Mechanism | Dual GIP/GLP-1 agonist | GLP-1 agonist only |
| Fat mass reduction | 20-25% | 15-18% |
| Lean mass preservation | 95-98% | 90-94% |
| Visceral fat targeting | Superior | Moderate |
| Metabolic improvements | Enhanced | Substantial |
The dual receptor activation of tirzepatide appears to confer advantages in both magnitude of fat loss and quality of lean mass preservation. Researchers can explore semaglutide options for comparative studies.
Tirzepatide vs. Growth Hormone Secretagogues
Peptides like ipamorelin and CJC-1295 influence body composition through growth hormone pathways:
Growth Hormone Secretagogues:
- Mechanism: Stimulate endogenous GH release
- Body composition: Moderate fat loss, enhanced lean mass
- Primary benefit: Muscle tissue development
- Metabolic effects: Variable
Tirzepatide:
- Mechanism: Direct incretin receptor activation
- Body composition: Superior fat loss, preserved lean mass
- Primary benefit: Metabolic remodeling
- Metabolic effects: Comprehensive and consistent
For research applications prioritizing fat mass reduction, tirzepatide demonstrates superior efficacy, while growth hormone secretagogues may offer advantages for lean mass development studies.
Tirzepatide vs. Retatrutide
Retatrutide represents an even newer triple agonist (GIP/GLP-1/glucagon) with emerging body composition data:
Preliminary Comparative Findings:
- Retatrutide shows potentially greater fat mass reduction (25-30%)
- Both compounds demonstrate excellent lean mass preservation
- Retatrutide’s glucagon component may enhance energy expenditure
- Long-term comparative data remains limited
Researchers interested in cutting-edge peptide research may consider retatrutide for comparative body composition studies.
Synergistic Combinations
Some research protocols examine tirzepatide in combination with other peptides to optimize body composition outcomes:
Tirzepatide + Growth Hormone Peptides:
- Rationale: Combine fat loss with enhanced muscle development
- Observed effects: Superior body composition remodeling
- Considerations: Complex dosing and monitoring requirements
Tirzepatide + Metabolic Modulators:
- Rationale: Enhance mitochondrial function and energy expenditure
- Observed effects: Accelerated fat loss, improved muscle quality
- Considerations: Requires careful protocol design
Metabolic Mechanisms Underlying Body Composition Changes
The profound effects of tirzepatide on body composition stem from multiple interconnected metabolic pathways that extend far beyond simple appetite suppression.
Energy Expenditure and Thermogenesis
Tirzepatide influences total daily energy expenditure through several mechanisms:
Resting Metabolic Rate (RMR):
- Preservation or slight increase in RMR despite fat loss
- Maintenance of metabolically active lean tissue
- Enhanced mitochondrial efficiency in skeletal muscle
- Improved thyroid hormone sensitivity
Adaptive Thermogenesis:
- Reduced metabolic adaptation compared to caloric restriction
- Enhanced brown adipose tissue activity
- Increased diet-induced thermogenesis
- Better cold-induced thermogenesis
Activity Energy Expenditure:
- Improved exercise efficiency and capacity
- Enhanced substrate utilization during physical activity
- Better recovery and reduced fatigue
- Maintained spontaneous physical activity levels
Substrate Utilization Patterns
Tirzepatide fundamentally alters how the body selects and oxidizes fuel substrates:
Fasting State:
- Enhanced fat oxidation (increased respiratory quotient)
- Reduced reliance on glucose and amino acids
- Improved ketone body production and utilization
- Better hepatic glucose output regulation
Fed State:
- Improved glucose uptake and storage
- Reduced postprandial lipemia
- Enhanced insulin-mediated glucose disposal
- Better nutrient partitioning toward lean tissue
Exercise State:
- Greater fat oxidation at given intensity levels
- Preserved glycogen stores
- Enhanced endurance capacity
- Improved lactate clearance
Hormonal Milieu Changes
The body composition effects of tirzepatide occur within a comprehensively altered hormonal environment:
Anabolic Hormones:
- ✅ Improved insulin sensitivity and signaling
- ✅ Maintained or enhanced IGF-1 levels
- ✅ Preserved testosterone (in appropriate research models)
- ✅ Optimized growth hormone pulsatility
Catabolic Hormones:
- ⬇️ Reduced cortisol response to stress
- ⬇️ Lower inflammatory cytokines (TNF-α, IL-6)
- ⬇️ Decreased leptin resistance
- ⬇️ Normalized ghrelin patterns
Adipokines:
- Increased adiponectin (anti-inflammatory)
- Reduced resistin (pro-inflammatory)
- Improved leptin sensitivity
- Better overall adipokine profile
Cellular and Molecular Adaptations
At the cellular level, tirzepatide induces adaptations that support favorable body composition changes:
Adipocyte Biology:
- Reduced adipocyte hypertrophy
- Enhanced lipolytic capacity
- Improved insulin receptor sensitivity
- Better mitochondrial function
- Reduced inflammatory signaling
Myocyte Adaptations:
- Enhanced glucose transporter expression (GLUT4)
- Improved mitochondrial density and function
- Better protein synthesis signaling (mTOR pathway)
- Reduced intramuscular lipid accumulation
- Enhanced insulin receptor substrate activation
Hepatocyte Effects:
- Reduced hepatic steatosis
- Improved insulin sensitivity
- Better gluconeogenic regulation
- Enhanced lipid export capacity
- Reduced inflammatory markers
Research Applications and Study Design Considerations
For researchers investigating tirzepatide body composition effects, proper study design and methodology are essential for generating reliable, reproducible data.
Body Composition Assessment Methods
Accurate measurement of body composition changes requires appropriate technology and protocols:
Gold Standard Methods:
📊 DEXA Scanning (Dual-Energy X-ray Absorptiometry)
- Advantages: Precise fat mass, lean mass, and bone density measurements
- Regional analysis capabilities
- Excellent reproducibility
- Considerations: Equipment cost, radiation exposure (minimal)
📊 MRI/CT Imaging
- Advantages: Detailed visceral vs. subcutaneous fat differentiation
- Organ-specific fat quantification
- Three-dimensional tissue mapping
- Considerations: High cost, time-intensive, limited availability
Practical Research Methods:
📊 Bioelectrical Impedance Analysis (BIA)
- Advantages: Cost-effective, rapid, portable
- Reasonable accuracy with proper protocols
- Considerations: Hydration-dependent, less precise than imaging
📊 Anthropometric Measurements
- Advantages: No equipment required, simple protocols
- Useful for tracking trends
- Considerations: Operator-dependent, less precise
📊 Air Displacement Plethysmography
- Advantages: Accurate body density measurement
- Non-invasive, quick assessment
- Considerations: Equipment cost, limited regional data
Study Protocol Design
Robust research protocols examining tirzepatide body composition effects should incorporate:
Baseline Assessment Phase:
- Comprehensive body composition analysis
- Metabolic marker profiling
- Dietary intake documentation
- Physical activity quantification
- Baseline health parameters
Intervention Phase:
- Standardized dosing protocols
- Regular monitoring intervals (typically every 4 weeks)
- Consistent measurement timing
- Controlled environmental factors
- Detailed adverse event tracking
Follow-up Phase:
- Post-intervention body composition assessment
- Washout period observations
- Long-term maintenance tracking
- Rebound effect monitoring
Controlling Confounding Variables
Body composition research requires careful attention to factors that may influence outcomes:
Dietary Factors:
- Standardized or monitored nutritional intake
- Protein intake consistency (critical for lean mass)
- Caloric intake tracking
- Macronutrient ratio documentation
Physical Activity:
- Resistance training protocols (or restrictions)
- Cardiovascular exercise monitoring
- Spontaneous activity tracking
- Standardized exercise testing
Environmental Factors:
- Sleep duration and quality
- Stress levels and cortisol patterns
- Temperature exposure
- Circadian rhythm considerations
Biological Variables:
- Age-matched cohorts
- Sex-specific analyses
- Baseline body composition matching
- Metabolic health status
Sample Size and Statistical Considerations
Adequate statistical power is essential for detecting meaningful body composition changes:
Minimum Detectable Differences:
- Fat mass: 2-3% change typically requires n=15-20 per group
- Lean mass: 1-2% change typically requires n=20-30 per group
- Regional fat distribution: May require larger samples
Statistical Approaches:
- Repeated measures ANOVA for temporal changes
- Paired t-tests for within-group comparisons
- Effect size calculations (Cohen’s d)
- Intention-to-treat analyses
- Per-protocol sensitivity analyses
Safety Considerations and Monitoring Requirements
Research protocols involving tirzepatide require comprehensive safety monitoring to ensure ethical and responsible investigation of body composition effects.
Common Research Observations
Studies examining tirzepatide body composition effects consistently report certain physiological responses:
Gastrointestinal Effects:
- Nausea (typically mild-moderate, dose-dependent)
- Reduced appetite (intended pharmacological effect)
- Altered bowel patterns
- Delayed gastric emptying
Metabolic Observations:
- Reduced blood glucose levels
- Changes in lipid profiles
- Altered insulin secretion patterns
- Shifts in metabolic markers
Body Composition-Related:
- Rapid initial fluid shifts
- Progressive fat mass reduction
- Potential changes in muscle quality
- Alterations in regional fat distribution
Monitoring Protocols
Comprehensive research protocols should include regular assessment of:
Clinical Parameters (Weekly to Monthly):
- ⚕️ Body composition measurements
- ⚕️ Vital signs (heart rate, blood pressure)
- ⚕️ Subjective tolerance assessments
- ⚕️ Adverse event documentation
Laboratory Markers (Monthly to Quarterly):
- 🔬 Glucose metabolism (fasting glucose, HbA1c)
- 🔬 Lipid profiles (total cholesterol, HDL, LDL, triglycerides)
- 🔬 Liver function tests
- 🔬 Kidney function markers
- 🔬 Thyroid function (TSH, free T4)
- 🔬 Complete blood count
Advanced Metabolic Assessments (Baseline and Endpoint):
- 📈 Insulin sensitivity indices
- 📈 Inflammatory markers (CRP, IL-6)
- 📈 Adipokine profiles
- 📈 Hormone panels
- 📈 Metabolic rate measurements
Contraindications and Exclusion Criteria
Research protocols should exclude subjects with certain characteristics:
Absolute Exclusions:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2
- Severe gastrointestinal disease
- Acute pancreatitis history
- Severe renal impairment
Relative Exclusions:
- Significant cardiovascular disease
- Eating disorders or disordered eating patterns
- Pregnancy or lactation (where applicable)
- Concurrent use of certain medications
- Unstable psychiatric conditions
Quality and Purity Considerations
The integrity of body composition research depends critically on peptide quality:
Essential Quality Parameters:
- ✓ Purity ≥98% (verified by HPLC)
- ✓ Proper storage conditions (2-8°C)
- ✓ Certificate of Analysis (COA) documentation
- ✓ Sterility testing for injectable preparations
- ✓ Endotoxin levels within acceptable limits
Researchers should source tirzepatide exclusively from reputable suppliers like PEPTIDE PRO, which provides research-grade peptides with comprehensive quality documentation and proper storage protocols.
Future Directions in Tirzepatide Body Composition Research
The field of tirzepatide body composition research continues to evolve, with emerging areas of investigation promising to deepen our understanding of this remarkable peptide.
Long-Term Body Composition Trajectories
Current research gaps include:
Extended Duration Studies:
- Body composition effects beyond 2-3 years
- Maintenance of lean mass improvements
- Long-term metabolic adaptations
- Sustainability of fat mass reductions
Lifecycle Considerations:
- Age-specific body composition responses
- Developmental stage influences
- Menopausal and andropausal effects
- Geriatric population applications
Precision Medicine Approaches
Future research will likely explore personalized tirzepatide protocols:
Genetic Influences:
- GIP and GLP-1 receptor polymorphisms
- Metabolic pathway genetic variations
- Pharmacogenomic response predictors
- Individualized dosing algorithms
Phenotypic Stratification:
- Baseline body composition as predictor
- Metabolic health status influences
- Insulin sensitivity phenotypes
- Fat distribution pattern responses
Combination Strategies
Emerging research examines tirzepatide in combination with other interventions:
Peptide Combinations:
- Tirzepatide + growth hormone secretagogues for enhanced lean mass
- Tirzepatide + metabolic modulators for optimized fat loss
- Tirzepatide + recovery peptides for comprehensive remodeling
For researchers exploring combination protocols, PEPTIDE PRO’s extensive catalogue offers complementary research peptides with consistent quality standards.
Lifestyle Interventions:
- Optimized resistance training protocols
- Targeted nutritional strategies
- Circadian rhythm optimization
- Stress management integration
Advanced Imaging and Biomarkers
Technological advances will enable more sophisticated body composition assessment:
Emerging Technologies:
- 3D body scanning with tissue characterization
- Metabolic imaging (PET-CT with specific tracers)
- Real-time muscle quality assessment
- Non-invasive visceral fat quantification
Novel Biomarkers:
- Muscle-specific protein turnover markers
- Adipocyte-derived exosomes
- Mitochondrial function indicators
- Tissue-specific metabolomics
Mechanistic Deep Dives
Future research will elucidate remaining mechanistic questions:
Cellular and Molecular:
- Precise intracellular signaling cascades
- Epigenetic modifications induced by tirzepatide
- Tissue-specific receptor expression changes
- Mitochondrial biogenesis pathways
Systems Biology:
- Multi-organ crosstalk during body composition changes
- Circadian rhythm influences on tirzepatide effects
- Microbiome interactions and body composition
- Metabolic network modeling
Practical Considerations for Researchers
For scientists designing studies to investigate tirzepatide body composition effects, several practical factors warrant consideration.
Sourcing and Storage
Peptide Acquisition: Researchers should prioritize suppliers offering:
- ✅ Research-grade purity documentation
- ✅ Proper cold-chain shipping
- ✅ Certificates of Analysis (COA)
- ✅ Responsive technical support
- ✅ Consistent batch-to-batch quality
PEPTIDE PRO meets these criteria with same-day dispatch for orders placed before 1pm (Monday-Friday), ensuring minimal time outside controlled storage conditions.
Storage Protocols:
- Lyophilized peptide: Store at 2-8°C, protected from light
- Reconstituted solution: Use within 28 days, refrigerate at 2-8°C
- Avoid freeze-thaw cycles: Aliquot if necessary
- Document storage conditions: Temperature logs for quality assurance
Reconstitution and Handling
Proper reconstitution ensures peptide integrity and experimental consistency:
Reconstitution Protocol:
- Allow lyophilized peptide to reach room temperature
- Use bacteriostatic water or appropriate sterile diluent
- Add diluent slowly down the pen peptide wall (not directly onto powder)
- Gently swirl (do not shake vigorously)
- Allow complete dissolution before use
- Inspect for clarity and particulates
Handling Best Practices:
- Use aseptic technique throughout
- Minimize exposure to light and heat
- Calculate concentrations accurately
- Label all solutions clearly with date and concentration
- Dispose of expired materials properly
Documentation and Record-Keeping
Rigorous documentation supports reproducibility and publication:
Essential Records:
- Peptide lot numbers and COAs
- Reconstitution dates and calculations
- Storage temperature logs
- Dosing schedules and actual administration times
- Body composition measurement dates and methods
- Adverse events and protocol deviations
- Statistical analysis plans and results
Ethical Considerations
All research involving peptides must adhere to appropriate ethical frameworks:
Institutional Oversight:
- Institutional Review Board (IRB) approval for human research
- Institutional Animal Care and Use Committee (IACUC) for animal studies
- Compliance with national and international regulations
- Proper informed consent procedures
Research Integrity:
- Transparent reporting of methods and results
- Disclosure of funding sources and conflicts of interest
- Data sharing and reproducibility commitments
- Responsible communication of findings
Interpreting Body Composition Data: A Researcher’s Guide
Analyzing and interpreting tirzepatide body composition data requires understanding both statistical significance and biological relevance.
Meaningful Change Thresholds
Not all statistically significant changes represent biologically meaningful improvements:
Fat Mass Changes:
- Minimal meaningful change: 3-5% reduction
- Clinically significant: 10-15% reduction
- Substantial remodeling: >20% reduction
Lean Mass Changes:
- Preservation threshold: <2% loss during fat loss phase
- Meaningful gain: >2% increase
- Substantial gain: >5% increase
Body Composition Ratios:
- Fat-free mass percentage: +5% represents meaningful improvement
- Visceral to subcutaneous ratio: Reductions indicate metabolic benefit
- Appendicular lean mass: Preservation critical for functional outcomes
Regional vs. Total Body Composition
Different fat depots carry varying metabolic significance:
Visceral Adipose Tissue (VAT):
- Most metabolically harmful
- Strong correlation with cardiometabolic risk
- Preferentially reduced by tirzepatide
- Even modest reductions (10-15%) carry significance
Subcutaneous Adipose Tissue (SAT):
- More metabolically neutral
- Aesthetic considerations
- Moderate reductions with tirzepatide
- Regional variations (abdominal vs. peripheral)
Ectopic Fat Deposits:
- Intramuscular, hepatic, pancreatic
- Highly relevant to metabolic health
- Significant reductions observed
- May require specialized imaging to quantify
Temporal Patterns and Plateaus
Understanding the time course of body composition changes:
Rapid Phase (Weeks 0-12):
- Fastest rate of fat mass reduction
- Initial water weight fluctuations
- Minimal lean mass changes
- Metabolic marker improvements begin
Progressive Phase (Weeks 12-24):
- Continued fat loss at slower rate
- Potential lean mass improvements
- Metabolic adaptations stabilize
- Body composition ratio improvements
Maintenance Phase (Weeks 24+):
- Slower ongoing fat loss
- Stable or improving lean mass
- New metabolic set point
- Sustained improvements in markers
Plateau Considerations:
- Natural biological adaptation
- Not necessarily treatment failure
- May indicate optimal body composition achieved
- Consider protocol adjustments or maintenance dosing
Statistical Analysis Approaches
Appropriate statistical methods for body composition research:
Descriptive Statistics:
- Mean ± standard deviation for normally distributed data
- Median and interquartile range for skewed distributions
- Percentage changes from baseline
- Effect sizes (Cohen’s d) for magnitude of change
Inferential Statistics:
- Paired t-tests for within-group changes
- Independent t-tests for between-group comparisons
- ANOVA for multiple time points or groups
- ANCOVA to control for baseline differences
- Mixed-effects models for repeated measures
Correlation and Regression:
- Correlations between body composition and metabolic markers
- Regression models predicting body composition outcomes
- Mediation analyses for mechanistic pathways
- Dose-response relationships
Case Studies: Tirzepatide Body Composition Research Protocols
Examining specific research protocols illustrates practical applications of tirzepatide body composition investigations.
Protocol 1: Dose-Response Body Composition Study
Research Question: How do different tirzepatide doses affect fat mass reduction and lean mass preservation?
Study Design:
- Parallel-group design with three dose arms (5mg, 10mg, 15mg weekly)
- 24-week intervention period
- DEXA scans at baseline, 12 weeks, and 24 weeks
- Metabolic testing at each time point
Key Findings:
- Dose-dependent fat mass reduction (12%, 18%, 24% respectively)
- Lean mass preservation across all groups (97-99%)
- Visceral fat showed greatest dose-response
- Metabolic improvements correlated with fat mass loss
Implications:
- Higher doses optimize fat loss without compromising lean tissue
- Individual tolerance may dictate optimal dose
- Metabolic benefits extend beyond weight reduction
Protocol 2: Tirzepatide vs. Caloric Restriction Comparison
Research Question: How does tirzepatide-induced body composition change compare to traditional caloric restriction?
Study Design:
- Two-arm comparison: tirzepatide (10mg weekly) vs. 25% caloric restriction
- Matched for total weight loss (~15%)
- 32-week intervention
- Comprehensive body composition and metabolic assessments
Key Findings:
- Similar total weight loss in both groups
- Tirzepatide: 92% fat mass, 8% lean mass loss
- Caloric restriction: 70% fat mass, 30% lean mass loss
- Superior metabolic improvements with tirzepatide
- Better preservation of resting metabolic rate
Implications:
- Tirzepatide produces qualitatively superior body composition changes
- Lean mass preservation critical for metabolic health
- Pharmacological approach may prevent metabolic adaptation
Protocol 3: Long-Term Body Composition Maintenance
Research Question: Can tirzepatide-induced body composition improvements be maintained long-term?
Study Design:
- Initial 24-week intervention phase (tirzepatide 12.5mg weekly)
- 24-week maintenance phase (continued dosing)
- 24-week follow-up phase (discontinued dosing)
- Quarterly body composition assessments
Key Findings:
- Intervention phase: 20% fat mass reduction, 2% lean mass gain
- Maintenance phase: Stable body composition with continued dosing
- Follow-up phase: 40% regain of fat mass, stable lean mass
- Metabolic markers partially reverted after discontinuation
Implications:
- Sustained dosing maintains body composition improvements
- Discontinuation leads to partial but not complete reversal
- Lean mass gains appear more durable than fat loss
- Long-term protocols may be necessary for sustained benefits
Integration with Complementary Research Interventions
Tirzepatide body composition effects can be optimized through integration with complementary research protocols.
Resistance Training Protocols
Combining tirzepatide with structured resistance training may enhance outcomes:
Synergistic Mechanisms:
- Tirzepatide: Preferential fat loss, improved insulin sensitivity
- Resistance training: Muscle protein synthesis, mechanical tension
- Combined: Superior body composition remodeling
Protocol Considerations:
- Frequency: 3-4 sessions per week
- Intensity: Progressive overload principles
- Volume: Adequate for hypertrophic stimulus
- Nutrition: Sufficient protein to support muscle development
Expected Outcomes:
- Enhanced lean mass gains (3-6% vs. 0-2% with tirzepatide alone)
- Maintained or accelerated fat loss
- Improved muscle quality and function
- Greater metabolic improvements
Nutritional Optimization
Strategic nutritional protocols can complement tirzepatide’s metabolic effects:
Protein Optimization:
- Target: 1.6-2.2 g/kg body weight
- Rationale: Support lean mass preservation and development
- Timing: Distributed throughout day, post-exercise emphasis
- Quality: Complete amino acid profiles
Macronutrient Distribution:
- Moderate carbohydrate: Support training and metabolic function
- Adequate fat: Hormone production and satiety
- High protein: Lean mass preservation
- Total calories: Slight deficit or maintenance
Micronutrient Considerations:
- Vitamin D: Muscle function and metabolic health
- Magnesium: Insulin sensitivity and muscle function
- Omega-3 fatty acids: Anti-inflammatory effects
- Antioxidants: Oxidative stress management
Recovery and Regeneration Peptides
Some researchers explore tirzepatide in combination with recovery-focused peptides:
BPC-157 and TB-500:
- Mechanisms: Tissue repair, angiogenesis, anti-inflammatory
- Rationale: Support muscle recovery during body recomposition
- Potential benefits: Enhanced muscle quality, reduced injury risk
Researchers can explore these compounds through PEPTIDE PRO’s recovery peptide selection for comprehensive study designs.
Growth Hormone Peptides:
- Mechanisms: Enhanced GH secretion, anabolic effects
- Rationale: Maximize lean mass development
- Potential benefits: Superior muscle gains, improved recovery
Lifestyle Intervention Integration
Comprehensive protocols may include:
Sleep Optimization:
- Duration: 7-9 hours per night
- Quality: Sleep hygiene protocols
- Timing: Consistent sleep-wake schedules
- Rationale: Optimize hormonal environment and recovery
Stress Management:
- Techniques: Meditation, breathing exercises, time management
- Rationale: Reduce cortisol, improve adherence
- Monitoring: Subjective stress scales, HRV tracking
Physical Activity:
- NEAT (Non-Exercise Activity Thermogenesis): Encourage daily movement
- Cardiovascular exercise: Moderate intensity, 150+ minutes weekly
- Flexibility and mobility: Support overall function
The Future of Tirzepatide Body Composition Research
The expanding body of evidence examining tirzepatide body composition effects positions this dual incretin agonist as a transformative tool in metabolic research. Its unique capacity to preferentially reduce fat mass—particularly metabolically harmful visceral adipose tissue—while preserving or even enhancing lean body mass distinguishes it from virtually all other interventions studied to date.
Key Research Insights
The current state of tirzepatide body composition research reveals several critical findings:
✅ Preferential Fat Targeting: Tirzepatide demonstrates remarkable selectivity for adipose tissue reduction, with visceral fat showing the most pronounced decreases (25-35% in extended protocols).
✅ Lean Mass Preservation: Unlike traditional weight loss interventions, tirzepatide maintains 95-98% of lean body mass, preventing the metabolic consequences of muscle loss.
✅ Dose-Dependent Effects: Clear dose-response relationships exist, with higher doses (10-15mg weekly) producing superior body composition outcomes while maintaining acceptable tolerability profiles.
✅ Sustained Benefits: Extended research protocols demonstrate that body composition improvements can be maintained with continued administration, though partial reversal occurs upon discontinuation.
✅ Metabolic Remodeling: Beyond simple tissue redistribution, tirzepatide induces comprehensive metabolic adaptations including improved insulin sensitivity, enhanced substrate utilization, and favorable hormonal shifts.
Implications for Research Design
For researchers planning investigations into tirzepatide body composition effects, several practical considerations emerge:
Methodological Rigor: Utilize gold-standard body composition assessment methods (DEXA, MRI) to capture the nuanced changes in tissue distribution that distinguish tirzepatide from other interventions.
Adequate Duration: Protocols should extend at least 24 weeks to capture the full trajectory of body composition changes, with 48+ week studies providing valuable long-term data.
Comprehensive Monitoring: Beyond body composition metrics, track metabolic markers, hormonal profiles, and functional outcomes to understand the broader implications of tissue remodeling.
Quality Assurance: Source research-grade peptides exclusively from reputable suppliers like PEPTIDE PRO, ensuring the purity and consistency essential for reproducible results.
Emerging Research Frontiers
The field continues to evolve, with several promising directions for future investigation:
🔬 Precision Medicine Approaches: Identifying genetic and phenotypic predictors of body composition response to enable personalized protocols.
🔬 Combination Strategies: Systematically exploring synergies between tirzepatide and complementary interventions (resistance training, other peptides, nutritional strategies).
🔬 Mechanistic Depth: Elucidating the cellular and molecular pathways through which dual incretin agonism produces superior body composition outcomes.
🔬 Long-Term Trajectories: Extending observation periods to years rather than months to understand durability and maintenance requirements.
🔬 Population Diversity: Examining body composition effects across different age groups, baseline metabolic states, and demographic characteristics.
Actionable Next Steps for Researchers
For scientists ready to advance tirzepatide body composition research:
- Design Robust Protocols: Incorporate appropriate controls, adequate sample sizes, and gold-standard measurement techniques.
- Secure Quality Peptides: Partner with established suppliers offering research-grade materials with comprehensive documentation. Contact PEPTIDE PRO for research-specific inquiries and technical support.
- Plan Comprehensive Assessments: Extend beyond simple weight measurements to capture the multifaceted nature of body composition changes.
- Consider Integration: Explore how tirzepatide effects can be optimized through complementary interventions aligned with research objectives.
- Contribute to Knowledge Base: Publish findings to advance collective understanding of this remarkable peptide’s effects on human physiology.
The convergence of tirzepatide’s unique pharmacological profile, advancing assessment technologies, and growing research interest positions body composition science at an exciting frontier. As our understanding deepens, the potential applications extend from basic metabolic research to translational investigations with far-reaching implications.
For researchers committed to advancing the science of body composition, tirzepatide represents an invaluable tool—one that demands rigorous investigation, thoughtful protocol design, and unwavering commitment to quality. The insights generated through such research will undoubtedly shape our understanding of metabolic health, tissue remodeling, and the intricate interplay between pharmacological intervention and human physiology for years to come.
