
Understanding tirzepatide dose escalation can mean the difference between achieving optimal therapeutic outcomes and experiencing unnecessary side effects. As one of the most promising dual GIP/GLP-1 receptor agonists currently being studied in metabolic research, tirzepatide demonstrates remarkable efficacy—but only when dose adjustments follow evidence-based protocols. Whether you’re a researcher investigating metabolic pathways or a laboratory professional conducting peptide studies, knowing precisely when to escalate dosage and which clinical markers to monitor is essential for maximizing research validity while maintaining proper experimental protocols.
Tirzepatide Dose Escalation: When to Increase & Signs to Watch represents a critical knowledge area for anyone working with this research peptide. The escalation protocol isn’t arbitrary—it’s carefully designed to balance therapeutic efficacy against tolerability profiles observed in clinical trials. This comprehensive guide examines the complete dose escalation framework, from the initial 2.5 mg starting dose through the maximum 15 mg weekly administration, providing researchers with the detailed information needed to understand optimal dosing strategies.
Key Takeaways
- Tirzepatide follows a structured dose escalation protocol starting at 2.5 mg weekly for 4 weeks, then increasing in 2.5 mg increments every 4 weeks based on tolerability and response
- Weight loss efficacy correlates with dosage levels: 5 mg achieves approximately 15% reduction, 10 mg produces 20% reduction, and 15 mg results in 21% weight loss in clinical studies
- Gastrointestinal side effects increase with higher doses, affecting 39% of subjects at 5 mg versus 49% at 15 mg, making gradual escalation essential for tolerability
- Dose increases should be considered when weight loss plateaus after initial response, indicating potential need for higher efficacy levels
- The 4-week minimum interval between dose adjustments allows adequate physiological adaptation and reduces adverse event frequency
Understanding Tirzepatide: Mechanism and Research Applications

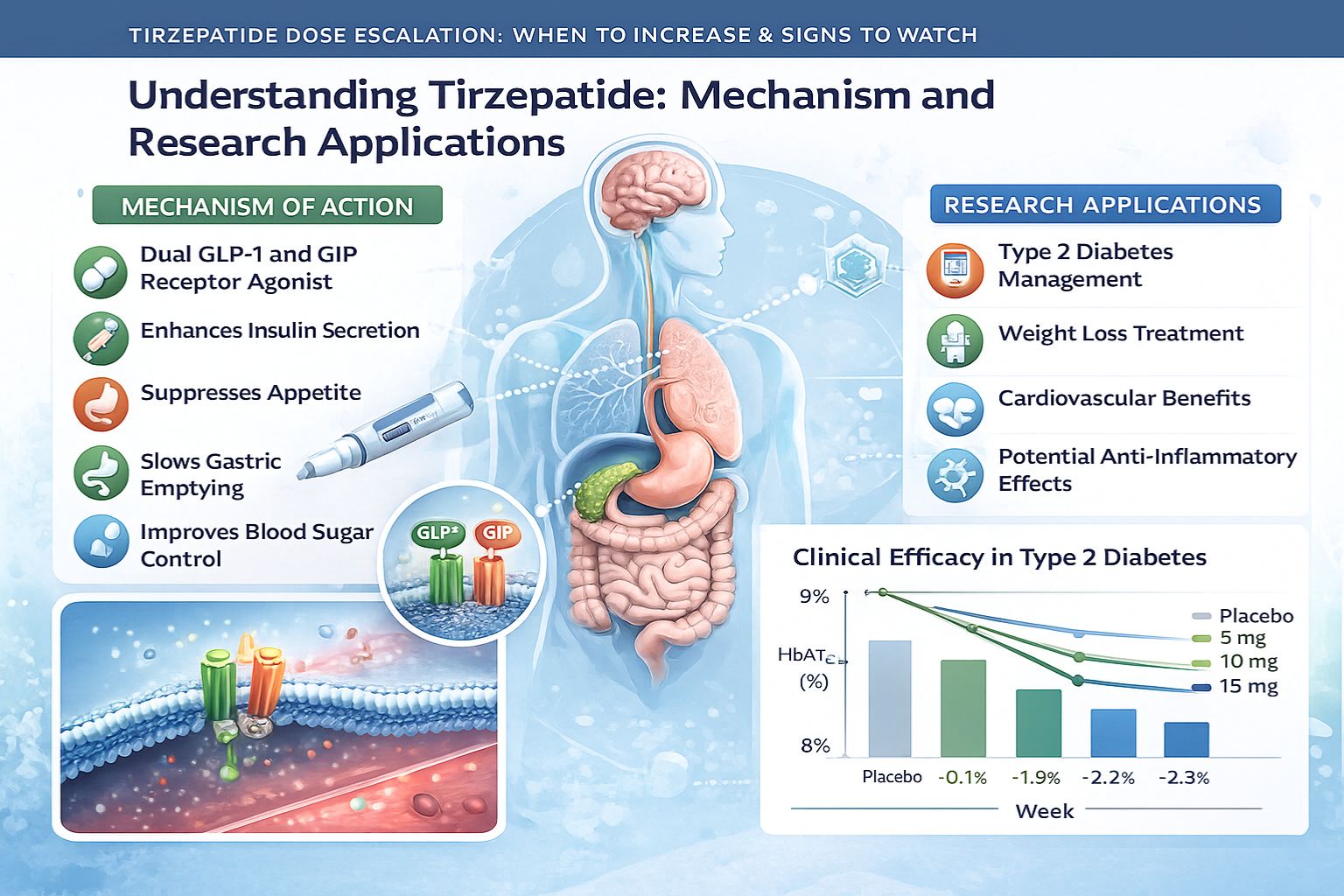
Tirzepatide represents a novel class of research peptides that function as dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonists. This dual mechanism distinguishes tirzepatide from single-receptor agonists, offering researchers unique opportunities to study combined incretin pathway activation.[1]
The peptide’s molecular structure enables simultaneous activation of both GIP and GLP-1 receptors, creating synergistic effects on glucose homeostasis, insulin secretion, and energy expenditure. In laboratory settings, tirzepatide has demonstrated significant impacts on metabolic parameters, making it a valuable compound for metabolic research applications.
Why Dose Escalation Matters in Research Protocols
Dose escalation protocols serve multiple critical functions in peptide research:
Tolerability Assessment 📊
Gradual dose increases allow researchers to identify optimal dosing thresholds while monitoring for adverse responses. Starting with lower doses and systematically increasing provides valuable data on dose-response relationships.
Pharmacokinetic Optimization
The body requires time to adapt to increasing peptide concentrations. Rapid dose escalation can overwhelm metabolic pathways, while gradual increases allow physiological systems to adjust, providing more reliable research data.
Safety Margin Establishment
Progressive dosing helps establish safety margins and identify potential toxicity thresholds, essential information for comprehensive research protocols.
For researchers sourcing high-purity research-grade peptides, understanding these escalation principles ensures experimental protocols align with established clinical frameworks.
The Standard Tirzepatide Dose Escalation Protocol
Tirzepatide Dose Escalation: When to Increase & Signs to Watch follows a precisely defined protocol validated through extensive clinical research. This standardized approach maximizes efficacy while minimizing adverse events.
Initial Starting Dose: 2.5 mg Weekly
The tirzepatide escalation protocol invariably begins with 2.5 mg administered subcutaneously once weekly for 4 weeks. This starting dose serves exclusively as an initiation phase and is not approved as a maintenance dosage in clinical applications.[2]
Why 2.5 mg?
- Allows gradual receptor activation without overwhelming GIP/GLP-1 pathways
- Minimizes initial gastrointestinal adaptation responses
- Establishes baseline tolerability before therapeutic doses
- Provides researchers with initial pharmacodynamic data
The 4-week duration at this starting dose is critical—shorter initiation periods correlate with higher rates of gastrointestinal side effects in clinical studies.
Progression Through Maintenance Doses
After the initial 4-week period at 2.5 mg, the standard escalation proceeds as follows:
| Week | Dose (mg) | Purpose | Expected Response |
|---|---|---|---|
| 1-4 | 2.5 | Initiation only | Minimal weight change, GI adaptation |
| 5-8 | 5.0 | First maintenance dose | Initial metabolic effects, ~5-8% weight reduction |
| 9-12 | 7.5 | Optional escalation | Enhanced efficacy, ~10-12% weight reduction |
| 13-16 | 10.0 | Standard maintenance | Significant effects, ~15-18% weight reduction |
| 17-20 | 12.5 | Higher maintenance | Near-maximal efficacy, ~18-20% weight reduction |
| 21+ | 15.0 | Maximum dose | Maximum efficacy, ~21% weight reduction |
Critical Protocol Requirements:
✅ Minimum 4-week intervals between each dose increase
✅ 2.5 mg increments for all escalation steps
✅ 15 mg maximum dose across all research applications
✅ Subcutaneous administration once weekly on the same day
Available Maintenance Dosages
The approved maintenance dosages for tirzepatide research applications are:
- 5 mg once weekly (first maintenance level)
- 10 mg once weekly (standard maintenance level)
- 15 mg once weekly (maximum maintenance level)
Notably, the 2.5 mg dose is not considered a maintenance dose—it exists solely for treatment initiation. The intermediate doses of 7.5 mg and 12.5 mg serve as optional escalation steps between the primary maintenance levels.[3]
Researchers working with tirzepatide formulations should ensure their protocols account for these specific dosing requirements.
When to Increase Tirzepatide Dosage: Clinical Indicators
Determining the optimal timing for dose escalation requires careful monitoring of multiple parameters. Tirzepatide Dose Escalation: When to Increase & Signs to Watch involves both efficacy markers and tolerability assessments.
Weight Loss Plateau Indicators
One of the primary signals for potential dose escalation is a weight loss plateau following initial response. Research data demonstrates distinct efficacy levels at different doses:
Dose-Dependent Weight Loss Efficacy:
- 5 mg dose: Approximately 15% total body weight reduction
- 10 mg dose: Approximately 20% total body weight reduction
- 15 mg dose: Approximately 21% total body weight reduction[4]
🔍 When to Consider Escalation:
If weight loss stabilizes at levels significantly below expected thresholds for the current dose (e.g., only 10% reduction on 5 mg when 15% is typical), escalation may be warranted after the minimum 4-week period.
Metabolic Response Assessment
Beyond weight metrics, metabolic markers provide valuable escalation guidance:
Glycemic Control Parameters
In metabolic research, suboptimal glucose regulation despite adequate treatment duration may indicate need for higher doses. Tirzepatide’s dual mechanism affects both insulin secretion and insulin sensitivity—inadequate response in these pathways suggests potential for dose optimization.
Lipid Profile Changes
Tirzepatide influences lipid metabolism through multiple mechanisms. Plateauing improvements in lipid parameters may signal opportunity for dose escalation to achieve additional metabolic benefits.
Appetite and Satiety Markers
The GLP-1 component significantly affects appetite regulation. If satiety effects diminish or appetite returns to baseline levels, higher doses may restore these regulatory effects.
Tolerability Confirmation
Before any dose increase, confirming adequate tolerability at the current dose is essential:
✓ Gastrointestinal symptoms resolved or minimal
✓ No persistent nausea or vomiting
✓ Stable injection site reactions
✓ No concerning cardiovascular symptoms
✓ Adequate nutritional intake maintained
Only when tolerability criteria are met should escalation proceed. Researchers should document these parameters thoroughly in experimental protocols.
Signs to Watch: Monitoring During Dose Escalation
Comprehensive monitoring during tirzepatide dose escalation is essential for both safety and research validity. Understanding which signs indicate successful adaptation versus concerning responses guides appropriate protocol adjustments.
Gastrointestinal Side Effects: The Primary Concern
Gastrointestinal (GI) adverse effects represent the most common tolerability challenge with tirzepatide, and their frequency increases with dosage:
GI Side Effect Prevalence by Dose:
- 5 mg dose: Approximately 39% experience nausea or diarrhea
- 15 mg dose: Approximately 49% experience nausea or diarrhea[5]
Common GI Manifestations:
🔸 Nausea (most frequent)
🔸 Diarrhea
🔸 Decreased appetite (therapeutic effect, but can be excessive)
🔸 Vomiting (less common, more concerning)
🔸 Constipation
🔸 Abdominal discomfort or pain
🔸 Dyspepsia
Severity Grading:
| Grade | Description | Action |
|---|---|---|
| Mild | Transient symptoms, no intervention needed | Continue monitoring |
| Moderate | Persistent symptoms, dietary modifications helpful | Consider dose pause |
| Severe | Symptoms interfering with normal function | Reduce or hold dose |
| Very Severe | Dehydration, inability to maintain nutrition | Discontinue, medical evaluation |
The gradual dose escalation protocol specifically aims to minimize these effects by allowing GI adaptation time. Rushing escalation or skipping intermediate doses significantly increases GI adverse event rates.
Systemic Adaptation Signs
Beyond GI effects, several systemic responses warrant monitoring:
Fatigue and Energy Levels
Initial fatigue is common during dose increases as metabolic pathways adjust. This typically resolves within 1-2 weeks. Persistent fatigue beyond 3 weeks may indicate inadequate caloric intake or other metabolic concerns.
Cardiovascular Responses
Monitor for:
- Heart rate changes (tirzepatide may cause modest increases)
- Blood pressure variations
- Palpitations or irregular heartbeat sensations
Injection Site Reactions
While generally mild, monitor for:
- Redness or swelling at injection sites
- Persistent nodules or lumps
- Increasing reaction severity with dose escalation
Hypoglycemic Symptoms
Though less common with tirzepatide than with some other agents, watch for:
- Shakiness or tremors
- Excessive sweating
- Confusion or difficulty concentrating
- Extreme hunger
Positive Response Indicators
Equally important is recognizing signs of successful dose escalation:
✅ Progressive weight reduction following dose increase
✅ Improved satiety and appetite control
✅ Enhanced energy levels after initial adaptation
✅ Stable GI tolerance without worsening symptoms
✅ Positive metabolic marker trends
✅ Sustained compliance with dosing schedule
These positive indicators suggest the current dose is appropriate and well-tolerated, supporting continuation or further escalation per protocol.
For researchers requiring comprehensive peptide support, access to quality compounds and proper storage guidance ensures experimental validity throughout dose escalation studies.
When NOT to Increase: Contraindications and Pause Criteria
Understanding when to avoid dose escalation is equally critical to the Tirzepatide Dose Escalation: When to Increase & Signs to Watch framework. Several scenarios warrant pausing or avoiding scheduled dose increases.
Persistent Side Effects
If GI or systemic side effects persist beyond the typical 2-3 week adaptation period, dose escalation should be postponed:
Pause Criteria:
🛑 Ongoing nausea affecting daily function or nutritional intake
🛑 Recurrent vomiting (more than occasional episodes)
🛑 Severe diarrhea causing dehydration risk
🛑 Persistent fatigue not improving after 3 weeks
🛑 Concerning cardiovascular symptoms
🛑 Injection site reactions worsening with each dose
In research settings, these observations provide valuable data about individual tolerability thresholds and may indicate the current dose represents the maximum tolerable level for that particular subject or model.
Health Status Changes
Changes in overall health status require reassessment before proceeding with escalation:
Medical Considerations:
- Acute illness or infection: Metabolic stress may alter peptide response
- New medication initiation: Potential drug interactions require evaluation
- Surgical procedures: Perioperative period may necessitate dose adjustments
- Pregnancy or lactation: Contraindicated in these states
- Hepatic or renal function changes: May affect peptide metabolism and clearance
Healthcare providers overseeing clinical applications should thoroughly evaluate any health status changes before authorizing dose increases.[6]
Adequate Response at Current Dose
If therapeutic goals are being met at the current dose, escalation may be unnecessary:
Consider Maintaining Current Dose When:
- Weight loss targets are being achieved
- Metabolic parameters show continued improvement
- Side effects are minimal or absent
- Subject/patient satisfaction is high
- Risk-benefit analysis favors stability over potential additional gains
The principle of “minimum effective dose” suggests using the lowest dose that achieves therapeutic objectives, minimizing exposure to higher-dose side effect risks.
Approaching Maximum Dose
As subjects approach the 15 mg maximum dose, particular caution is warranted:
The 15 mg once weekly dose represents the absolute maximum recommended across all tirzepatide applications. No escalation beyond this level is supported by current research, and doing so would represent off-protocol administration with unknown safety implications.
At 12.5 mg and 15 mg doses, the incremental efficacy gains become smaller (21% weight loss at 15 mg versus 20% at 10 mg), while side effect rates continue increasing. Researchers must carefully weigh whether the final escalation step provides meaningful additional benefit.
Practical Dose Escalation Management Strategies
Successfully navigating tirzepatide dose escalation requires more than understanding the protocol—practical management strategies significantly impact tolerability and outcomes.
Timing Dose Increases Strategically
Day of Week Consistency
Maintaining the same weekly injection day provides schedule predictability and helps identify temporal patterns in side effects. Many researchers recommend choosing a day when subjects can rest the following day if needed (e.g., Friday evening for weekend recovery).
Seasonal Considerations
In longer research protocols, consider avoiding dose increases during:
- Holiday periods (dietary changes may confound results)
- High-stress periods (may amplify side effects)
- Extreme weather (may affect activity levels and metabolic parameters)
Minimum Interval Adherence
The 4-week minimum between increases is not arbitrary—it represents the time required for:
- Steady-state plasma concentrations to establish
- GI adaptation to complete
- Metabolic effects to fully manifest
- Accurate assessment of dose-specific responses
Rushing this timeline compromises both safety and research validity.
Dietary Modifications to Support Escalation
Dietary strategies can significantly reduce GI side effects during dose escalation:
Recommended Approaches:
️ Smaller, more frequent meals rather than large portions 🍽
️ Bland, low-fat foods during the first week after dose increase 🍽
️ Adequate hydration (especially important given GI effects) 🍽
️ Avoiding trigger foods (spicy, greasy, or highly processed items) 🍽
️ Protein prioritization to maintain lean mass during weight loss 🍽
️ Fiber moderation initially, then gradual increase for GI health
These modifications don’t alter the peptide’s mechanism but can substantially improve tolerability during adaptation periods.
Injection Technique Optimization
Proper administration technique influences both efficacy and tolerability:
Best Practices:
- Rotation of injection sites (abdomen, thigh, upper arm) to minimize local reactions
- Room temperature administration (remove from refrigeration 30 minutes before injection)
- Slow injection technique to reduce discomfort
- Proper needle depth for subcutaneous (not intramuscular) delivery
- Consistent timing within the weekly schedule
For research applications requiring multiple peptide formulations, maintaining meticulous injection protocols ensures experimental consistency.
Documentation and Monitoring Protocols
Comprehensive documentation enables informed escalation decisions:
Essential Tracking Parameters:
📋 Weight and body composition (weekly minimum)
📋 Side effect log (daily during first 2 weeks after increase)
📋 Dietary intake (particularly during adaptation periods)
📋 Activity levels (may influence metabolic outcomes)
📋 Concurrent medications (potential interactions)
📋 Metabolic markers (glucose, lipids, etc., per protocol schedule)
📋 Quality of life assessments (standardized instruments)
This data provides the evidence base for escalation decisions and contributes to broader research understanding of tirzepatide dose-response relationships.
Missed Doses and Schedule Disruptions
Even with careful planning, dose schedule disruptions occur. Understanding how to manage these situations maintains protocol integrity.
The 4-Day Rule for Missed Doses
If a scheduled weekly dose is missed, the following protocol applies:
Within 4 Days (96 Hours):
Administer the missed dose as soon as remembered, then resume the regular weekly schedule from that new day.
Beyond 4 Days:
Skip the missed dose entirely and administer the next dose on the regularly scheduled day. Do not double-dose to “catch up.”[7]
Impact on Escalation Timeline
Missed doses may necessitate timeline adjustments:
Single Missed Dose:
Generally does not require restarting the 4-week interval at the current dose level. Continue with planned escalation if tolerability and efficacy remain appropriate.
Multiple Missed Doses or Extended Gap:
If more than 2 consecutive doses are missed or a gap exceeds 2 weeks:
- Consider restarting at the previous lower dose
- Re-establish tolerability before proceeding with planned escalation
- Extend the current dose period to ensure adequate assessment
Documentation Requirements:
All schedule disruptions should be thoroughly documented in research protocols, as they may influence outcome interpretation and statistical analyses.
Special Populations and Dose Escalation Considerations
Certain populations may require modified escalation approaches, though tirzepatide is labeled “For Research Use Only” and not approved for general clinical use.
Age-Related Considerations
Older Adult Populations:
Research in older subjects suggests:
- Similar efficacy across age groups
- Potentially higher GI side effect sensitivity
- More conservative escalation timelines may improve tolerability
- Careful monitoring for dehydration risk with GI effects
Younger Adult Populations:
Generally demonstrate:
- Good tolerability across dose ranges
- Robust metabolic responses
- Standard escalation protocols typically appropriate
Baseline Metabolic Status
Higher Baseline BMI:
Subjects with higher initial body mass index may:
- Require full dose escalation to maximum levels for optimal outcomes
- Demonstrate more pronounced weight loss at higher doses
- Tolerate escalation well due to larger metabolic reserve
Lower Baseline BMI:
Subjects closer to normal weight ranges may:
- Achieve therapeutic goals at lower maintenance doses
- Experience more pronounced side effects at higher doses
- Benefit from conservative escalation with careful response monitoring
Concurrent Medication Considerations
Insulin or Insulin Secretagogues:
When used alongside medications affecting glucose regulation:
- Hypoglycemia risk increases, particularly at higher tirzepatide doses
- Dose adjustments of concurrent medications may be necessary
- More frequent glucose monitoring warranted during escalation
Other Weight-Affecting Medications:
Concurrent use of medications that influence weight or appetite requires:
- Careful attribution of effects to tirzepatide versus other agents
- Potential modification of escalation decisions based on combined effects
- Enhanced monitoring for additive side effects
For researchers exploring comprehensive peptide portfolios including tirzepatide and complementary compounds, understanding these interaction considerations is essential for protocol design.
Long-Term Maintenance: After Reaching Target Dose
Once optimal dosing is achieved, long-term maintenance strategies become paramount.
Determining Your Optimal Maintenance Dose
The “optimal” dose is highly individualized and represents the intersection of:
Efficacy Achievement:
- Therapeutic goals met (weight loss targets, metabolic parameters)
- Continued progressive improvement or stable maintenance
- Sustained appetite and satiety regulation
Tolerability Profile:
- Minimal or manageable side effects
- Good quality of life maintenance
- Sustainable long-term adherence
Risk-Benefit Balance:
- Benefits clearly outweigh any ongoing side effects
- No emerging safety concerns
- Appropriate for individual health status
For some subjects, the optimal maintenance dose may be 5 mg weekly; for others, 15 mg may be necessary and well-tolerated. The standardized escalation protocol allows identification of this individual optimum.
Monitoring During Maintenance Phase
Even after escalation completion, ongoing monitoring remains important:
Reduced-Frequency Assessments:
- Weight: Monthly (versus weekly during escalation)
- Metabolic markers: Quarterly (versus monthly during escalation)
- Side effect assessment: Each administration initially, then as needed
- Comprehensive evaluation: Every 6 months minimum
Dose Reduction Considerations
Circumstances that may warrant dose reduction from maintenance levels:
Tolerability Issues:
If side effects emerge or worsen during maintenance, dose reduction may restore tolerability while maintaining partial therapeutic benefit.
Therapeutic Goal Achievement:
Once weight loss targets are met, some protocols explore dose reduction to the minimum effective maintenance level.
Health Status Changes:
New medical conditions, medications, or physiological changes may necessitate dose adjustment.
Dose reduction typically follows the reverse of escalation: decrease by 2.5 mg increments with 4-week intervals to assess response at each level.
Tirzepatide Dose Escalation in Research Settings
For laboratories and research institutions working with tirzepatide, dose escalation protocols require additional considerations beyond clinical applications.
Research Protocol Design
Well-designed tirzepatide research protocols incorporate:
Standardized Escalation Schedules:
Following the established 2.5 mg starting dose with 4-week intervals and 2.5 mg increments ensures comparability with existing literature and clinical frameworks.
Control Groups:
Appropriate controls (placebo, alternative dosing schedules, comparator peptides) strengthen research validity and allow isolation of tirzepatide-specific effects.
Blinding Procedures:
Where feasible, blinding of subjects and researchers to dose levels reduces bias in subjective outcome assessments.
Statistical Power Calculations:
Sample sizes should account for expected dose-response relationships and anticipated dropout rates due to side effects.
Quality Assurance in Research Applications
Research-grade tirzepatide requires:
Purity Verification:
High-purity peptides (≥98%) ensure consistent dosing and minimize confounding from impurities. Research-grade peptide suppliers should provide certificates of analysis (COAs) documenting purity levels.
Proper Storage:
Lyophilized tirzepatide should be stored at 2-8°C in controlled conditions. After reconstitution, refrigerated storage with prompt use maintains peptide stability and dosing accuracy.
Reconstitution Protocols:
Standardized reconstitution procedures using appropriate diluents (typically bacteriostatic water) ensure consistent concentration and dosing precision across the research protocol.
Handling Procedures:
Proper handling under controlled conditions prevents degradation and maintains research validity throughout dose escalation studies.
Regulatory and Ethical Considerations
Research applications of tirzepatide must adhere to:
Clear Labeling:
All research peptides must be clearly labeled “For Research Use Only” with no implications for human therapeutic use outside approved clinical trials.
Institutional Oversight:
Appropriate institutional review board (IRB) or ethics committee approval for any research involving human subjects.
Informed Consent:
Comprehensive informed consent processes that clearly communicate dose escalation protocols, potential side effects, and research objectives.
Adverse Event Reporting:
Established procedures for identifying, documenting, and reporting adverse events, particularly during dose escalation phases when side effect risk is elevated.
Comparing Tirzepatide Dose Escalation to Other Peptides
Understanding tirzepatide’s dose escalation protocol in context with other metabolic peptides provides valuable perspective.
Tirzepatide vs. Semaglutide
Semaglutide (GLP-1 agonist only) follows a different escalation schedule:
| Parameter | Tirzepatide | Semaglutide |
|---|---|---|
| Starting Dose | 2.5 mg weekly | 0.25 mg weekly |
| Escalation Increment | 2.5 mg | Variable (0.25→0.5→1.0→1.7→2.4 mg) |
| Time Between Increases | 4 weeks minimum | 4 weeks minimum |
| Maximum Dose | 15 mg weekly | 2.4 mg weekly |
| Mechanism | Dual GIP/GLP-1 | GLP-1 only |
The higher absolute doses of tirzepatide reflect its dual mechanism and different molecular weight, not necessarily greater potency. Both require gradual escalation to optimize tolerability.
Researchers working with semaglutide formulations alongside tirzepatide should maintain distinct protocols for each peptide.
Tirzepatide vs. Liraglutide
Liraglutide represents an earlier GLP-1 agonist with daily rather than weekly administration:
- Daily dosing (versus weekly for tirzepatide)
- Escalation from 0.6 mg to 3.0 mg over several weeks
- More frequent dose adjustments possible due to daily administration
- Different side effect temporal patterns due to dosing frequency
The weekly administration of tirzepatide simplifies escalation protocols and may improve adherence in longer research studies.
Tirzepatide vs. Retatrutide
Retatrutide represents a newer triple agonist (GIP/GLP-1/glucagon):
- Similar weekly administration schedule
- Different starting doses and escalation increments
- Potentially enhanced efficacy due to triple mechanism
- Distinct side effect profile requiring separate monitoring protocols
As research with retatrutide expands, comparative dose escalation studies will provide valuable insights into optimal protocols for multi-agonist peptides.
Frequently Asked Questions About Tirzepatide Dose Escalation
How long does it take to reach the maximum tirzepatide dose?
Following the standard protocol with 4-week intervals between increases:
- Week 1-4: 2.5 mg (initiation)
- Week 5-8: 5 mg (first maintenance)
- Week 9-12: 7.5 mg (optional)
- Week 13-16: 10 mg (standard maintenance)
- Week 17-20: 12.5 mg (optional)
- Week 21+: 15 mg (maximum)
Minimum time to maximum dose: 20 weeks (approximately 5 months) if escalating at every opportunity. Many subjects reach their optimal maintenance dose before the maximum, extending on intermediate doses based on response and tolerability.
Can I skip intermediate doses and escalate faster?
No. Skipping intermediate doses or shortening the 4-week intervals between increases significantly increases the risk of:
- Severe gastrointestinal side effects
- Poor tolerability leading to discontinuation
- Inadequate assessment of dose-specific responses
- Compromised research validity in experimental protocols
The established escalation schedule is evidence-based and designed to optimize the balance between efficacy and tolerability. Deviating from this protocol is not recommended.
What if I experience side effects at a higher dose?
If side effects become problematic after dose escalation:
Option 1: Temporary Dose Pause
Remain at the current dose for an additional 4 weeks to allow further adaptation before considering additional escalation.
Option 2: Dose Reduction
Return to the previous well-tolerated dose level. Many subjects find their optimal maintenance dose is not the maximum dose.
Option 3: Supportive Management
Implement dietary modifications, hydration strategies, and other supportive measures while maintaining the current dose.
Option 4: Discontinuation
If side effects are severe or persistent despite interventions, discontinuation may be necessary. This should be done under appropriate supervision in clinical settings.
Is the 2.5 mg dose effective for weight loss?
The 2.5 mg dose is designated as an initiation dose only and is not approved as a maintenance dose. While some metabolic effects may be observed at this level, the therapeutic efficacy for weight loss is substantially lower than at maintenance doses (5 mg, 10 mg, or 15 mg).
Research data specifically supports the 5 mg, 10 mg, and 15 mg doses as maintenance levels with established efficacy profiles. The 2.5 mg dose serves to minimize initial side effects and allow gradual physiological adaptation.
How does body weight affect dose escalation decisions?
Body weight influences dose escalation in several ways:
Higher Baseline Weight:
- May require escalation to higher maintenance doses (10-15 mg) for optimal outcomes
- Typically demonstrates good tolerability across the dose range
- Larger absolute weight loss at higher doses
Lower Baseline Weight:
- May achieve therapeutic goals at lower maintenance doses (5-7.5 mg)
- Potentially more sensitive to side effects at higher doses
- Dose escalation decisions based more on metabolic parameters than absolute weight loss
Individual response variability means dose escalation should be guided by the specific subject’s tolerability and efficacy profile rather than weight alone.
Conclusion: Optimizing Tirzepatide Dose Escalation for Best Outcomes
Tirzepatide Dose Escalation: When to Increase & Signs to Watch represents a critical framework for maximizing this peptide’s therapeutic potential while maintaining appropriate safety margins. The evidence-based protocol—starting at 2.5 mg weekly for 4 weeks, then escalating in 2.5 mg increments every 4 weeks to a maximum of 15 mg—has been carefully designed based on extensive clinical research demonstrating optimal efficacy-tolerability balance.
Key Principles for Successful Escalation
Patience and Adherence to Protocol
The 4-week minimum interval between dose increases is not arbitrary—it represents the time required for physiological adaptation, steady-state achievement, and accurate response assessment. Rushing this timeline compromises both safety and outcomes.
Individualized Decision-Making
While the escalation schedule is standardized, the optimal maintenance dose varies considerably between individuals. Some subjects achieve therapeutic goals at 5 mg weekly, while others require 15 mg for maximum benefit. Careful monitoring of both efficacy markers and tolerability indicators guides these individualized decisions.
Comprehensive Monitoring
Successful dose escalation requires systematic tracking of weight, metabolic parameters, side effects, and quality of life indicators. This documentation provides the evidence base for informed escalation decisions and contributes to broader research understanding.
Proactive Side Effect Management
Gastrointestinal effects represent the primary tolerability challenge, affecting 39-49% of subjects depending on dose level. Dietary modifications, hydration strategies, and appropriate timing of dose increases significantly improve tolerability and reduce discontinuation rates.
Actionable Next Steps
For researchers and laboratory professionals working with tirzepatide:
- Establish Standardized Protocols: Develop comprehensive dose escalation protocols that align with evidence-based frameworks while addressing your specific research objectives.
- Source High-Quality Peptides: Ensure research-grade tirzepatide with verified purity (≥98%) and appropriate storage conditions. PEPTIDE PRO provides research-grade peptides with full documentation and quality assurance.
- Implement Robust Monitoring Systems: Create systematic data collection procedures for tracking dose-response relationships, side effects, and long-term outcomes.
- Plan for Individual Variability: Design protocols with sufficient flexibility to accommodate individual differences in tolerability and response while maintaining scientific rigor.
- Stay Current with Emerging Research: The tirzepatide research landscape continues evolving. Regular review of new publications ensures protocols incorporate the latest evidence.
- Maintain Regulatory Compliance: Ensure all research applications adhere to appropriate institutional oversight, clearly label peptides “For Research Use Only,” and maintain comprehensive documentation.
The Future of Tirzepatide Dose Optimization
As research with tirzepatide expands, several areas warrant continued investigation:
Personalized Escalation Strategies: Future research may identify biomarkers or genetic factors that predict optimal escalation timelines and maintenance doses for individual subjects.
Combination Approaches: Studies examining tirzepatide in combination with other metabolic peptides may reveal synergistic effects and modified escalation requirements.
Long-Term Maintenance Optimization: Extended follow-up studies will clarify optimal long-term maintenance strategies, including potential for dose reduction after goal achievement.
Alternative Escalation Schedules: While the standard 4-week interval protocol is well-established, research into alternative timing strategies may identify approaches that further optimize tolerability without compromising efficacy.
Understanding Tirzepatide Dose Escalation: When to Increase & Signs to Watch empowers researchers to design rigorous protocols, clinicians to optimize therapeutic approaches, and the scientific community to advance metabolic research. The careful balance between progressive dose increases and adequate adaptation time represents the cornerstone of successful tirzepatide application across research and clinical contexts.
For researchers ready to begin or expand their work with tirzepatide, contact PEPTIDE PRO for research-grade peptides, comprehensive product information, and expert support for your experimental protocols.
References
[1] Frias JP, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. New England Journal of Medicine. 2021;385(6):503-515.
[2] U.S. Food and Drug Administration. Tirzepatide Prescribing Information. 2022.
[3] Rosenstock J, et al. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet. 2021;398(10295):143-155.
[4] Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. New England Journal of Medicine. 2022;387(3):205-216.
[5] Dahl D, et al. Effect of Subcutaneous Tirzepatide vs Placebo Added to Titrated Insulin Glargine on Glycemic Control in Patients With Type 2 Diabetes: The SURPASS-5 Randomized Clinical Trial. JAMA. 2022;327(6):534-545.
[6] European Medicines Agency. Assessment Report for Tirzepatide. 2022.
[7] Ludvik B, et al. Once-weekly tirzepatide versus once-daily insulin degludec as add-on to metformin with or without SGLT2 inhibitors in patients with type 2 diabetes (SURPASS-3): a randomised, open-label, parallel-group, phase 3 trial. Lancet. 2021;398(10300):583-598.
