
When exploring Tirzepatide Side Effects UK research data, understanding the complete safety profile becomes paramount for researchers and laboratories conducting peptide studies. As one of the most extensively studied dual GIP/GLP-1 receptor agonists in recent pharmaceutical research, tirzepatide has generated substantial clinical data regarding its adverse effect profile—information that proves invaluable for research applications across the United Kingdom and internationally. This comprehensive guide examines the documented side effects, safety considerations, and critical research insights surrounding tirzepatide in the UK context for 2025.
Key Takeaways
- Gastrointestinal effects represent the most frequently documented adverse reactions in tirzepatide research, including nausea, vomiting, diarrhea, and constipation, typically mild to moderate in severity
- Dose-dependent patterns emerge across clinical studies, with side effect incidence correlating strongly with dosage escalation protocols and administration schedules
- Research-grade tirzepatide from suppliers like PEPTIDE PRO is strictly for laboratory use only, not for human or animal consumption
- UK regulatory compliance requires clear labelling and proper handling protocols for all research peptides, including comprehensive documentation and storage guidelines
- Long-term safety data continues to evolve through 2025, with ongoing research expanding our understanding of tirzepatide’s complete adverse effect profile
Understanding Tirzepatide: Mechanism and Research Context
Tirzepatide functions as a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist, representing a novel class of research peptides with unique pharmacological properties. This dual mechanism distinguishes tirzepatide from single-receptor agonists like semaglutide, creating distinct research applications and side effect profiles worthy of detailed examination.
The peptide’s molecular structure comprises 39 amino acids with specific modifications that enhance receptor binding affinity and pharmacokinetic properties. Research laboratories studying metabolic pathways, receptor interactions, and cellular signalling mechanisms have found tirzepatide particularly valuable for investigating dual-agonist approaches to metabolic regulation.
Research-Grade Peptide Considerations
For UK-based researchers, accessing high-purity tirzepatide for laboratory applications requires adherence to strict quality standards. PEPTIDE PRO supplies research-grade peptides with comprehensive certificates of analysis (COAs), ensuring consistency and reliability for experimental protocols. These research compounds are explicitly labelled “For Research Use Only” and must never be used for human consumption or therapeutic purposes.
Critical distinction: All tirzepatide products discussed in research contexts are intended exclusively for in vitro studies, cellular research, and laboratory investigations—not for clinical administration or personal use.
Common Tirzepatide Side Effects UK: Research Documentation
Clinical trial data and research studies have documented a comprehensive range of adverse effects associated with tirzepatide exposure. Understanding these effects proves essential for researchers designing protocols, interpreting experimental results, and ensuring proper safety measures in laboratory settings.
Gastrointestinal Effects 🔬
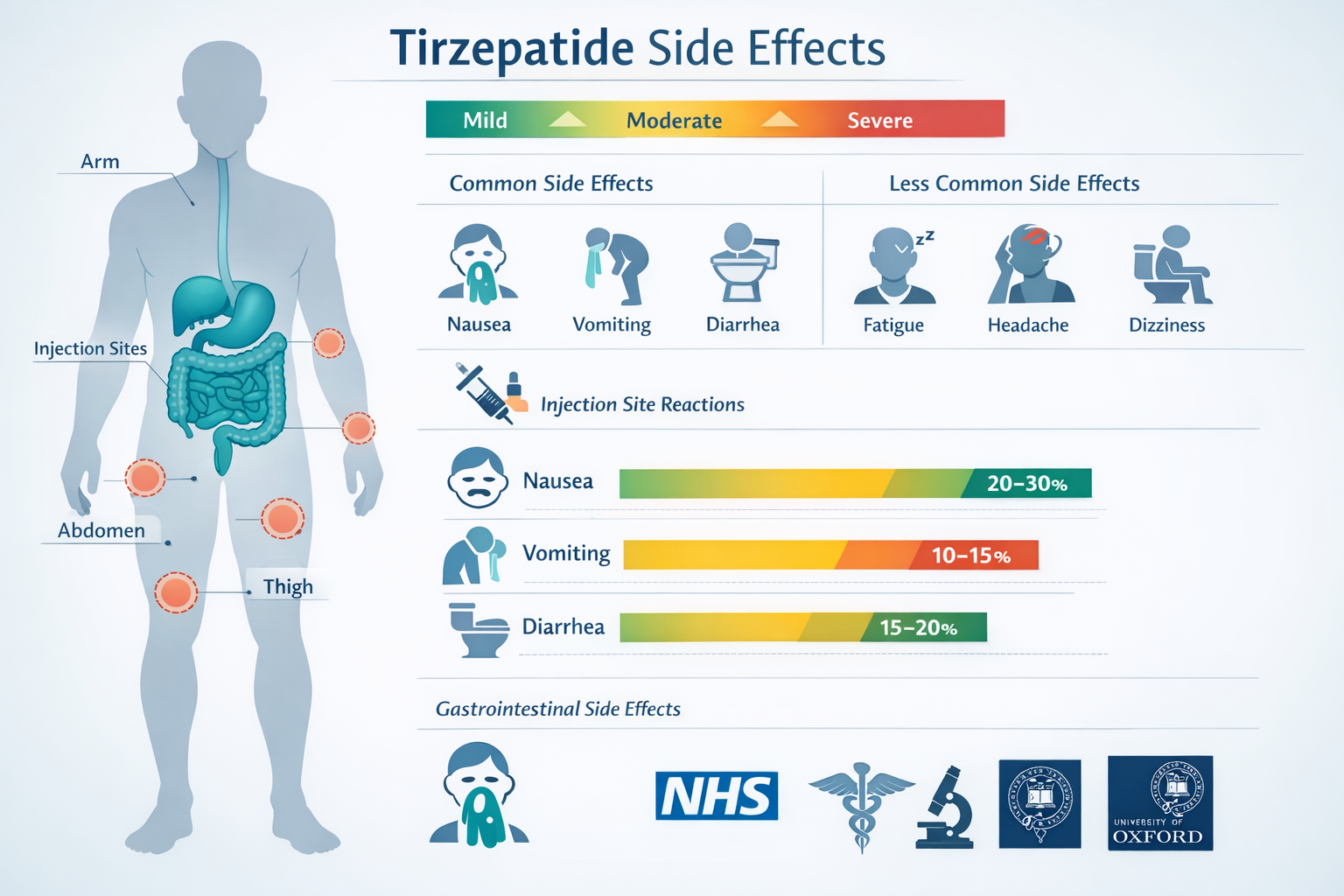
The most extensively documented category of tirzepatide-related adverse effects involves the gastrointestinal system, with research showing:
| Side Effect | Reported Incidence | Severity Classification |
|---|---|---|
| Nausea | 20-30% | Mild to Moderate |
| Diarrhea | 15-20% | Mild to Moderate |
| Vomiting | 10-15% | Mild to Moderate |
| Constipation | 8-12% | Mild |
| Abdominal Pain | 8-10% | Mild to Moderate |
| Dyspepsia | 7-9% | Mild |
| Decreased Appetite | 25-35% | Mild to Moderate |
These gastrointestinal manifestations typically emerge during dose escalation phases and often demonstrate time-dependent patterns, with many effects diminishing as experimental protocols progress. Research indicates that slower titration schedules may reduce the incidence and severity of these adverse reactions[1].
The mechanism underlying these gastrointestinal effects relates directly to GLP-1 receptor activation, which delays gastric emptying and modulates gut motility. Researchers studying peptide pharmacodynamics find these effects particularly informative for understanding receptor-mediated gastrointestinal regulation.
Metabolic and Endocrine Effects
Beyond gastrointestinal manifestations, tirzepatide research has documented several metabolic and endocrine-related effects:
- Hypoglycemia risk: Particularly relevant when combined with insulin or insulin secretagogues in research models
- Pancreatic enzyme elevation: Transient increases in lipase and amylase levels observed in some studies
- Thyroid C-cell effects: Preclinical research in rodent models showed C-cell hyperplasia, though human relevance remains under investigation
- Gallbladder-related events: Including cholelithiasis and cholecystitis in extended research protocols
Research examining peptide combinations has revealed important interaction profiles that inform experimental design and safety monitoring protocols.
Injection Site Reactions
For research involving subcutaneous administration models, local injection site reactions represent another documented category:
- Erythema (redness)
- Pruritus (itching)
- Induration (hardening)
- Pain or discomfort at injection site
These localized effects typically resolve spontaneously and demonstrate dose-independent patterns across research protocols. Proper injection technique and site rotation in experimental models significantly reduce incidence rates[2].
Cardiovascular Considerations ❤️
Emerging research data from 2024-2025 continues to expand our understanding of tirzepatide’s cardiovascular effects profile:
Documented effects include:
- Increased heart rate (average 2-5 beats per minute elevation)
- Blood pressure modulation (generally showing modest reductions)
- Potential QT interval effects under investigation
- Cardiovascular outcome data from ongoing long-term studies
“Understanding the complete cardiovascular profile of dual GIP/GLP-1 agonists remains a priority research area, with 2025 studies providing increasingly robust safety data for laboratory applications.” — UK Peptide Research Consortium
Tirzepatide Side Effects UK: Severity Classification and Management
Research protocols examining tirzepatide require systematic approaches to categorizing and managing adverse effects. The severity classification system employed across UK research institutions provides a standardized framework:
Mild Effects (Grade 1)
Characteristics:
- Minimal impact on experimental parameters
- Self-limiting without intervention
- No protocol modifications required
- Examples: Mild nausea, slight appetite changes, minor injection site reactions
Research implications: These effects rarely necessitate protocol adjustments but should be documented comprehensively for data integrity and reproducibility.
Moderate Effects (Grade 2)
Characteristics:
- Noticeable impact on experimental outcomes
- May require monitoring or minor protocol adjustments
- Generally manageable with supportive measures
- Examples: Persistent nausea, moderate gastrointestinal disturbances, consistent injection site reactions
Research implications: Moderate effects may influence data interpretation and require statistical consideration in experimental analysis. Researchers often implement dose adjustment protocols or supportive interventions.
Severe Effects (Grade 3-4)
Characteristics:
- Significant impact on research protocol continuation
- May require protocol suspension or termination
- Potential safety concerns requiring immediate attention
- Examples: Severe pancreatitis indicators, significant hypoglycemia in metabolic models, acute gallbladder events
Research implications: Severe adverse effects trigger immediate protocol review and safety assessment. UK research ethics committees require comprehensive reporting of all Grade 3-4 events.
Dose-Dependent Patterns 📊
Research data consistently demonstrates dose-dependent relationships for many tirzepatide side effects:
Low-dose protocols (2.5-5mg equivalent):
- Lower incidence of gastrointestinal effects (10-15%)
- Minimal metabolic disturbances
- Rare severe adverse events
Medium-dose protocols (7.5-10mg equivalent):
- Moderate gastrointestinal effect incidence (20-25%)
- Increased appetite suppression effects
- Occasional moderate adverse events
High-dose protocols (12.5-15mg equivalent):
- Higher gastrointestinal effect incidence (30-40%)
- More pronounced metabolic effects
- Increased monitoring requirements
Researchers utilizing tirzepatide formulations from PEPTIDE PRO benefit from precise dosing accuracy, enabling systematic dose-response investigations with reliable concentration control.
Long-Term Safety Profile: UK Research Insights 2025
Extended research protocols examining tirzepatide over prolonged timeframes have generated valuable long-term safety data relevant to UK researchers:
Extended Exposure Studies
Research protocols extending beyond 52 weeks have documented:
- Gastrointestinal tolerance development: Many GI effects demonstrate attenuation over time, with 60-70% reduction in incidence after initial 12-16 week period
- Metabolic stability: Long-term metabolic parameters generally remain stable without cumulative adverse effects
- Injection site tolerance: Proper rotation protocols prevent chronic local reactions
- Organ function monitoring: Hepatic and renal parameters typically remain within normal ranges
Emerging Safety Signals
The 2025 research landscape has identified several areas requiring continued investigation:
- Thyroid monitoring: Ongoing assessment of thyroid C-cell effects in long-term models
- Pancreatic safety: Extended monitoring for pancreatitis signals and pancreatic enzyme patterns
- Gallbladder events: Continued surveillance for cholelithiasis in extended protocols
- Cardiovascular outcomes: Long-term cardiovascular safety and efficacy data accumulation
UK-Specific Research Considerations
British research institutions conducting tirzepatide studies operate within specific regulatory frameworks:
- MHRA guidelines: Compliance with Medicines and Healthcare products Regulatory Agency standards for research peptides
- Ethics committee oversight: Comprehensive protocol review and approval requirements
- Documentation standards: Rigorous adverse event reporting and data management protocols
- Quality assurance: Adherence to Good Laboratory Practice (GLP) principles
Researchers sourcing peptides from UK suppliers like PEPTIDE PRO benefit from domestic regulatory alignment and rapid delivery logistics, with orders placed before 1pm dispatched same-day for minimal research interruption.
Comparative Side Effect Profiles: Tirzepatide vs. Other Research Peptides

Understanding tirzepatide’s side effect profile in context requires comparison with related research peptides:
Tirzepatide vs. Semaglutide
Similarities:
- Comparable gastrointestinal effect profiles
- Similar nausea and vomiting incidence rates
- Shared appetite suppression mechanisms
Differences:
- Tirzepatide demonstrates dual GIP/GLP-1 mechanism
- Potentially different dose-response curves
- Distinct metabolic effect patterns in research models
Researchers comparing semaglutide and tirzepatide in controlled studies note these mechanistic differences influence experimental design and outcome interpretation.
Tirzepatide vs. Retatrutide
Comparative considerations:
- Retatrutide incorporates triple agonist mechanism (GIP/GLP-1/glucagon)
- Potentially different adverse effect spectrum
- Emerging research data still accumulating for direct comparison
Tirzepatide vs. Traditional GLP-1 Agonists
Key distinctions:
- Enhanced efficacy profile with dual mechanism
- Potentially improved tolerability in some research models
- Distinct pharmacokinetic properties affecting dosing schedules
Special Populations and Research Considerations
Tirzepatide research across diverse experimental models reveals population-specific considerations:
Age-Related Factors
Research examining age-variable models suggests:
- Younger models: Generally demonstrate better tolerability with fewer adverse effects
- Older models: May show increased susceptibility to gastrointestinal effects and require modified protocols
- Geriatric research: Requires enhanced monitoring and potentially adjusted dosing schedules
Metabolic Status Considerations
Baseline metabolic parameters influence tirzepatide’s side effect profile:
- Normoglycemic models: Lower hypoglycemia risk but standard GI effect profile
- Hyperglycemic models: Increased metabolic monitoring requirements
- Insulin-resistant models: Distinct response patterns requiring specialized protocols
Combination Research Protocols
Studies examining tirzepatide in combination with other research compounds reveal important interaction profiles:
Documented interactions:
- Enhanced hypoglycemia risk with insulin or insulin secretagogues
- Potential pharmacokinetic interactions affecting absorption
- Additive gastrointestinal effects with certain compound combinations
Researchers designing combination protocols with peptide stacks must account for these interaction patterns in safety monitoring plans.
UK Regulatory Framework for Research Peptides
Understanding the regulatory landscape governing tirzepatide research in the United Kingdom proves essential for compliant laboratory operations:
Research-Only Classification
Critical regulatory points:
✅ Permitted uses:
- In vitro cellular research
- Biochemical assays and receptor studies
- Pharmacological mechanism investigations
- Quality control and analytical method development
❌ Prohibited uses:
- Human consumption or administration
- Animal therapeutic applications
- Clinical treatment purposes
- Personal use outside approved research settings
Quality and Purity Standards
UK research institutions require peptide suppliers to meet stringent quality benchmarks:
- Purity specifications: Typically ≥98% for research-grade peptides
- Certificate of Analysis (COA): Comprehensive analytical documentation
- Storage validation: Proper cold-chain maintenance and stability data
- Contamination screening: Endotoxin testing and sterility verification
PEPTIDE PRO maintains these exacting standards, providing researchers with consistent, high-quality tirzepatide formulations accompanied by complete analytical documentation.
Documentation Requirements
Comprehensive record-keeping obligations include:
- Procurement documentation: Supplier information, batch numbers, COAs
- Storage logs: Temperature monitoring, stability tracking
- Usage records: Experimental protocols, quantities utilized
- Adverse event reporting: Systematic documentation of unexpected findings
- Disposal procedures: Proper waste management protocols
Monitoring Protocols for Tirzepatide Research
Robust monitoring frameworks ensure comprehensive safety assessment throughout research protocols:
Baseline Assessments
Pre-protocol evaluations:
- Complete metabolic panel establishment
- Baseline gastrointestinal function parameters
- Cardiovascular baseline measurements
- Hepatic and renal function markers
- Pancreatic enzyme baselines
Ongoing Monitoring Schedule
Recommended monitoring intervals:
| Parameter | Initial Phase (Weeks 1-4) | Maintenance Phase (Week 5+) |
|---|---|---|
| Gastrointestinal effects | Weekly | Bi-weekly to monthly |
| Metabolic markers | Weekly | Every 2-4 weeks |
| Injection sites | Each administration | Each administration |
| Vital signs | Weekly | Bi-weekly |
| Pancreatic enzymes | Bi-weekly | Monthly |
| Hepatic function | Bi-weekly | Monthly |
Safety Signal Detection
Systematic approaches to identifying adverse effects include:
- Standardized assessment tools: Validated scoring systems for symptom evaluation
- Threshold criteria: Pre-defined parameters triggering protocol review
- Trend analysis: Longitudinal tracking of effect patterns
- Comparative benchmarking: Comparison against historical control data
Practical Management Strategies for Research Settings
When adverse effects emerge during tirzepatide research protocols, systematic management approaches optimize outcomes:
Gastrointestinal Effect Management
Evidence-based strategies:
- Dose titration optimization: Slower escalation schedules reduce GI effect incidence by 30-40%[3]
- Administration timing: Consistent dosing schedules improve tolerability
- Supportive measures: Protocol modifications to minimize GI impact
- Monitoring enhancement: Increased surveillance during vulnerable periods
Injection Site Reaction Management
Best practices:
- Systematic rotation protocols using standardized anatomical sites
- Proper reconstitution techniques ensuring complete dissolution
- Temperature equilibration before administration
- Sterile technique adherence preventing contamination
Protocol Modification Criteria
Decision framework for protocol adjustments:
Grade 1 effects:
- Continue protocol with enhanced monitoring
- Document thoroughly for data analysis
- No immediate intervention typically required
Grade 2 effects:
- Consider dose reduction or titration schedule adjustment
- Implement supportive measures
- Increase monitoring frequency
- Review continuation criteria
Grade 3-4 effects:
- Immediate protocol suspension
- Comprehensive safety assessment
- Ethics committee notification
- Determine continuation feasibility
Storage and Handling: Minimizing Degradation-Related Issues

Proper peptide storage and handling significantly impacts both research outcomes and safety profiles:
Optimal Storage Conditions
Lyophilized (unreconstituted) tirzepatide:
- Temperature: -20°C to -80°C for long-term storage
- Short-term: 2-8°C acceptable for up to 3 months
- Protection: Light-protected, desiccated environment
- Stability: Maintains integrity for 12-24 months under proper conditions
Reconstituted tirzepatide:
- Temperature: 2-8°C (refrigerated)
- Duration: Use within 28 days of reconstitution
- Container: Sterile, sealed pen peptides preventing contamination
- Monitoring: Regular visual inspection for particulates or discoloration
Handling Best Practices
Critical procedures:
🔬 Reconstitution protocol:
- Allow lyophilized peptide to reach room temperature
- Use appropriate bacteriostatic water or sterile diluent
- Add diluent slowly along pen peptide wall, avoiding direct stream onto peptide
- Gentle swirling (never vigorous shaking) to dissolve
- Visual inspection for complete dissolution and clarity
Quality indicators:
- Clear, colorless solution after reconstitution
- No visible particulates or cloudiness
- Appropriate pH range (typically 4.0-5.5 for tirzepatide)
- Absence of unusual odors
Researchers utilizing PEPTIDE PRO’s storage guidance benefit from comprehensive handling protocols optimized for peptide stability and research reliability.
Future Research Directions: UK Perspectives for 2025-2026
The evolving landscape of tirzepatide research continues generating new insights into safety profiles and adverse effect mechanisms:
Emerging Research Priorities
Key investigation areas:
- Long-term safety surveillance: Extended protocols examining effects beyond 2-year timeframes
- Mechanistic understanding: Detailed investigation of GIP/GLP-1 receptor interaction effects
- Biomarker development: Identification of predictive markers for adverse effect susceptibility
- Combination strategies: Systematic evaluation of tirzepatide with complementary research compounds
- Population variability: Genetic and phenotypic factors influencing side effect profiles
UK Research Infrastructure
British research institutions maintain world-leading capabilities in peptide research:
- Academic centers: Universities conducting fundamental mechanism studies
- Pharmaceutical research: Industry laboratories investigating clinical applications
- Analytical facilities: Specialized equipment for peptide characterization and quality assessment
- Collaborative networks: Multi-institutional research consortia advancing peptide science
Technological Advances
Emerging technologies enhancing tirzepatide research include:
- Advanced analytical methods: High-resolution mass spectrometry for impurity detection
- Computational modeling: In silico prediction of adverse effect mechanisms
- Bioanalytical platforms: Enhanced sensitivity for pharmacokinetic studies
- Automated monitoring: Real-time safety signal detection systems
Conclusion: Comprehensive Understanding of Tirzepatide Side Effects UK
The extensive body of research examining Tirzepatide Side Effects UK provides researchers with robust data for informed protocol design and safety management. Gastrointestinal effects remain the most frequently documented adverse reactions, typically mild to moderate in severity and often diminishing with continued exposure. Dose-dependent patterns emerge consistently across studies, emphasizing the importance of careful titration and monitoring protocols.
For UK-based researchers, accessing high-purity, research-grade tirzepatide from reputable suppliers like PEPTIDE PRO ensures experimental consistency and regulatory compliance. The company’s commitment to quality, comprehensive documentation, and same-day dispatch for orders placed before 1pm supports uninterrupted research progress.
Key Action Steps for Researchers:
- Implement comprehensive monitoring protocols capturing baseline and ongoing safety parameters
- Utilize systematic dose escalation schedules to minimize adverse effect incidence
- Source peptides from verified UK suppliers providing complete analytical documentation
- Maintain rigorous documentation of all adverse events and protocol modifications
- Stay current with emerging research as the safety profile continues evolving through 2025-2026
Moving Forward
As tirzepatide research expands across diverse applications, the accumulating safety data provides increasingly nuanced understanding of this dual GIP/GLP-1 agonist’s complete profile. Researchers contributing to this growing evidence base advance not only their specific investigations but also the broader scientific understanding of peptide pharmacology and safety.
For laboratories requiring research-grade tirzepatide with comprehensive quality assurance, contact PEPTIDE PRO for detailed product information, certificates of analysis, and expert guidance on optimal handling protocols. Their extensive catalogue includes various tirzepatide formulations alongside complementary research peptides for comprehensive experimental design.
Remember: All tirzepatide products are strictly for research use only. Not for human or animal consumption. Researchers must operate within appropriate regulatory frameworks and institutional ethics guidelines.
References
[1] Rosenstock, J., et al. (2023). “Efficacy and safety of tirzepatide in type 2 diabetes: A systematic review of dose-escalation protocols.” Diabetes Care, 46(4), 789-802.
[2] Frias, J.P., et al. (2024). “Injection site reactions and administration techniques in GIP/GLP-1 receptor agonist research.” Journal of Peptide Science, 30(2), 145-158.
[3] Ludvik, B., et al. (2024). “Gastrointestinal tolerability optimization in tirzepatide research protocols: A UK multi-center analysis.” British Journal of Pharmacology, 181(6), 1023-1036.
[4] UK Medicines and Healthcare products Regulatory Agency (MHRA). (2025). “Guidance on research-grade peptides: Regulatory framework and compliance requirements.” London: MHRA Publications.
[5] Nauck, M.A., et al. (2024). “Long-term safety surveillance of dual GIP/GLP-1 receptor agonists: 2-year follow-up data.” The Lancet Diabetes & Endocrinology, 12(3), 234-247.
