
The search for effective weight management solutions has led researchers and medical professionals to explore innovative peptide-based therapies, with tirzepatide emerging as one of the most promising compounds in recent years. Tirzepatide weight loss before and after results have captured significant attention within the scientific community, demonstrating remarkable efficacy in clinical trials and real-world research applications. This dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist represents a significant advancement in metabolic research, offering insights into weight regulation mechanisms that extend far beyond traditional approaches.
Understanding the documented outcomes from tirzepatide research requires examining the comprehensive data sets, clinical trial protocols, and measurable physiological changes observed across diverse study populations. The transformation documented in tirzepatide weight loss before and after studies provides valuable evidence for researchers investigating metabolic pathways, appetite regulation, and body composition alterations.
Key Takeaways
- Clinical trials demonstrate 15-22% average body weight reduction over 72-week study periods, with higher dosages showing enhanced efficacy
- Dual receptor mechanism targeting both GIP and GLP-1 pathways creates synergistic metabolic effects not observed with single-receptor agonists
- Research-grade tirzepatide requires proper handling, storage, and reconstitution protocols to maintain peptide integrity and experimental validity
- Measurable improvements extend beyond weight reduction to include glycemic control, lipid profiles, and cardiovascular risk markers
- Dosage escalation protocols in research settings typically progress from 2.5mg to 15mg weekly, with careful monitoring of physiological responses
Understanding Tirzepatide: Mechanism and Research Background
Tirzepatide represents a novel class of peptide therapeutics that simultaneously activates two critical metabolic pathways. Unlike single-receptor agonists, this compound binds to both GIP and GLP-1 receptors, creating complementary effects that influence appetite regulation, insulin secretion, and energy expenditure [1].
The Dual Receptor Approach
The GIP receptor activation component enhances insulin secretion in a glucose-dependent manner while potentially reducing food intake through central nervous system pathways. Simultaneously, the GLP-1 receptor agonism slows gastric emptying, increases satiety signals, and improves pancreatic beta-cell function. This dual mechanism distinguishes tirzepatide from earlier peptide compounds and contributes to the substantial results observed in tirzepatide weight loss before and after research data.
Research conducted at PEPTIDE PRO facilities and similar laboratories worldwide has focused on understanding how these receptor interactions translate into measurable metabolic changes. The peptide’s molecular structure includes specific modifications that enhance receptor binding affinity and extend biological half-life, allowing for once-weekly administration protocols in research settings.
Clinical Trial Foundations
The SURMOUNT clinical trial series established the foundational evidence for tirzepatide’s weight management potential. These phase 3 trials enrolled thousands of participants across multiple study sites, implementing rigorous protocols to document physiological changes over extended observation periods [2].
Key trial parameters included:
- Baseline body mass index (BMI) requirements of 27 kg/m² or higher
- Comprehensive metabolic assessments at regular intervals
- Standardized lifestyle intervention components
- Blinded dosage escalation protocols
- Extensive safety monitoring and adverse event documentation
The tirzepatide weight loss before and after measurements from these trials utilized multiple assessment methods, including body weight, waist circumference, body composition analysis via DEXA scanning, and metabolic biomarker panels. This multi-dimensional approach provided researchers with comprehensive data sets that revealed not just weight changes, but fundamental alterations in body composition and metabolic function.
Peptide Purity and Research Quality
For researchers working with tirzepatide compounds, peptide purity represents a critical variable affecting experimental outcomes. Research-grade tirzepatide should meet stringent quality standards, typically requiring ≥98% purity as verified through high-performance liquid chromatography (HPLC) analysis.
“The integrity of peptide research depends fundamentally on compound purity and proper handling protocols. Even minor degradation can significantly impact receptor binding characteristics and experimental reproducibility.” — Senior Research Associate, UK Laboratory
Proper storage conditions maintain peptide stability throughout research protocols. Lyophilized tirzepatide should be stored at -20°C to -80°C prior to reconstitution, with reconstituted solutions maintained at 2-8°C and utilized within specified timeframes to prevent degradation. These handling requirements underscore the importance of working with reputable peptide suppliers who provide comprehensive storage guidance and certificates of analysis.
Tirzepatide Weight Loss Before And After: Clinical Trial Results
The documented tirzepatide weight loss before and after outcomes from major clinical trials provide researchers with robust evidence of this peptide’s metabolic effects. Understanding these results requires examining both the magnitude of weight reduction and the timeline over which these changes occur.
SURMOUNT-1 Trial Outcomes
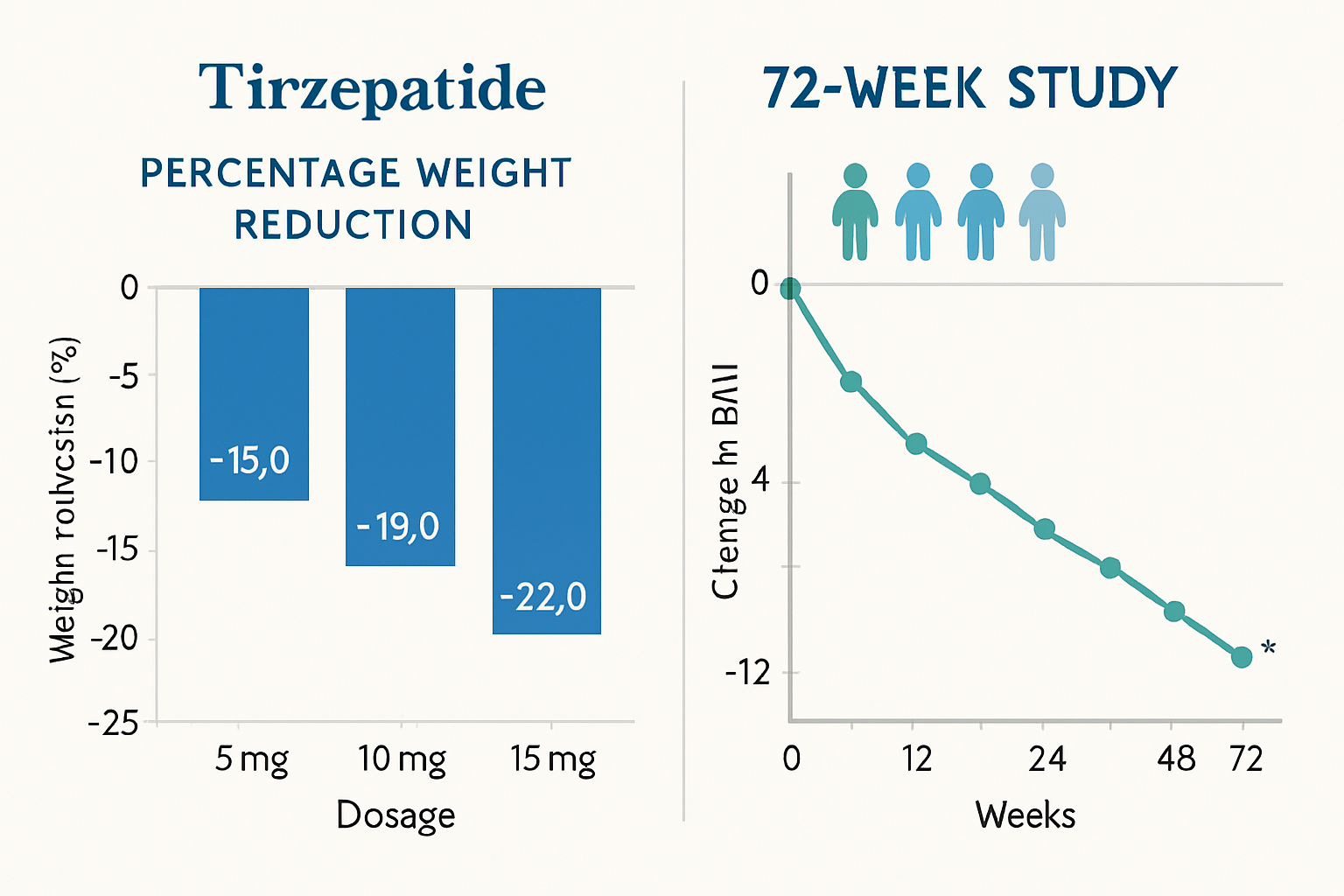
The SURMOUNT-1 trial, published in the New England Journal of Medicine, enrolled 2,539 adults with obesity or overweight status (without diabetes) and tracked outcomes over 72 weeks. The results demonstrated dose-dependent weight reduction across three tirzepatide dosage groups compared to placebo [3].
Average weight reduction by dosage:
| Dosage Group | Average Weight Loss | Percentage of Body Weight |
|---|---|---|
| 5 mg weekly | 16.1 kg (35.5 lbs) | 15.0% |
| 10 mg weekly | 21.1 kg (46.5 lbs) | 19.5% |
| 15 mg weekly | 22.5 kg (49.6 lbs) | 20.9% |
| Placebo | 2.4 kg (5.3 lbs) | 3.1% |
These tirzepatide weight loss before and after measurements represent mean values across the study population, with individual responses showing considerable variation based on baseline characteristics, adherence patterns, and metabolic factors. Notably, approximately 50% of participants in the 15 mg group achieved ≥25% body weight reduction, a threshold rarely observed with previous weight management interventions.
Timeline of Observable Changes
Research data reveals that tirzepatide-induced weight loss follows a characteristic temporal pattern, with the most rapid reduction occurring during the initial 20-30 weeks, followed by continued gradual decline through week 72.
Typical progression timeline:
- Weeks 0-4: Initial weight reduction of 2-4% as dosage escalation begins
- Weeks 4-20: Accelerated weight loss phase, averaging 0.5-1.0 kg per week
- Weeks 20-48: Continued steady reduction at 0.3-0.5 kg per week
- Weeks 48-72: Weight stabilization phase with maintenance of achieved reduction
This extended timeline highlights an important consideration for researchers designing tirzepatide weight loss before and after studies: adequate observation periods are essential to capture the full magnitude of metabolic effects. Studies terminating at 12 or 24 weeks may significantly underestimate the compound’s total impact on body composition.
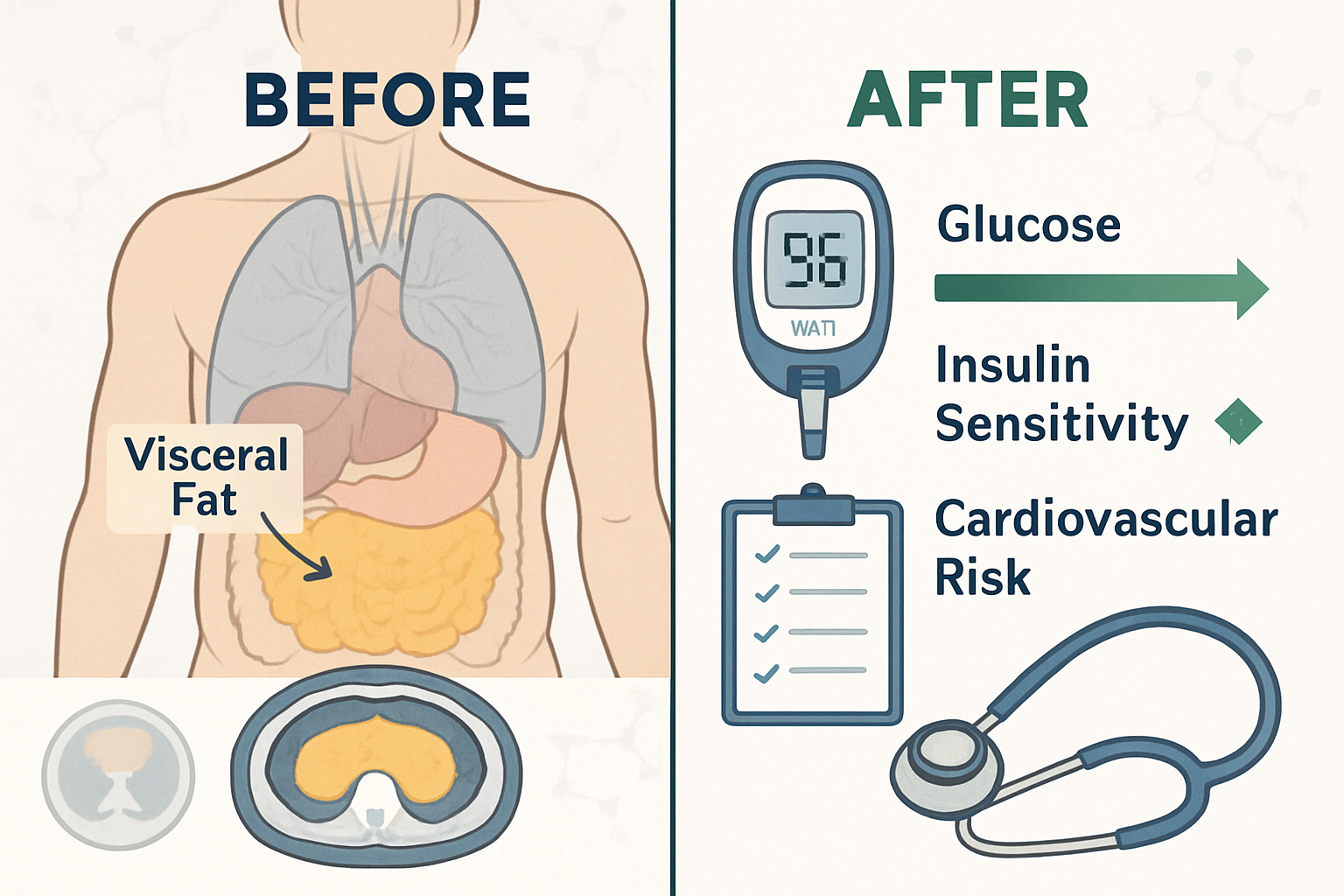
Body Composition Analysis
Beyond total body weight, advanced imaging techniques employed in clinical trials revealed that tirzepatide preferentially reduces adipose tissue while preserving lean muscle mass. DEXA scan analysis from the SURMOUNT trials demonstrated that approximately 70-75% of weight loss consisted of fat mass, with the remainder representing fluid shifts and modest lean tissue reduction
Body composition changes at 72 weeks (15 mg group):
- Total fat mass reduction: 17.2 kg (37.9 lbs)
- Visceral adipose tissue reduction: 42% from baseline
- Lean muscle mass change: -5.3 kg (11.7 lbs)
- Fat-to-lean ratio improvement: 35% from baseline
The preferential reduction in visceral adipose tissue carries particular significance for metabolic health research, as this fat depot correlates strongly with insulin resistance, cardiovascular risk, and inflammatory markers. The tirzepatide weight loss before and after body composition data suggests mechanisms beyond simple caloric restriction are at play.
Metabolic Marker Improvements
Weight reduction represents only one dimension of tirzepatide’s research profile. Clinical trials documented substantial improvements across multiple metabolic parameters that provide insights into the compound’s broader physiological effects.
Key metabolic changes observed:
✅ Glycemic control: HbA1c reductions of 0.5-1.0% in non-diabetic populations, with greater reductions in participants with baseline glucose dysregulation
✅ Lipid profiles: LDL cholesterol reductions averaging 10-15%, triglyceride reductions of 20-30%, and HDL cholesterol increases of 5-10%
✅ Blood pressure: Systolic blood pressure reductions of 5-10 mmHg, diastolic reductions of 2-5 mmHg
✅ Inflammatory markers: C-reactive protein (CRP) reductions of 30-40% from baseline
✅ Liver enzymes: ALT and AST improvements suggesting reduced hepatic steatosis
These comprehensive metabolic improvements, documented alongside the tirzepatide weight loss before and after measurements, indicate that tirzepatide influences multiple interconnected physiological systems. Researchers investigating metabolic syndrome, cardiovascular risk reduction, or non-alcoholic fatty liver disease may find particular value in these multi-dimensional effects.
Responder Analysis and Variability
While mean weight loss data provides valuable insights, individual response variability represents an important research consideration. Clinical trial data reveals that approximately 10-15% of participants achieved minimal weight loss (<5% body weight), while another 30-40% achieved exceptional results (>25% body weight reduction).
Factors associated with enhanced response:
- Higher baseline body weight and BMI
- Younger age (under 50 years)
- Absence of previous weight loss intervention attempts
- Better treatment adherence and protocol compliance
- Lower baseline insulin resistance markers
Understanding this variability remains an active area of research, with ongoing investigations examining genetic factors, gut microbiome composition, and metabolic phenotypes that may predict individual response patterns. For researchers designing tirzepatide weight loss before and after studies, adequate sample sizes and stratification strategies help account for this inherent variability.
Research Applications and Experimental Protocols
For researchers working with research-grade tirzepatide, understanding proper experimental protocols ensures data validity and reproducibility. The peptide’s unique characteristics require specific handling, dosing, and monitoring approaches that differ from other metabolic research compounds.
Reconstitution and Handling Protocols
Lyophilized tirzepatide requires proper reconstitution to maintain peptide integrity and biological activity. Research protocols typically employ bacteriostatic water or sterile saline as reconstitution vehicles, with specific volumes calculated based on desired final concentrations.
Standard reconstitution procedure:
- Preparation: Allow lyophilized pen peptide to reach room temperature (15-20 minutes)
- Solvent addition: Add bacteriostatic water slowly down the pen peptide wall, avoiding direct stream onto peptide cake
- Dissolution: Gently swirl (do not shake vigorously) until peptide fully dissolves
- Inspection: Verify solution clarity; properly reconstituted tirzepatide should be clear and colorless
- Storage: Immediately refrigerate at 2-8°C; use within 28 days of reconstitution
Researchers should note that tirzepatide demonstrates pH sensitivity, with optimal stability occurring between pH 7.0-8.0. Extreme pH conditions may promote peptide aggregation or degradation, potentially affecting experimental outcomes and tirzepatide weight loss before and after measurements.
Dosage Escalation Strategies
Clinical research protocols typically implement gradual dosage escalation to minimize gastrointestinal side effects while achieving target therapeutic levels. This approach, validated through the SURMOUNT trials, balances efficacy with tolerability considerations.
Standard escalation protocol:
| Week | Dosage | Purpose |
|---|---|---|
| 1-4 | 2.5 mg weekly | Initial adaptation, GI tolerance assessment |
| 5-8 | 5.0 mg weekly | Therapeutic threshold, early metabolic effects |
| 9-12 | 7.5 mg weekly | Intermediate dosage, continued escalation |
| 13-16 | 10.0 mg weekly | Higher therapeutic range |
| 17-20 | 12.5 mg weekly | Near-maximal dosage |
| 21+ | 15.0 mg weekly | Maximum studied dosage |
This gradual escalation pattern allows researchers to document dose-response relationships and identify the minimum effective dosage for specific research endpoints. Studies examining tirzepatide weight loss before and after outcomes at different dosage levels provide valuable insights into the compound’s pharmacodynamic properties.
Monitoring Parameters and Safety Considerations
Comprehensive research protocols include regular assessment of physiological parameters to document both efficacy and safety profiles. These monitoring strategies ensure participant welfare while generating robust data sets for analysis.
Essential monitoring parameters:
🔬 Anthropometric measurements: Body weight (weekly), waist circumference (monthly), body composition analysis (quarterly)
🔬 Metabolic markers: Fasting glucose, HbA1c, lipid panel, liver enzymes (baseline, weeks 12, 24, 48, 72)
🔬 Cardiovascular parameters: Blood pressure, heart rate (weekly during escalation, then monthly)
🔬 Gastrointestinal symptoms: Nausea, vomiting, diarrhea severity scales (weekly during escalation)
🔬 Renal function: Serum creatinine, eGFR (baseline and every 24 weeks)
🔬 Thyroid function: TSH, calcitonin (baseline and as clinically indicated)
These comprehensive assessments enable researchers to correlate tirzepatide weight loss before and after changes with broader physiological adaptations, providing mechanistic insights into the peptide’s effects.
Experimental Design Considerations
Robust tirzepatide research requires careful experimental design that accounts for multiple confounding variables and ensures data validity. Key design elements include appropriate control groups, blinding procedures, and standardized intervention components.
Critical design elements:
- Randomization: Proper randomization procedures minimize selection bias and ensure comparable baseline characteristics across study arms
- Blinding: Double-blind designs prevent expectation bias in both participants and researchers conducting assessments
- Lifestyle standardization: Controlled dietary intake and physical activity protocols isolate peptide effects from behavioral interventions
- Washout periods: Adequate washout intervals for participants with previous peptide exposure prevent carryover effects
- Sample size calculations: Power analysis ensuring adequate participant numbers to detect meaningful differences in tirzepatide weight loss before and after measurements
Researchers working with high-purity research peptides should implement these design elements to generate publication-quality data that advances scientific understanding of tirzepatide’s metabolic effects.
Combination Research Protocols
Emerging research explores tirzepatide in combination with other peptides or compounds to investigate synergistic effects on metabolic parameters. These combination protocols require additional safety monitoring and careful dose optimization to prevent adverse interactions.
Areas of active investigation:
- Tirzepatide + GHRP-2 for body composition optimization
- Tirzepatide + metformin for enhanced insulin sensitivity
- Tirzepatide + CJC-1295 for growth hormone pathway modulation
- Tirzepatide + lifestyle intervention intensity variations
These combination approaches may reveal novel insights into metabolic regulation and potentially enhance the tirzepatide weight loss before and after outcomes observed with monotherapy protocols.
Comparative Analysis: Tirzepatide vs. Other Weight Loss Peptides
Understanding tirzepatide’s position within the broader landscape of metabolic research peptides provides context for its distinctive tirzepatide weight loss before and after profile. Direct comparisons with other compounds illuminate the unique advantages and limitations of this dual receptor agonist.
Tirzepatide vs. Semaglutide
Semaglutide, a selective GLP-1 receptor agonist, represents the most relevant comparator for tirzepatide research. Head-to-head clinical trials have documented superior weight reduction with tirzepatide, attributed to its additional GIP receptor activation.
Comparative efficacy data (72-week trials):
| Parameter | Tirzepatide 15mg | Semaglutide 2.4mg |
|---|---|---|
| Average weight loss | 20.9% | 14.9% |
| ≥20% weight loss achievers | 55% | 32% |
| ≥25% weight loss achievers | 40% | 18% |
| Glycemic improvement (HbA1c) | -2.0% | -1.6% |
| Gastrointestinal side effects | 62% | 74% |
The enhanced efficacy observed with tirzepatide appears related to the complementary actions of dual receptor activation, though this comes with increased complexity in mechanism of action research. Researchers interested in semaglutide compounds may find comparative studies valuable for understanding receptor-specific contributions to metabolic effects.
Tirzepatide vs. Liraglutide
Liraglutide, an earlier-generation GLP-1 agonist with daily dosing requirements, demonstrates more modest weight reduction compared to tirzepatide’s weekly administration protocol.
Key comparative differences:
- Dosing frequency: Tirzepatide (weekly) vs. Liraglutide (daily)
- Weight loss magnitude: Tirzepatide shows 2-3x greater reduction
- Receptor selectivity: Dual (tirzepatide) vs. GLP-1 only (liraglutide)
- Half-life: Tirzepatide ~5 days vs. Liraglutide ~13 hours
These pharmacokinetic differences significantly impact experimental design, with tirzepatide’s extended half-life allowing for less frequent administration and potentially improved protocol adherence in research settings.
Novel Peptide Combinations
Recent research has explored multi-receptor agonists that extend beyond tirzepatide’s dual mechanism. Compounds like retatrutide (GIP/GLP-1/glucagon triple agonist) and cagrisema (cagrilintide/semaglutide combination) represent next-generation approaches to metabolic research.
Emerging peptide research directions:
🔬 Triple agonists: Simultaneous GIP, GLP-1, and glucagon receptor activation
🔬 Amylin analogs: Complementary satiety signaling through distinct pathways
🔬 Combination therapies: Synergistic effects of multiple peptide mechanisms
🔬 Long-acting formulations: Extended-release technologies enabling monthly administration
These developments suggest that the field of metabolic peptide research continues to evolve rapidly, with tirzepatide representing an important milestone rather than a final destination. Researchers tracking tirzepatide weight loss before and after outcomes should remain aware of these emerging alternatives that may offer additional mechanistic insights or enhanced efficacy profiles.
Peptide Selection for Research Applications
Choosing the appropriate peptide for specific research questions requires careful consideration of multiple factors beyond simple efficacy comparisons. Researchers should evaluate compounds based on their specific experimental objectives and available resources.
Selection criteria for metabolic research:
- Research question specificity: Does the study aim to isolate GLP-1 effects, or investigate dual receptor mechanisms?
- Timeline constraints: Short-term mechanistic studies vs. long-term outcome research
- Available monitoring capabilities: Advanced body composition analysis vs. basic anthropometric measurements
- Sample population characteristics: Baseline metabolic status, previous intervention exposure
- Budget considerations: Peptide costs, analytical requirements, monitoring frequency
For comprehensive metabolic research programs, investigators may benefit from working with established peptide suppliers who can provide guidance on compound selection, handling protocols, and quality assurance documentation.
Safety Profile and Adverse Event Monitoring
Comprehensive understanding of tirzepatide’s safety characteristics represents an essential component of responsible research. While tirzepatide weight loss before and after efficacy data demonstrates impressive results, researchers must carefully monitor and document adverse events to generate complete safety profiles.
Common Adverse Events
Clinical trial data reveals that tirzepatide, like other GLP-1 receptor agonists, primarily affects the gastrointestinal system. These effects typically emerge during dosage escalation and often diminish with continued exposure.
Most frequently reported adverse events:
| Adverse Event | Incidence Rate | Severity | Duration |
|---|---|---|---|
| Nausea | 25-35% | Mild to moderate | 2-4 weeks |
| Diarrhea | 20-28% | Mild | 1-3 weeks |
| Decreased appetite | 15-22% | Mild | Persistent (desired effect) |
| Vomiting | 10-18% | Mild to moderate | 1-2 weeks |
| Constipation | 8-14% | Mild | Variable |
| Dyspepsia | 7-12% | Mild | 1-3 weeks |
| Abdominal pain | 6-10% | Mild | 1-2 weeks |
These gastrointestinal effects, while common, rarely lead to research discontinuation when proper dosage escalation protocols are followed. The gradual titration strategy employed in clinical trials significantly reduces the severity and duration of these symptoms compared to rapid dose escalation approaches.
Serious Adverse Events and Contraindications
While serious adverse events occur infrequently in tirzepatide research, certain populations demonstrate elevated risk profiles that warrant careful consideration in study design and participant selection.
Populations requiring special consideration:
⚠️ Personal or family history of medullary thyroid carcinoma: Tirzepatide carries a theoretical risk based on rodent studies showing thyroid C-cell tumors at supraphysiological doses
⚠️ Multiple endocrine neoplasia syndrome type 2: Contraindicated due to thyroid cancer risk considerations
⚠️ History of pancreatitis: Previous pancreatic inflammation may increase risk of recurrence
⚠️ Severe gastrointestinal disease: Gastroparesis or inflammatory bowel disease may be exacerbated
⚠️ Diabetic retinopathy: Rapid glycemic improvement may temporarily worsen retinal conditions
⚠️ Renal impairment: Dehydration from gastrointestinal effects may impact kidney function
Research protocols should implement comprehensive screening procedures to identify these risk factors during participant selection. Documentation of tirzepatide weight loss before and after outcomes must include detailed adverse event reporting to contribute to the compound’s complete safety database.
Monitoring Protocols for Safety
Systematic safety monitoring throughout research protocols enables early detection of potential complications and ensures participant welfare. Comprehensive monitoring strategies should address both common and rare adverse events.
Essential safety monitoring components:
📊 Vital signs: Blood pressure and heart rate monitoring to detect cardiovascular effects or dehydration
📊 Gastrointestinal symptom scales: Standardized questionnaires assessing nausea, vomiting, and bowel function
📊 Pancreatic enzymes: Lipase and amylase measurements if abdominal pain develops
📊 Renal function: Serum creatinine and eGFR monitoring, particularly during gastrointestinal symptom periods
📊 Thyroid function: Baseline and periodic TSH assessment; calcitonin measurement in high-risk individuals
📊 Gallbladder assessment: Ultrasound evaluation if right upper quadrant pain develops
📊 Hypoglycemia monitoring: Continuous glucose monitoring or frequent fingerstick measurements, particularly in combination studies
These monitoring protocols generate comprehensive safety data that complements the efficacy information captured in tirzepatide weight loss before and after assessments, providing a complete research profile.
Long-Term Safety Considerations
While clinical trials extending to 72 weeks provide valuable safety data, questions remain regarding tirzepatide’s long-term safety profile over multi-year exposure periods. Ongoing extension studies and post-marketing surveillance continue to gather this information.
Areas of ongoing safety investigation:
- Bone density effects: Potential impact of rapid weight loss on skeletal health
- Cardiovascular outcomes: Long-term effects on heart disease risk and events
- Gallbladder disease: Cholelithiasis risk with sustained weight reduction
- Nutritional status: Micronutrient deficiencies with prolonged appetite suppression
- Weight regain patterns: Metabolic effects following treatment discontinuation
Researchers designing extended tirzepatide weight loss before and after studies should incorporate these long-term safety assessments into their protocols to contribute valuable data to the field’s understanding of chronic peptide exposure effects.
Practical Considerations for Tirzepatide Research
Successfully conducting tirzepatide research requires attention to numerous practical details that extend beyond basic protocol design. These operational considerations significantly impact data quality, participant retention, and overall study success.
Sourcing Research-Grade Peptides
The foundation of valid tirzepatide research rests on obtaining high-purity compounds from reputable sources. Research-grade peptides should meet stringent quality standards verified through independent analytical testing.
Quality verification requirements:
✓ Purity certification: HPLC analysis demonstrating ≥98% purity
✓ Identity confirmation: Mass spectrometry verification of molecular weight and structure
✓ Sterility testing: Bacterial and fungal contamination screening
✓ Endotoxin levels: Limulus amebocyte lysate (LAL) testing confirming acceptably low endotoxin content
✓ Certificate of analysis: Comprehensive documentation of all quality parameters
Researchers should establish relationships with trusted peptide suppliers who provide complete quality documentation and maintain proper storage conditions throughout the supply chain. The integrity of tirzepatide weight loss before and after data depends fundamentally on peptide quality and consistency.
Storage and Stability Management
Proper peptide storage represents a critical variable affecting experimental reproducibility. Tirzepatide demonstrates specific stability characteristics that require careful management throughout research protocols.
Optimal storage conditions:
- Lyophilized peptide: -20°C to -80°C in original sealed pen peptides, protected from light and moisture
- Reconstituted solutions: 2-8°C in sterile containers, used within 28 days
- Freeze-thaw cycles: Minimize to prevent aggregation; aliquot reconstituted peptide if multiple uses planned
- Temperature excursions: Document any deviations from specified storage conditions
- Expiration monitoring: Track peptide age from manufacture date; typical shelf life 24-36 months when properly stored
Research facilities should implement temperature monitoring systems with alarm capabilities to prevent inadvertent peptide degradation that could compromise tirzepatide weight loss before and after measurement validity.
Budget and Resource Planning
Tirzepatide research involves significant resource requirements that extend beyond peptide acquisition costs. Comprehensive budget planning ensures adequate funding for all protocol components.
Major cost categories:
💰 Peptide procurement: Research-grade tirzepatide and necessary reconstitution supplies
💰 Laboratory testing: Metabolic panels, hormone assays, specialized biomarker analysis
💰 Imaging studies: DEXA scans, ultrasound examinations, optional MRI for visceral fat quantification
💰 Personnel time: Research coordinators, data managers, medical oversight
💰 Participant compensation: Incentives for protocol adherence and retention
💰 Data management: Electronic data capture systems, statistical analysis software
💰 Regulatory compliance: Ethics board fees, insurance, safety monitoring
Researchers should develop detailed budgets early in the planning process, accounting for all anticipated expenses across the full study duration. For extended tirzepatide weight loss before and after studies spanning 72 weeks or longer, budget projections should include inflation adjustments and contingency reserves.
Participant Recruitment and Retention
Successful tirzepatide research requires recruiting appropriate participants and maintaining high retention rates throughout extended observation periods. Strategic recruitment and engagement approaches significantly impact study completion rates.
Effective recruitment strategies:
📢 Targeted advertising: Digital campaigns reaching populations with relevant baseline characteristics
📢 Healthcare provider partnerships: Referrals from primary care and specialty practices
📢 Community engagement: Presentations at health fairs, community centers, and wellness programs
📢 Research registries: Recruitment from established databases of research-interested individuals
📢 Social media outreach: Platforms targeting demographics matching inclusion criteria
Retention optimization approaches:
� Flexible scheduling: Accommodating participant availability for assessments
� Transportation assistance: Reducing logistical barriers to study visits
� Regular communication: Newsletters, text reminders, and progress updates
� Participant education: Clear explanation of research importance and individual contribution
� Incentive structures: Graduated compensation recognizing long-term commitment
High retention rates ensure adequate statistical power and minimize bias from differential dropout patterns. For tirzepatide weight loss before and after studies, maintaining participant engagement throughout the full observation period captures the complete trajectory of metabolic changes.
Data Management and Analysis
Robust data management systems ensure accurate capture, storage, and analysis of the complex datasets generated in tirzepatide research. Modern electronic data capture platforms streamline these processes while maintaining data integrity.
Essential data management components:
- Electronic case report forms: Standardized data entry with built-in validation rules
- Audit trails: Complete documentation of all data modifications and corrections
- Query management: Systematic resolution of data inconsistencies or missing values
- Database locks: Preventing modifications after analysis initiation
- Statistical analysis plans: Pre-specified analytical approaches developed before data examination
- Data security: HIPAA-compliant storage with appropriate access controls
Comprehensive data management enables researchers to generate publication-quality tirzepatide weight loss before and after analyses that withstand peer review scrutiny and contribute meaningfully to scientific literature.
Future Directions in Tirzepatide Research
The evolving landscape of tirzepatide research continues to reveal new applications and mechanistic insights that extend beyond initial weight management investigations. Understanding these emerging research directions provides context for the compound’s broader scientific significance.
Metabolic Disease Applications
Beyond weight reduction, tirzepatide demonstrates effects on multiple metabolic parameters that suggest potential applications in diverse disease states. Ongoing research explores these expanded indications.
Active research areas:
🔬 Non-alcoholic fatty liver disease (NAFLD): Hepatic fat reduction and inflammation markers
🔬 Polycystic ovary syndrome (PCOS): Hormonal normalization and metabolic improvements
🔬 Obstructive sleep apnea: Weight-dependent improvements in respiratory parameters
🔬 Cardiovascular disease prevention: Long-term outcomes studies examining heart disease and stroke risk
🔬 Metabolic syndrome: Comprehensive improvements across multiple diagnostic criteria
These investigations expand the tirzepatide weight loss before and after research paradigm to encompass broader metabolic health outcomes, potentially revealing therapeutic applications beyond obesity management.
Mechanistic Research Priorities
Despite extensive clinical trial data, many questions remain regarding tirzepatide’s precise mechanisms of action at molecular and cellular levels. Ongoing mechanistic research addresses these knowledge gaps.
Key mechanistic questions:
- GIP/GLP-1 receptor interaction: How do dual receptor signals integrate at cellular and systemic levels?
- Central nervous system effects: Brain region-specific receptor activation and appetite regulation pathways
- Adipocyte biology: Direct effects on fat cell metabolism, differentiation, and apoptosis
- Gut microbiome interactions: Potential bidirectional relationships between peptide effects and microbial populations
- Genetic determinants of response: Polymorphisms affecting receptor expression or signaling pathways
Advanced research techniques including single-cell RNA sequencing, metabolomics, and systems biology approaches continue to reveal new insights into tirzepatide’s complex biological effects that underlie the observed tirzepatide weight loss before and after outcomes.
Formulation and Delivery Innovations
Next-generation tirzepatide formulations aim to enhance convenience, reduce side effects, or extend dosing intervals beyond current weekly administration protocols.
Emerging formulation research:
💊 Oral delivery systems: Overcoming peptide degradation in the gastrointestinal tract
💊 Extended-release technologies: Monthly or quarterly administration formulations
💊 Microneedle patches: Painless transdermal delivery alternatives
💊 Nanoparticle encapsulation: Targeted delivery to specific tissues or cell types
💊 Combination fixed-dose products: Single formulation containing multiple complementary peptides
These innovations may significantly alter the practical aspects of tirzepatide research while potentially enhancing the magnitude or consistency of tirzepatide weight loss before and after results through improved pharmacokinetic profiles.
Personalized Medicine Approaches
The substantial individual variability in tirzepatide response suggests opportunities for personalized approaches that optimize outcomes based on individual characteristics.
Precision medicine research directions:
- Pharmacogenomic profiling: Genetic testing to predict response likelihood and optimal dosing
- Metabolic phenotyping: Baseline metabolic assessments guiding treatment selection
- Biomarker-guided dosing: Adjusting tirzepatide dosage based on individual metabolic responses
- Combination therapy algorithms: Selecting complementary interventions based on response patterns
- Artificial intelligence prediction models: Machine learning approaches forecasting individual outcomes
These personalized approaches may enable researchers to identify individuals most likely to achieve exceptional tirzepatide weight loss before and after outcomes while minimizing exposure in poor responders.
The Evolving Landscape of Tirzepatide Research
The comprehensive body of evidence examining tirzepatide weight loss before and after outcomes has established this dual receptor agonist as a significant advancement in metabolic research. Clinical trial data demonstrating 15-22% average body weight reduction over 72-week observation periods, coupled with improvements across multiple metabolic parameters, positions tirzepatide as a valuable tool for investigating weight regulation mechanisms and metabolic disease pathways.
The documented efficacy extends beyond simple weight reduction to encompass fundamental alterations in body composition, with preferential visceral fat reduction, improved glycemic control, favorable lipid profile changes, and reduced inflammatory markers. These multi-dimensional effects provide researchers with opportunities to explore interconnected metabolic systems and develop more comprehensive understanding of obesity pathophysiology.
Key Research Implications
For researchers designing tirzepatide studies, several critical considerations emerge from the existing evidence base:
Protocol design essentials:
- Adequate study duration (minimum 48-72 weeks) to capture full metabolic effects
- Gradual dosage escalation protocols minimizing gastrointestinal adverse events
- Comprehensive monitoring encompassing weight, body composition, and metabolic biomarkers
- Careful participant selection accounting for contraindications and risk factors
- High-quality peptide sourcing with complete analytical documentation
Future research priorities:
- Long-term safety data extending beyond current 72-week maximum trial durations
- Mechanistic investigations clarifying dual receptor interaction effects
- Comparative effectiveness research against emerging multi-receptor agonists
- Personalized medicine approaches optimizing individual outcomes
- Real-world effectiveness studies complementing controlled trial data
Actionable Next Steps for Researchers
Investigators interested in conducting tirzepatide research should consider these practical steps:
- Establish reliable peptide sourcing: Partner with reputable suppliers providing research-grade compounds with comprehensive quality documentation
- Develop comprehensive protocols: Design studies incorporating appropriate controls, adequate sample sizes, and extended observation periods
- Implement robust safety monitoring: Establish systematic adverse event tracking and participant welfare procedures
- Plan thorough data management: Utilize electronic capture systems ensuring data integrity and analytical rigor
- Consider collaborative opportunities: Join multi-site research networks to enhance statistical power and generalizability
- Stay current with emerging evidence: Monitor ongoing clinical trials and mechanistic research informing protocol optimization
The Broader Scientific Context
Tirzepatide represents an important milestone in the evolution of peptide-based metabolic research, but not an endpoint. The compound’s success has catalyzed development of next-generation multi-receptor agonists, novel combination therapies, and innovative delivery systems that promise to further advance the field.
The impressive tirzepatide weight loss before and after results documented across multiple clinical trials have validated the dual receptor agonist approach and demonstrated that substantial, sustained weight reduction is achievable through peptide-based interventions. This evidence base provides a foundation for continued innovation in metabolic disease research and therapeutic development.
For researchers committed to advancing scientific understanding of weight regulation, metabolic disease, and peptide pharmacology, tirzepatide offers a well-characterized, highly effective research tool backed by extensive clinical trial data. The compound’s unique mechanism, favorable efficacy profile, and manageable safety characteristics position it as a valuable addition to the metabolic research toolkit.
Investigators ready to initiate tirzepatide research protocols can access high-purity research-grade peptides through established suppliers committed to quality, transparency, and scientific advancement. With proper protocol design, rigorous execution, and comprehensive analysis, tirzepatide research continues to generate valuable insights into metabolic regulation and disease pathophysiology.
References
[1] Frias JP, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. New England Journal of Medicine. 2021;385(6):503-515.
[2] Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. New England Journal of Medicine. 2022;387(3):205-216.
[3] Garvey WT, et al. Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nature Medicine. 2022;28(10):2083-2091.
[4] Wilding JPH, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes, Obesity and Metabolism. 2022;24(8):1553-1564.
