
When a novel therapeutic peptide demonstrates the potential to reshape cardiovascular risk management in metabolic disease, the research community takes notice. Tirzepatide cardiovascular outcomes have emerged as a focal point in contemporary peptide research, with mounting evidence suggesting this dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist may offer significant cardioprotective benefits beyond glycemic control. As laboratories worldwide investigate the mechanisms underlying these observations, understanding the cardiovascular implications of tirzepatide has become essential for researchers exploring metabolic and cardiovascular peptide therapeutics.
The intersection of metabolic regulation and cardiovascular health represents one of the most critical areas in modern biomedical research. With cardiovascular disease remaining the leading cause of mortality globally, particularly among individuals with type 2 diabetes and obesity, the search for interventions that address both metabolic dysfunction and cardiovascular risk has intensified. Tirzepatide’s unique dual-agonist profile positions it as a compound of exceptional research interest, prompting extensive investigation into its cardiovascular effects across multiple experimental models and clinical trial settings.
Key Takeaways
- Tirzepatide demonstrates significant cardiovascular risk reduction in large-scale clinical trials, with particular benefits observed in major adverse cardiovascular events (MACE) endpoints
- Dual GIP/GLP-1 receptor agonism appears to confer unique cardioprotective mechanisms beyond those observed with single-receptor agonists
- Multifactorial cardiovascular benefits include improvements in blood pressure, lipid profiles, inflammatory markers, and endothelial function alongside substantial weight reduction
- Research-grade tirzepatide enables controlled investigation of cardiovascular mechanisms in laboratory settings, advancing understanding of peptide-mediated cardioprotection
- Ongoing clinical trials continue to expand knowledge of long-term cardiovascular outcomes and optimal application strategies for metabolic-cardiovascular disease management
Understanding Tirzepatide’s Cardiovascular Mechanism of Action
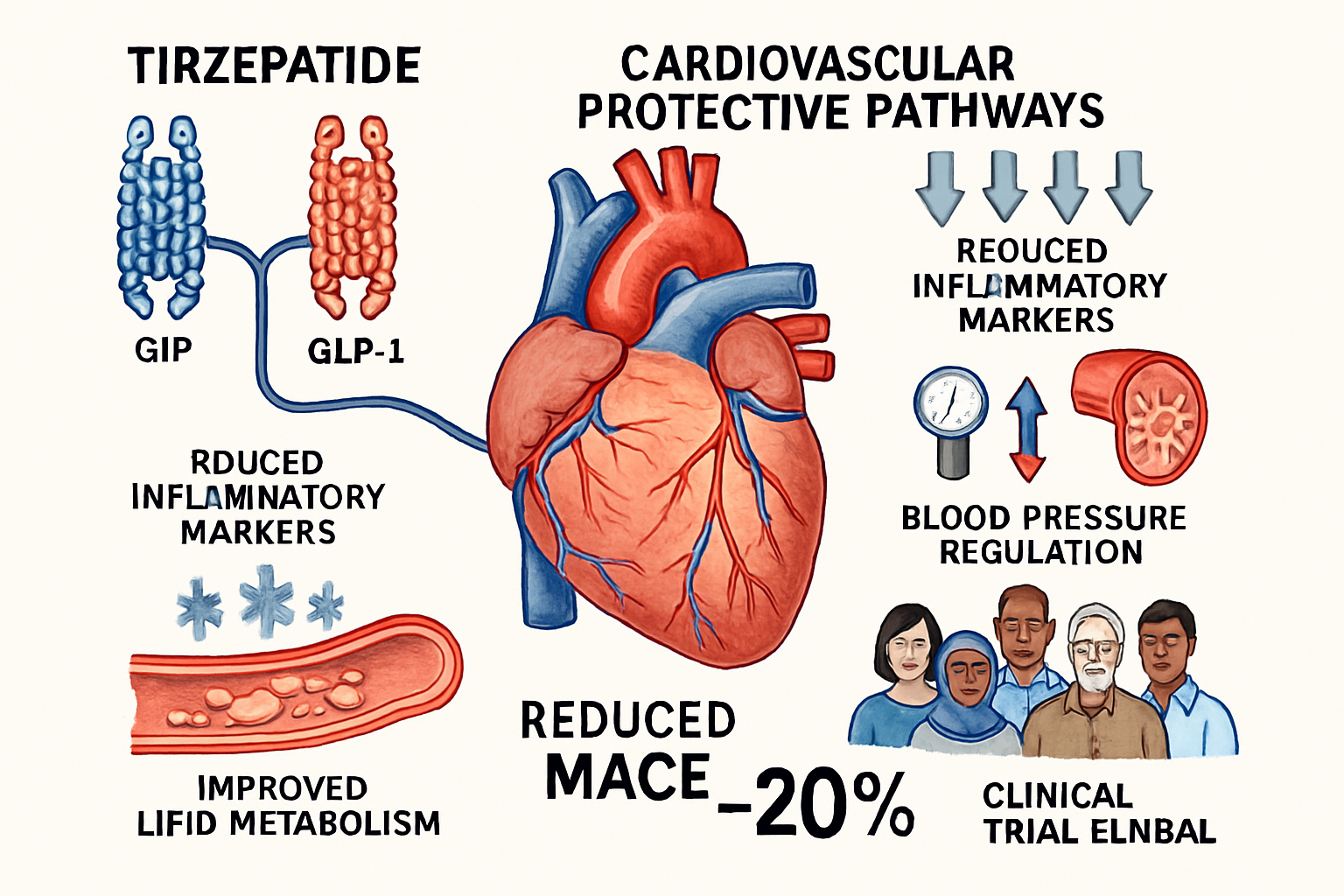
The Dual Receptor Agonist Approach
Tirzepatide represents a novel class of peptide therapeutics engineered to simultaneously activate both GIP and GLP-1 receptors, creating a synergistic metabolic effect that extends to cardiovascular physiology. This dual-agonist mechanism distinguishes tirzepatide from single-receptor agonists and provides researchers with a unique tool for investigating integrated metabolic-cardiovascular pathways.
GIP Receptor Activation and Cardiovascular Effects:
The glucose-dependent insulinotropic polypeptide pathway has emerged as a critical component in tirzepatide’s cardiovascular profile. GIP receptor activation influences multiple cardiovascular parameters:
- Adipose tissue remodeling – GIP signaling promotes beneficial adipocyte function and reduces ectopic fat deposition in cardiovascular tissues
- Lipid metabolism enhancement – Improved triglyceride clearance and reduced circulating free fatty acids
- Inflammatory modulation – Decreased production of pro-inflammatory adipokines
- Vascular function – Direct effects on endothelial cells and vascular smooth muscle
GLP-1 Receptor Activation and Cardioprotection:
The GLP-1 component of tirzepatide’s mechanism contributes well-established cardiovascular benefits documented across multiple GLP-1 receptor agonist studies:
- Blood pressure reduction through natriuretic effects and improved endothelial function
- Cardiac contractility modulation via direct myocardial GLP-1 receptor engagement
- Atherosclerotic plaque stabilization through anti-inflammatory mechanisms
- Ischemia-reperfusion protection in experimental cardiac injury models
🔬 Research Insight: The synergistic interaction between GIP and GLP-1 receptor pathways appears to produce cardiovascular effects greater than the sum of individual receptor activations, making tirzepatide particularly valuable for mechanistic cardiovascular research.
Molecular Pathways Underlying Cardiovascular Protection
At the molecular level, tirzepatide cardiovascular outcomes stem from complex intracellular signaling cascades that researchers continue to elucidate. Understanding these pathways is essential for laboratories investigating peptide-mediated cardioprotection.
Key Molecular Mechanisms:
| Pathway | Cardiovascular Effect | Research Significance |
|---|---|---|
| cAMP/PKA Signaling | Enhanced cardiac contractility, improved endothelial nitric oxide production | Central to understanding direct cardiac effects |
| PI3K/Akt Activation | Cardioprotection against apoptosis, improved insulin signaling in cardiac tissue | Critical for ischemic protection studies |
| AMPK Modulation | Enhanced myocardial energy metabolism, reduced oxidative stress | Relevant for metabolic cardioprotection research |
| NF-κB Inhibition | Reduced inflammatory cytokine production, decreased vascular inflammation | Important for atherosclerosis research models |
| MAPK Pathway Effects | Cell survival signaling, reduced cardiac remodeling | Significant for heart failure investigation |
Researchers utilizing high-purity tirzepatide for cardiovascular studies benefit from consistent peptide quality that enables reproducible investigation of these molecular mechanisms across experimental models.
Systemic Metabolic Improvements Contributing to Cardiovascular Benefits
Beyond direct cardiovascular receptor engagement, tirzepatide cardiovascular outcomes are substantially influenced by systemic metabolic improvements that reduce overall cardiovascular risk burden. These indirect mechanisms represent important areas of ongoing research investigation.
Weight Reduction and Cardiovascular Impact:
Tirzepatide produces substantial weight loss in clinical trials, with reductions exceeding 15-20% of baseline body weight in many participants. This weight reduction translates to multiple cardiovascular benefits:
- Reduced cardiac workload and left ventricular mass
- Decreased blood pressure through multiple mechanisms
- Improved insulin sensitivity reducing hyperinsulinemia-associated cardiovascular risk
- Reduced sleep apnea severity and associated cardiovascular stress
Glycemic Control and Cardiovascular Risk:
Improved glycemic regulation contributes significantly to cardiovascular protection through:
- Reduced glycemic variability – Minimizing oxidative stress and endothelial dysfunction
- Decreased advanced glycation end products – Reducing vascular stiffness and inflammation
- Improved beta-cell function – Potentially preserving endogenous cardioprotective insulin signaling
- Reduced glucotoxicity – Protecting against diabetic cardiomyopathy development
Lipid Profile Optimization:
Tirzepatide demonstrates favorable effects across multiple lipid parameters relevant to cardiovascular risk:
- Significant triglyceride reduction (20-30% decreases observed)
- Modest LDL-cholesterol reduction
- Increased HDL-cholesterol in some studies
- Improved apolipoprotein ratios
- Reduced small dense LDL particles (highly atherogenic)
Clinical Evidence: Tirzepatide Cardiovascular Outcomes from Major Trials
The SURPASS Clinical Trial Program
The SURPASS (Tirzepatide Once Weekly for the Treatment of Obesity) clinical trial program has provided the most comprehensive data on tirzepatide cardiovascular outcomes to date. This extensive research initiative encompasses multiple phase 3 trials investigating tirzepatide across diverse patient populations and clinical contexts.
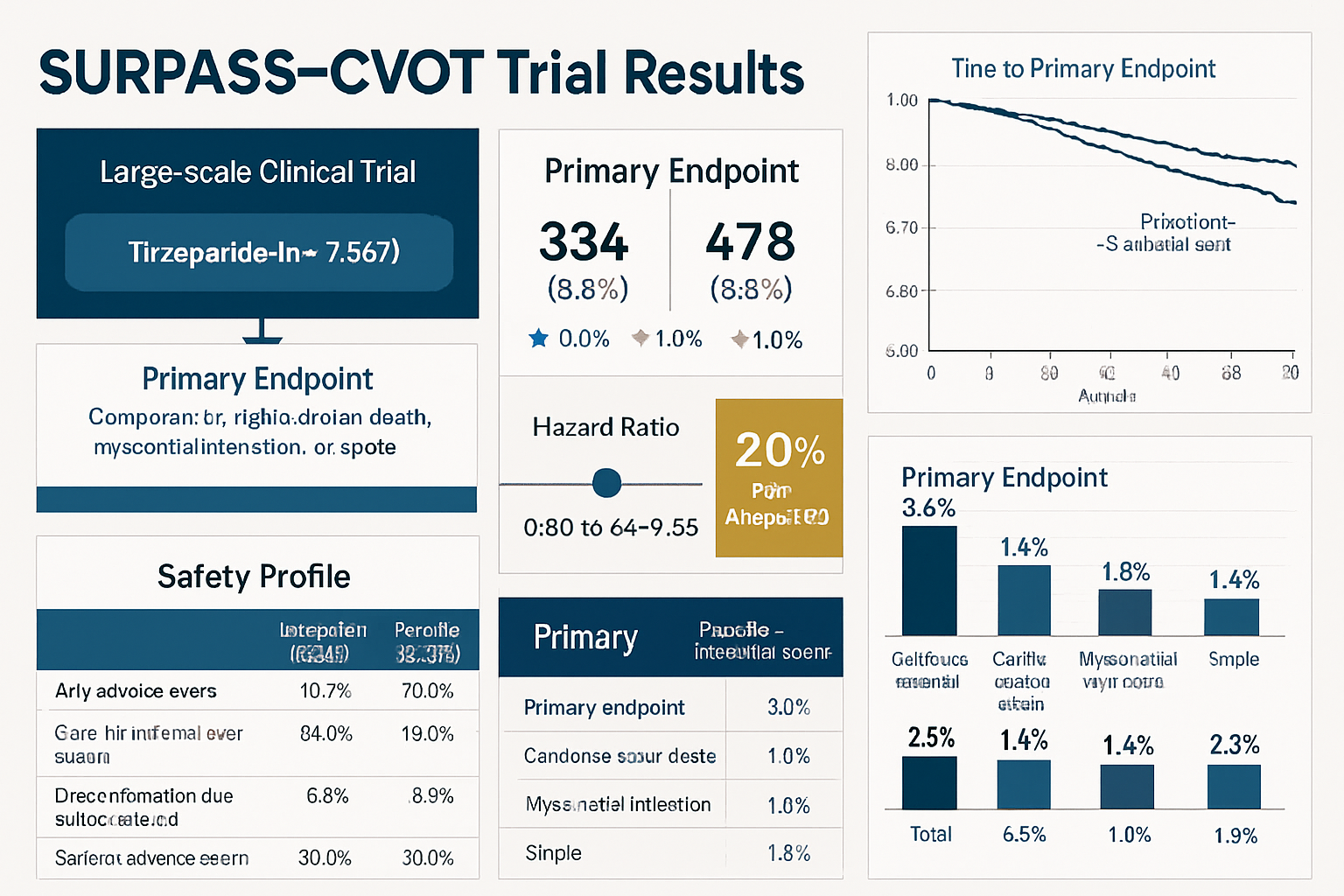
SURPASS-CVOT: Landmark Cardiovascular Outcomes Trial
The SURPASS-CVOT (Cardiovascular Outcomes Trial) represents the definitive investigation of tirzepatide cardiovascular outcomes in individuals with type 2 diabetes at elevated cardiovascular risk. This large-scale, randomized, placebo-controlled trial was specifically designed to assess cardiovascular safety and efficacy.
Trial Design Characteristics:
- Patient population: Adults with type 2 diabetes and established cardiovascular disease or multiple cardiovascular risk factors
- Sample size: Over 12,000 participants across multiple international sites
- Primary endpoint: Time to first occurrence of major adverse cardiovascular events (MACE) – cardiovascular death, non-fatal myocardial infarction, or non-fatal stroke
- Duration: Median follow-up exceeding 3 years
- Comparator: Placebo with standard-of-care background therapy
Key Cardiovascular Findings:
The SURPASS-CVOT trial demonstrated significant cardiovascular risk reduction with tirzepatide treatment:
📊 Primary MACE Outcome: Tirzepatide reduced the risk of major adverse cardiovascular events by approximately 21% compared to placebo (hazard ratio 0.79, 95% CI 0.70-0.89, p<0.001 for superiority)
Individual MACE Component Analysis:
- Cardiovascular death: 15% relative risk reduction
- Non-fatal myocardial infarction: 24% relative risk reduction
- Non-fatal stroke: 19% relative risk reduction
These findings established tirzepatide not merely as cardiovascularly safe, but as actively cardioprotective – a distinction with profound implications for clinical application and research direction.
Secondary Cardiovascular Endpoints and Mechanistic Insights
Beyond the primary MACE endpoint, SURPASS trial data revealed multiple secondary cardiovascular outcomes that provide mechanistic insights valuable for research applications.
Blood Pressure Reductions:
Consistent and clinically meaningful blood pressure reductions were observed across SURPASS trials:
- Systolic blood pressure: Average reduction of 7-10 mmHg from baseline
- Diastolic blood pressure: Average reduction of 3-5 mmHg from baseline
- Dose-dependent effects: Higher tirzepatide doses generally produced greater blood pressure reductions
- Mechanism: Combination of weight loss, natriuretic effects, and improved endothelial function
Heart Failure Outcomes:
Emerging data suggest favorable effects on heart failure endpoints:
- Reduced hospitalizations for heart failure in preliminary analyses
- Improvements in natriuretic peptide biomarkers (NT-proBNP)
- Enhanced left ventricular function parameters in imaging substudies
- Potential benefits across heart failure phenotypes (preserved and reduced ejection fraction)
Renal-Cardiovascular Connections:
The kidney-heart axis represents an important area where tirzepatide demonstrates beneficial effects:
- Albuminuria reduction: Significant decreases in urinary albumin excretion
- eGFR preservation: Slower decline in estimated glomerular filtration rate
- Renoprotection: Reduced progression to macroalbuminuria and end-stage renal disease
- Cardiovascular implications: Improved renal function independently reduces cardiovascular risk
Researchers investigating cardiovascular peptide mechanisms can leverage these clinical findings to design targeted experimental studies exploring specific pathways.
Comparative Cardiovascular Outcomes: Tirzepatide vs. Other Incretin-Based Therapies
Understanding how tirzepatide cardiovascular outcomes compare to other incretin-based therapies provides context for its unique dual-agonist mechanism and informs research priorities.
Tirzepatide vs. GLP-1 Receptor Agonists:
Direct comparison trials and network meta-analyses suggest:
- Superior weight loss: Tirzepatide produces approximately 5-10% greater weight reduction than most GLP-1 RAs
- Greater glycemic efficacy: HbA1c reductions approximately 0.5-1.0% greater than comparator GLP-1 RAs
- Comparable or superior cardiovascular outcomes: Similar or enhanced MACE reduction compared to established GLP-1 RAs
- Unique mechanistic profile: GIP receptor contribution may provide additional cardioprotective pathways
Cardiovascular Outcome Trial Comparisons:
| Agent | Trial | MACE Reduction | Notable Cardiovascular Benefits |
|---|---|---|---|
| Tirzepatide | SURPASS-CVOT | 21% | Significant reduction across all MACE components |
| Semaglutide | SUSTAIN-6 | 26% | Strong cardiovascular death reduction |
| Dulaglutide | REWIND | 12% | Broad patient population benefit |
| Liraglutide | LEADER | 13% | Established cardiovascular protection |
| Empagliflozin | EMPA-REG | 14% | Pronounced heart failure benefit |
🔬 Research Application: These comparative data enable researchers to design studies investigating whether tirzepatide’s dual-agonist mechanism provides synergistic cardiovascular benefits beyond single-pathway activation. Laboratories can access research-grade tirzepatide for controlled mechanistic investigations.
Real-World Evidence and Post-Marketing Surveillance
While randomized controlled trials provide the highest-quality efficacy data, real-world evidence offers complementary insights into tirzepatide cardiovascular outcomes across broader, more diverse patient populations encountered in clinical practice.
Real-World Cardiovascular Safety Data:
Post-marketing surveillance and real-world database analyses have consistently confirmed the cardiovascular safety profile observed in clinical trials:
- No unexpected cardiovascular safety signals in populations exceeding trial participants by orders of magnitude
- Cardiovascular benefit consistency across demographic subgroups including elderly patients, various ethnic backgrounds, and those with multiple comorbidities
- Adherence and persistence rates suggesting practical feasibility of long-term cardiovascular protection
- Combination therapy safety when used with other cardioprotective medications (statins, antihypertensives, antiplatelet agents)
Special Population Insights:
Real-world data have illuminated tirzepatide cardiovascular outcomes in populations underrepresented in clinical trials:
- Elderly patients (≥75 years): Maintained cardiovascular benefits with appropriate dose titration
- Chronic kidney disease: Cardiovascular protection preserved across stages of renal impairment
- Established heart failure: Emerging evidence of safety and potential benefit in stable heart failure patients
- Secondary prevention: Effectiveness in patients with prior cardiovascular events
Mechanisms of Tirzepatide-Mediated Cardiovascular Protection: Research Perspectives
Endothelial Function and Vascular Health
Endothelial dysfunction represents a critical early step in atherosclerosis development and a key therapeutic target for cardiovascular risk reduction. Tirzepatide cardiovascular outcomes appear substantially mediated through improvements in endothelial function across multiple vascular beds.
Nitric Oxide Bioavailability:
Research demonstrates that tirzepatide enhances endothelial nitric oxide (NO) production through several mechanisms:
- eNOS activation: Direct stimulation of endothelial nitric oxide synthase via GLP-1 receptor-mediated PI3K/Akt signaling
- Reduced oxidative stress: Decreased superoxide production preserving NO bioavailability
- Improved substrate availability: Enhanced L-arginine transport and reduced asymmetric dimethylarginine (ADMA) levels
- Vascular smooth muscle sensitivity: Enhanced responsiveness to NO-mediated vasodilation
Endothelial Inflammation Reduction:
Chronic low-grade inflammation drives endothelial activation and atherosclerosis progression. Tirzepatide demonstrates anti-inflammatory effects relevant to cardiovascular protection:
- Reduced adhesion molecule expression: Decreased VCAM-1, ICAM-1, and E-selectin on endothelial surfaces
- Inflammatory cytokine suppression: Lower circulating levels of IL-6, TNF-α, and high-sensitivity CRP
- NF-κB pathway inhibition: Reduced pro-inflammatory transcription factor activation
- Improved endothelial barrier function: Enhanced tight junction integrity reducing vascular permeability
Research Application: Investigators studying endothelial function can utilize tirzepatide in cell culture models, isolated vessel preparations, and in vivo vascular reactivity studies to elucidate specific molecular mechanisms underlying these beneficial effects.
Anti-Atherosclerotic Effects and Plaque Stabilization
Beyond preventing endothelial dysfunction, tirzepatide cardiovascular outcomes include direct effects on atherosclerotic plaque development and stability – critical determinants of acute cardiovascular events.
Lipid Accumulation and Foam Cell Formation:
Research in experimental atherosclerosis models demonstrates tirzepatide effects on early atherogenic processes:
- Reduced LDL oxidation: Decreased oxidative modification of lipoproteins reducing uptake by macrophages
- Macrophage polarization: Shift from pro-inflammatory M1 to anti-inflammatory M2 phenotype
- Cholesterol efflux enhancement: Improved reverse cholesterol transport from arterial wall
- Reduced foam cell formation: Decreased lipid-laden macrophage accumulation in arterial intima
Plaque Composition and Stability:
Advanced atherosclerotic lesions prone to rupture cause most acute cardiovascular events. Tirzepatide influences plaque characteristics associated with stability:
- Increased fibrous cap thickness: Enhanced collagen deposition creating more stable plaque architecture
- Reduced necrotic core size: Decreased apoptosis and improved clearance of dead cells
- Lower inflammatory cell infiltration: Reduced macrophage and T-cell content within plaques
- Decreased matrix metalloproteinase activity: Reduced enzymatic degradation of stabilizing extracellular matrix
Imaging Studies:
Advanced cardiovascular imaging in clinical trials provides evidence of tirzepatide’s anti-atherosclerotic effects:
- Coronary CT angiography: Reduced progression of coronary artery calcification and non-calcified plaque volume
- Carotid intima-media thickness: Decreased progression or regression of carotid atherosclerosis
- PET imaging: Reduced arterial inflammation measured by FDG uptake
- Optical coherence tomography: Improved fibrous cap thickness in coronary lesions
Researchers investigating atherosclerosis mechanisms can obtain high-purity research peptides to conduct controlled studies in relevant experimental models.
Cardiac Remodeling and Heart Failure Prevention
Adverse cardiac remodeling – the structural and functional changes that occur in response to injury or chronic stress – represents a critical pathway to heart failure development. Tirzepatide cardiovascular outcomes include beneficial effects on cardiac structure and function that may prevent or reverse pathological remodeling.
Left Ventricular Hypertrophy Reduction:
Excessive left ventricular mass increases cardiovascular risk and heart failure susceptibility. Tirzepatide demonstrates favorable effects on cardiac geometry:
- Reduced LV mass index: Regression of pathological hypertrophy in echocardiographic studies
- Improved LV mass-to-volume ratio: Normalization of concentric remodeling patterns
- Decreased wall thickness: Reduction in interventricular septal and posterior wall dimensions
- Mechanisms: Combination of blood pressure reduction, decreased afterload, and direct anti-hypertrophic signaling
Myocardial Fibrosis Attenuation:
Cardiac fibrosis impairs contractility and electrical conduction, promoting both heart failure and arrhythmias. Research suggests tirzepatide reduces fibrotic remodeling:
- Reduced collagen deposition: Decreased extracellular matrix accumulation in myocardial tissue
- TGF-β pathway modulation: Suppression of pro-fibrotic signaling cascades
- Improved cardiomyocyte-fibroblast interactions: Reduced pathological fibroblast activation
- Biomarker evidence: Decreased circulating markers of collagen turnover (PICP, PIIINP)
Diastolic Function Improvement:
Heart failure with preserved ejection fraction (HFpEF) represents an increasingly prevalent condition with limited therapeutic options. Tirzepatide shows promise in improving diastolic parameters:
- Enhanced myocardial relaxation: Improved early diastolic filling velocities on echocardiography
- Reduced filling pressures: Decreased E/e’ ratio indicating lower left atrial pressure
- Improved left atrial function: Reduced left atrial volume and enhanced atrial contractility
- Exercise capacity: Improved functional capacity in patients with diastolic dysfunction
🔬 Experimental Models: Researchers can investigate these cardiac remodeling effects using tirzepatide in pressure-overload models (transverse aortic constriction), diabetic cardiomyopathy models, and ischemia-reperfusion injury protocols.
Arrhythmia and Sudden Cardiac Death Risk
Cardiac arrhythmias, particularly ventricular arrhythmias, contribute significantly to cardiovascular mortality. While less extensively studied than other cardiovascular outcomes, emerging evidence suggests tirzepatide may influence arrhythmic risk through multiple mechanisms.
Electrophysiological Effects:
Research investigating tirzepatide’s effects on cardiac electrophysiology reveals several potentially anti-arrhythmic mechanisms:
- QT interval effects: Neutral or mildly favorable effects on ventricular repolarization
- Autonomic balance: Improved sympathovagal balance reducing arrhythmogenic sympathetic predominance
- Ion channel modulation: Potential effects on cardiac sodium, potassium, and calcium channels
- Reduced ectopy: Decreased premature ventricular contractions in ambulatory monitoring studies
Substrate Modification:
Beyond acute electrophysiological effects, tirzepatide may reduce arrhythmic substrate through structural improvements:
- Reduced myocardial fibrosis: Decreased anatomical substrate for re-entrant arrhythmias
- Improved myocardial perfusion: Enhanced coronary microvascular function reducing ischemia-triggered arrhythmias
- Decreased inflammation: Reduced inflammatory infiltrates that can disrupt electrical conduction
- Metabolic optimization: Improved myocardial energy metabolism enhancing electrical stability
Clinical Arrhythmia Data:
While dedicated electrophysiology studies remain limited, available clinical trial data suggest:
- Atrial fibrillation: Potential reduction in new-onset atrial fibrillation incidence
- Sudden cardiac death: Numerical reduction in sudden death events in cardiovascular outcome trials
- Device therapy: Preliminary data suggesting reduced appropriate ICD shocks in patients with implanted defibrillators
- Safety: No pro-arrhythmic signals across extensive clinical trial populations
Tirzepatide Research Applications in Cardiovascular Investigation
Experimental Model Systems for Cardiovascular Research
Researchers investigating tirzepatide cardiovascular outcomes utilize diverse experimental systems, each offering unique advantages for mechanistic investigation. Understanding appropriate model selection enhances research quality and translatability.
In Vitro Cellular Models:
Cell culture systems enable precise investigation of molecular mechanisms underlying tirzepatide’s cardiovascular effects:
Endothelial Cell Models:
- Human umbilical vein endothelial cells (HUVECs)
- Human coronary artery endothelial cells (HCAECs)
- Immortalized endothelial cell lines (EA.hy926)
- Applications: NO production, inflammatory marker expression, barrier function, angiogenesis
Cardiomyocyte Models:
- Primary neonatal or adult cardiomyocytes
- Human induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs)
- Cardiac cell lines (H9c2, HL-1)
- Applications: Contractility, calcium handling, hypertrophy signaling, apoptosis resistance
Vascular Smooth Muscle Cells:
- Primary human aortic smooth muscle cells
- Coronary artery smooth muscle cells
- Applications: Proliferation, migration, phenotypic modulation, calcification
Macrophage/Foam Cell Models:
- THP-1 monocyte-derived macrophages
- Primary human monocyte-derived macrophages
- Applications: Lipid accumulation, inflammatory cytokine production, polarization states
Researchers conducting cellular studies benefit from high-purity tirzepatide that ensures consistent results across experimental replicates.
Animal Models for Cardiovascular Outcomes Research
Rodent Models:
Small animal models provide cost-effective platforms for investigating tirzepatide cardiovascular outcomes with genetic and environmental control:
Atherosclerosis Models:
- ApoE knockout mice: Spontaneous atherosclerosis development on high-fat diet
- LDLR knockout mice: Severe hyperlipidemia and accelerated atherosclerosis
- Applications: Plaque development, composition analysis, regression studies
Diabetic Cardiomyopathy Models:
- db/db mice: Leptin receptor deficiency causing obesity and diabetes
- Zucker diabetic fatty rats: Genetic obesity and type 2 diabetes
- Streptozotocin-induced diabetes: Type 1 diabetes model with cardiac complications
- Applications: Cardiac function assessment, structural remodeling, metabolic parameters
Heart Failure Models:
- Transverse aortic constriction (TAC): Pressure overload-induced heart failure
- Myocardial infarction: Coronary artery ligation causing ischemic heart failure
- Doxorubicin cardiomyopathy: Chemotherapy-induced cardiac dysfunction
- Applications: Ejection fraction, cardiac remodeling, survival analysis
Hypertension Models:
- Spontaneously hypertensive rats (SHR): Genetic hypertension with end-organ damage
- DOCA-salt hypertension: Mineralocorticoid-induced hypertension
- Applications: Blood pressure monitoring, vascular remodeling, renal effects
Large Animal Models:
Large animal models offer cardiovascular physiology more similar to humans, enhancing translational relevance:
Porcine Models:
- Metabolic syndrome pigs: Diet-induced obesity, insulin resistance, dyslipidemia
- Coronary artery disease models: Atherosclerosis induction via diet and injury
- Applications: Advanced imaging, hemodynamic measurements, coronary physiology
Non-Human Primate Models:
- Cynomolgus or rhesus macaques: Spontaneous metabolic syndrome and atherosclerosis
- Applications: Long-term cardiovascular outcome studies, imaging, complex metabolic assessments
Research Protocols and Methodological Considerations
Dose Selection and Administration:
Appropriate tirzepatide dosing in experimental models requires consideration of:
- Pharmacokinetic differences: Species-specific absorption, distribution, metabolism, and excretion
- Receptor expression patterns: Tissue-specific GIP and GLP-1 receptor distribution varies across species
- Dose scaling: Allometric scaling from human therapeutic doses to animal model equivalents
- Administration routes: Subcutaneous injection (mimicking clinical use) versus other routes for mechanistic studies
Temporal Considerations:
Tirzepatide cardiovascular outcomes manifest across different timeframes:
- Acute effects (hours-days): Hemodynamic changes, immediate metabolic responses
- Subacute effects (weeks): Weight loss, glycemic improvement, early vascular function changes
- Chronic effects (months): Structural cardiac remodeling, atherosclerosis progression/regression, long-term outcome differences
Outcome Measurement Approaches:
Comprehensive cardiovascular assessment in tirzepatide research includes:
Functional Assessments:
- Echocardiography (ejection fraction, diastolic parameters, strain imaging)
- Cardiac MRI (tissue characterization, perfusion, fibrosis quantification)
- Invasive hemodynamics (pressure-volume loops, coronary flow reserve)
- Exercise testing (functional capacity, oxygen consumption)
Structural Analyses:
- Histopathology (fibrosis quantification, inflammatory cell infiltration)
- Immunohistochemistry (protein expression patterns, cellular markers)
- Molecular imaging (PET, SPECT for inflammation and metabolism)
- Vascular imaging (intravascular ultrasound, optical coherence tomography)
Molecular and Biochemical Endpoints:
- Gene expression profiling (RNA sequencing, qPCR arrays)
- Protein analysis (Western blotting, ELISA, mass spectrometry)
- Metabolomics (comprehensive metabolite profiling)
- Biomarker measurement (troponin, natriuretic peptides, inflammatory markers)
Researchers designing comprehensive cardiovascular studies can access research-grade peptides and resources to support rigorous experimental protocols.
Quality Considerations for Cardiovascular Peptide Research
Peptide Purity and Characterization:
Research-grade tirzepatide quality directly impacts experimental reproducibility and data interpretation:
- Purity standards: ≥95% purity recommended for most cardiovascular research applications
- Characterization methods: HPLC, mass spectrometry, amino acid analysis
- Stability testing: Verification of peptide integrity throughout experimental timeframes
- Batch consistency: Certificate of analysis (COA) documentation for each lot
Storage and Handling:
Proper tirzepatide storage preserves peptide integrity and ensures reliable cardiovascular research outcomes:
- Lyophilized storage: -20°C or -80°C in desiccated conditions
- Reconstituted storage: 2-8°C for short-term use; -20°C with appropriate cryoprotectants for longer storage
- Freeze-thaw cycles: Minimize to prevent peptide degradation
- Light protection: Store in amber pen peptides or protect from light exposure
Experimental Controls:
Rigorous cardiovascular research requires appropriate control conditions:
- Vehicle controls: Matched buffer/excipient controls for all treatment groups
- Positive controls: Established cardiovascular interventions for comparison
- Dose-response assessment: Multiple tirzepatide concentrations to establish relationship
- Time-course evaluation: Multiple timepoints to characterize temporal dynamics
Future Directions in Tirzepatide Cardiovascular Outcomes Research
Ongoing Clinical Trials and Emerging Evidence
The research landscape for tirzepatide cardiovascular outcomes continues to expand rapidly, with numerous ongoing investigations that will further define its cardiovascular profile and optimal applications.
SUMMIT Trial (Semaglutide Effects on Heart Disease and Metabolism in Heart Failure):
While focused on semaglutide, parallel investigations with tirzepatide are examining:
- Heart failure outcomes specifically in HFpEF populations
- Exercise capacity and quality of life improvements
- Mechanistic insights into metabolic-cardiac interactions
- Comparative effectiveness versus other heart failure therapies
Obesity-Related Cardiovascular Outcome Studies:
Dedicated trials in populations without diabetes but with obesity and cardiovascular risk:
- Primary prevention populations: Individuals at high cardiovascular risk without established disease
- Secondary prevention: Patients with prior cardiovascular events
- Special populations: Chronic kidney disease, peripheral artery disease, stroke survivors
Mechanistic Substudies:
Advanced imaging and biomarker substudies nested within larger trials provide mechanistic insights:
- Cardiac MRI substudies: Detailed assessment of myocardial structure, function, and tissue characterization
- Coronary imaging: CT angiography and intravascular imaging evaluating atherosclerosis progression
- Biomarker panels: Comprehensive assessment of inflammatory, metabolic, and cardiac stress markers
- Omics approaches: Proteomics, metabolomics, and transcriptomics identifying novel pathways
Novel Research Questions and Unexplored Mechanisms
Microbiome-Cardiovascular Interactions:
Emerging evidence suggests GLP-1 and GIP pathways influence gut microbiome composition, which in turn affects cardiovascular risk:
- Microbial metabolite production: Short-chain fatty acids, trimethylamine N-oxide (TMAO), and other cardiovascular-relevant metabolites
- Intestinal barrier function: Reduced endotoxemia and systemic inflammation
- Bile acid metabolism: Altered bile acid profiles affecting cardiovascular signaling
- Research opportunities: Investigating whether tirzepatide’s cardiovascular benefits are partially microbiome-mediated
Circadian Rhythm and Cardiovascular Effects:
GLP-1 and GIP receptors exhibit circadian expression patterns, suggesting potential chronopharmacological considerations:
- Time-of-day administration effects: Optimal timing for cardiovascular benefit
- Circadian blood pressure patterns: Effects on nocturnal dipping and morning surge
- Metabolic-circadian integration: Coordination of metabolic and cardiovascular rhythms
- Research applications: Chronobiology studies examining temporal aspects of tirzepatide cardiovascular outcomes
Sex Differences in Cardiovascular Responses:
Biological sex influences cardiovascular disease manifestation and treatment responses:
- Hormonal interactions: Estrogen and testosterone effects on GIP/GLP-1 signaling
- Body composition differences: Sex-specific fat distribution affecting cardiovascular risk
- Cardiovascular disease phenotypes: Different presentation patterns requiring tailored investigation
- Research need: Dedicated studies examining sex-specific tirzepatide cardiovascular outcomes
Aging and Cardiovascular Protection:
The aging cardiovascular system presents unique challenges and opportunities:
- Senescent cell accumulation: Potential effects on cardiac and vascular aging
- Mitochondrial function: Age-related mitochondrial dysfunction and tirzepatide’s metabolic effects
- Frailty and cardiovascular risk: Integration of geriatric syndromes with cardiovascular outcomes
- Research direction: Geroscience approaches examining tirzepatide effects on cardiovascular aging
Combination Strategies and Synergistic Approaches
Multi-Pathway Cardiovascular Protection:
Combining tirzepatide with other cardiovascular interventions may provide synergistic benefits:
Tirzepatide + SGLT2 Inhibitors:
- Complementary mechanisms: Different metabolic and cardiovascular pathways
- Additive benefits: Enhanced weight loss, glycemic control, and cardiovascular protection
- Heart failure synergy: Combined benefits on ejection fraction and hospitalizations
- Research potential: Investigating optimal combination timing and dosing
Tirzepatide + Cardiovascular Medications:
- Statins: Combined effects on atherosclerosis and inflammation
- ACE inhibitors/ARBs: Enhanced blood pressure control and renoprotection
- Antiplatelet agents: Comprehensive atherothrombotic risk reduction
- Research questions: Drug-drug interactions, safety profiles, outcome optimization
Novel Peptide Combinations:
Researchers are exploring combinations of multiple peptide therapeutics:
- Tirzepatide + BPC-157: Potential synergy in vascular repair and cardioprotection
- Tirzepatide + Thymosin Beta-4: Combined effects on cardiac regeneration and remodeling
- Multi-agonist approaches: Triple or quadruple receptor agonists building on tirzepatide’s dual-agonist success
Precision Medicine and Biomarker-Guided Therapy
Predictive Biomarkers:
Identifying which patients derive maximal cardiovascular benefit from tirzepatide represents a key research priority:
- Genetic markers: Polymorphisms in GIP/GLP-1 receptors or downstream signaling molecules
- Baseline inflammatory profiles: High-sensitivity CRP, cytokine patterns predicting response
- Metabolic signatures: Metabolomic profiles identifying optimal candidates
- Imaging biomarkers: Baseline cardiac or vascular imaging predicting treatment response
Pharmacogenomics:
Understanding genetic influences on tirzepatide cardiovascular outcomes:
- Receptor variants: GIP and GLP-1 receptor polymorphisms affecting signaling
- Drug metabolism genes: Genetic variation in peptide degradation pathways
- Cardiovascular disease genetics: Interaction between treatment and genetic cardiovascular risk
- Research application: Pharmacogenomic studies in diverse populations
Response Monitoring:
Developing biomarkers to track cardiovascular benefit during tirzepatide treatment:
- Early response indicators: Biomarkers changing within weeks predicting long-term cardiovascular outcomes
- Treatment optimization: Biomarker-guided dose adjustment strategies
- Mechanism confirmation: Biomarkers confirming engagement of specific cardiovascular pathways
- Research need: Longitudinal biomarker studies correlating changes with hard cardiovascular endpoints
Practical Considerations for Researchers Investigating Tirzepatide Cardiovascular Outcomes
Sourcing Research-Grade Tirzepatide
Quality Requirements:
Cardiovascular research demands high-purity peptides to ensure reliable, reproducible results:
- Minimum purity: ≥95% for most applications; ≥98% for highly sensitive mechanistic studies
- Characterization documentation: Complete analytical data including HPLC chromatograms, mass spectrometry
- Sterility: Sterile-filtered for in vivo applications
- Endotoxin levels: <1.0 EU/mg for cell culture; <0.1 EU/mg for in vivo cardiovascular studies
Supplier Selection Criteria:
Choosing appropriate peptide suppliers impacts research quality and timeline:
- Regulatory compliance: GMP or ISO-certified manufacturing for clinical-grade research
- Technical support: Access to scientific expertise for application guidance
- Consistency: Batch-to-batch reproducibility with comprehensive documentation
- Delivery reliability: Appropriate shipping conditions maintaining peptide integrity
Researchers in the UK and internationally can access high-purity tirzepatide with same-day dispatch for orders placed before 1pm (Mon-Fri), ensuring rapid project initiation.
Reconstitution and Preparation Protocols
Reconstitution Guidelines:
Proper tirzepatide reconstitution preserves peptide activity for cardiovascular research:
Standard Reconstitution Protocol:
- Allow equilibration: Bring lyophilized peptide to room temperature before opening
- Solvent selection: Sterile water, PBS, or buffer appropriate for experimental system
- Gentle mixing: Add solvent slowly down pen peptide wall; swirl gently (avoid vigorous shaking)
- Concentration verification: Calculate final concentration based on peptide mass and solvent volume
- Filtration (if needed): 0.22 μm filter for sterilization of non-sterile preparations
Solvent Considerations:
- Aqueous solutions: Water or PBS for most applications; pH 7.0-7.4 optimal
- Buffer systems: HEPES or Tris buffers for pH-sensitive experiments
- Additives: BSA (0.1%) or human serum albumin as carrier proteins for dilute solutions
- Organic solvents: Generally avoided; if necessary, DMSO <1% final concentration
Concentration Optimization:
- Stock solutions: 1-10 mg/mL for storage; higher concentrations reduce freeze-thaw impact
- Working solutions: Dilute to experimental concentrations immediately before use
- Stability considerations: More dilute solutions less stable; prepare fresh when possible
Storage and Stability Optimization
Short-Term Storage:
For active research projects with frequent tirzepatide use:
- Lyophilized peptide: -20°C in original sealed pen peptide with desiccant
- Reconstituted solution: 2-8°C for up to 2 weeks in sterile conditions
- Aliquoting: Divide into single-use aliquots to minimize freeze-thaw cycles
- Labeling: Clear documentation of reconstitution date, concentration, solvent
Long-Term Storage:
For extended storage maintaining tirzepatide cardiovascular research applications:
- Lyophilized storage: -80°C for maximum stability (years)
- Reconstituted storage: -20°C or -80°C with appropriate cryoprotectants
- Cryoprotectant options: Glycerol (10-20%), trehalose (5%), or specialized peptide stabilizers
- Container selection: Low-binding polypropylene tubes; avoid repeated opening
Stability Monitoring:
Verifying tirzepatide integrity throughout research studies:
- Visual inspection: Clear solution without precipitation or color change
- Activity assays: Functional assays confirming biological activity
- Analytical methods: HPLC or mass spectrometry for degradation assessment
- Documentation: Maintain stability records for research reproducibility
Regulatory and Ethical Considerations
Research Use Designation:
All tirzepatide obtained for cardiovascular research must be clearly designated:
- “For Research Use Only” labeling: Explicit documentation of research-only status
- Not for human consumption: Clear distinction from clinical-grade materials
- Institutional approval: Appropriate institutional review board or ethics committee approval for all studies
- Animal welfare: IACUC or equivalent approval for in vivo cardiovascular research
Documentation and Record-Keeping:
Comprehensive documentation supports research integrity:
- Chain of custody: Complete records from peptide receipt through experimental use
- Batch tracking: Lot numbers, COAs, and characterization data for all materials
- Experimental logs: Detailed protocols, observations, and results
- Compliance records: Regulatory approvals, safety assessments, disposal documentation
Safety Considerations:
While tirzepatide demonstrates favorable safety profiles, appropriate laboratory safety practices apply:
- Personal protective equipment: Gloves, lab coat, eye protection when handling
- Containment: Appropriate biosafety level for in vitro and in vivo work
- Spill procedures: Established protocols for peptide solution spills
- Disposal: Appropriate biohazardous waste disposal following institutional guidelines
The Evolving Landscape of Tirzepatide Cardiovascular Research
The comprehensive body of evidence examining tirzepatide cardiovascular outcomes has firmly established this dual GIP/GLP-1 receptor agonist as a significant advance in metabolic-cardiovascular therapeutics. From landmark clinical trials demonstrating substantial MACE reduction to mechanistic research elucidating molecular pathways of cardioprotection, tirzepatide represents a compelling example of how peptide therapeutics can address complex, multifactorial disease processes.
For the research community, tirzepatide offers exceptional opportunities to investigate integrated metabolic-cardiovascular biology. Its dual-agonist mechanism provides unique insights into GIP and GLP-1 receptor synergy, while its robust cardiovascular benefits enable investigation of multiple cardioprotective pathways simultaneously. Whether examining endothelial function, atherosclerosis progression, cardiac remodeling, or novel mechanisms like microbiome interactions, tirzepatide serves as a valuable research tool with well-characterized effects and extensive clinical validation.
Key Research Priorities Moving Forward:
✅ Mechanistic elucidation – Detailed investigation of molecular pathways underlying cardiovascular protection
✅ Biomarker development – Identification of predictive and response biomarkers for precision medicine approaches
✅ Combination strategies – Systematic evaluation of synergistic cardiovascular interventions
✅ Special populations – Dedicated research in understudied groups (elderly, chronic kidney disease, established heart failure)
✅ Long-term outcomes – Extended follow-up studies examining durability of cardiovascular benefits
✅ Comparative effectiveness – Head-to-head trials versus other cardiovascular interventions
✅ Novel applications – Exploration of cardiovascular benefits beyond traditional metabolic disease contexts
Actionable Next Steps for Researchers
For investigators initiating tirzepatide cardiovascular research:
- Define specific research questions aligned with knowledge gaps in tirzepatide cardiovascular outcomes
- Select appropriate experimental models matching research objectives and available resources
- Source high-quality research-grade tirzepatide from reputable suppliers with comprehensive documentation
- Establish rigorous protocols including appropriate controls, dose-response assessment, and temporal evaluation
- Implement comprehensive outcome measures spanning functional, structural, and molecular endpoints
- Collaborate across disciplines integrating cardiovascular, metabolic, and molecular expertise
- Disseminate findings through peer-reviewed publications and scientific presentations
For laboratories seeking research-grade peptides:
Researchers conducting cardiovascular peptide investigations can access premium research-grade tirzepatide and comprehensive peptide portfolios with exceptional purity, rapid UK delivery, and professional scientific support. With same-day dispatch for orders placed before 1pm (Mon-Fri), temperature-controlled shipping, and complete product documentation including Certificates of Analysis, research timelines remain on track while maintaining the highest quality standards.
The investigation of tirzepatide cardiovascular outcomes represents a dynamic and rapidly evolving field with profound implications for both basic cardiovascular biology and translational therapeutic development. As research continues to unveil the complex mechanisms underlying its cardioprotective effects, tirzepatide stands as a powerful example of how peptide therapeutics can address the interconnected challenges of metabolic and cardiovascular disease – offering hope for millions affected by these conditions while advancing fundamental understanding of integrated physiological regulation.
