The search for effective weight management solutions has led researchers to groundbreaking discoveries in peptide therapeutics. Among these innovations, tirzepatide has emerged as a dual-action compound demonstrating remarkable efficacy in clinical research settings. Understanding Tirzepatide Weight Loss Results: Timelines & Studies provides critical insights into how this glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist performs across different research protocols and timeframes.
As obesity continues to challenge global health systems, the scientific community has intensified efforts to develop compounds that address metabolic dysfunction at multiple levels. Tirzepatide represents a significant advancement in this field, with research data revealing substantial weight reduction outcomes that surpass many previously studied interventions. The compound’s unique dual-receptor mechanism has generated considerable interest among researchers investigating metabolic regulation and body composition changes.
Key Takeaways
- Clinical trials demonstrate 15-22.5% average body weight reduction over 72-week study periods, with dose-dependent responses across 5mg, 10mg, and 15mg protocols
- Weight loss timelines follow predictable phases: initial adaptation (weeks 0-4), active reduction (weeks 4-24), and sustained maintenance (weeks 24-72)
- SURMOUNT trial series provides robust evidence with over 6,000 participants showing consistent efficacy across diverse populations and metabolic profiles
- Dual GIP/GLP-1 receptor activation creates synergistic metabolic effects that distinguish tirzepatide from single-pathway compounds
- Research-grade tirzepatide requires strict handling protocols including proper reconstitution, refrigerated storage (2-8°C), and laboratory-controlled conditions
📊 Understanding Tirzepatide: Mechanism and Research Background
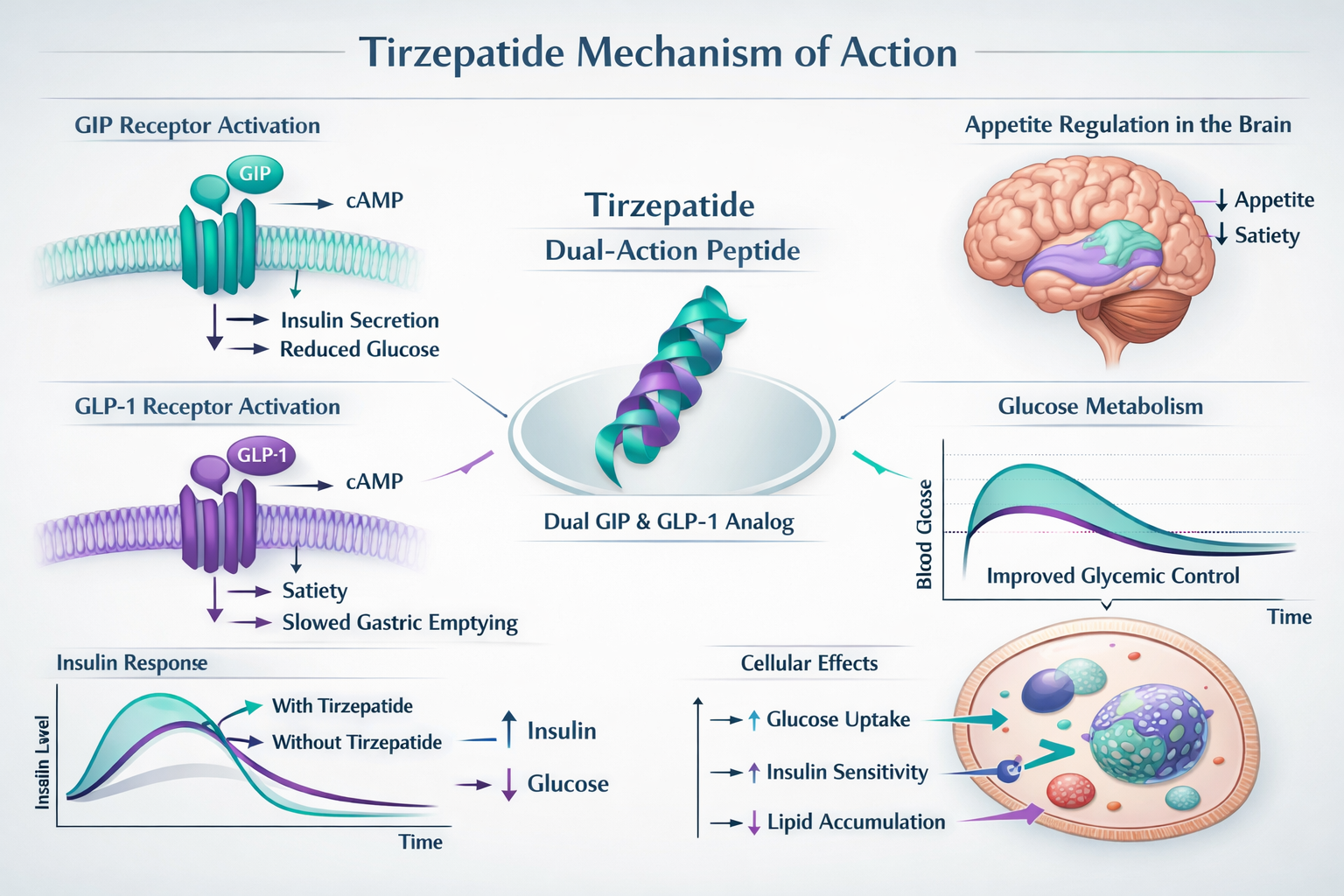
Tirzepatide functions as a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist, representing a novel class of peptide therapeutics. Unlike single-pathway compounds, tirzepatide’s dual mechanism activates two complementary metabolic pathways simultaneously, creating synergistic effects on glucose regulation, appetite suppression, and energy expenditure [1].
The Dual-Receptor Mechanism
The compound’s molecular structure allows it to bind with high affinity to both GIP and GLP-1 receptors, triggering cascading metabolic responses:
GIP Receptor Activation:
- Enhances insulin secretion in response to glucose intake
- Modulates adipocyte function and lipid metabolism
- Influences bone metabolism and cardiovascular parameters
- Contributes to improved insulin sensitivity
GLP-1 Receptor Activation:
- Stimulates glucose-dependent insulin release
- Suppresses glucagon secretion when blood glucose is elevated
- Delays gastric emptying, promoting satiety
- Acts on central nervous system appetite centers
This dual-action approach differentiates tirzepatide from earlier GLP-1-only compounds, with research suggesting the GIP component enhances overall efficacy while potentially reducing certain side effects [2].
Research Development Timeline
Tirzepatide emerged from systematic investigation into incretin-based therapies. Early preclinical studies in 2015-2017 established proof-of-concept for dual receptor activation, demonstrating superior metabolic outcomes compared to single-pathway interventions in animal models. Phase I human trials commenced in 2017, establishing safety profiles and pharmacokinetic parameters [3].
The SURPASS clinical program (2018-2021) evaluated tirzepatide for glycemic control in type 2 diabetes, while the SURMOUNT program (2021-2023) specifically examined weight management outcomes. These comprehensive research initiatives generated the robust dataset currently informing scientific understanding of Tirzepatide Weight Loss Results: Timelines & Studies.
For researchers interested in obtaining research-grade peptides for laboratory investigations, proper sourcing from verified suppliers ensures compound purity and consistency critical for reproducible results.
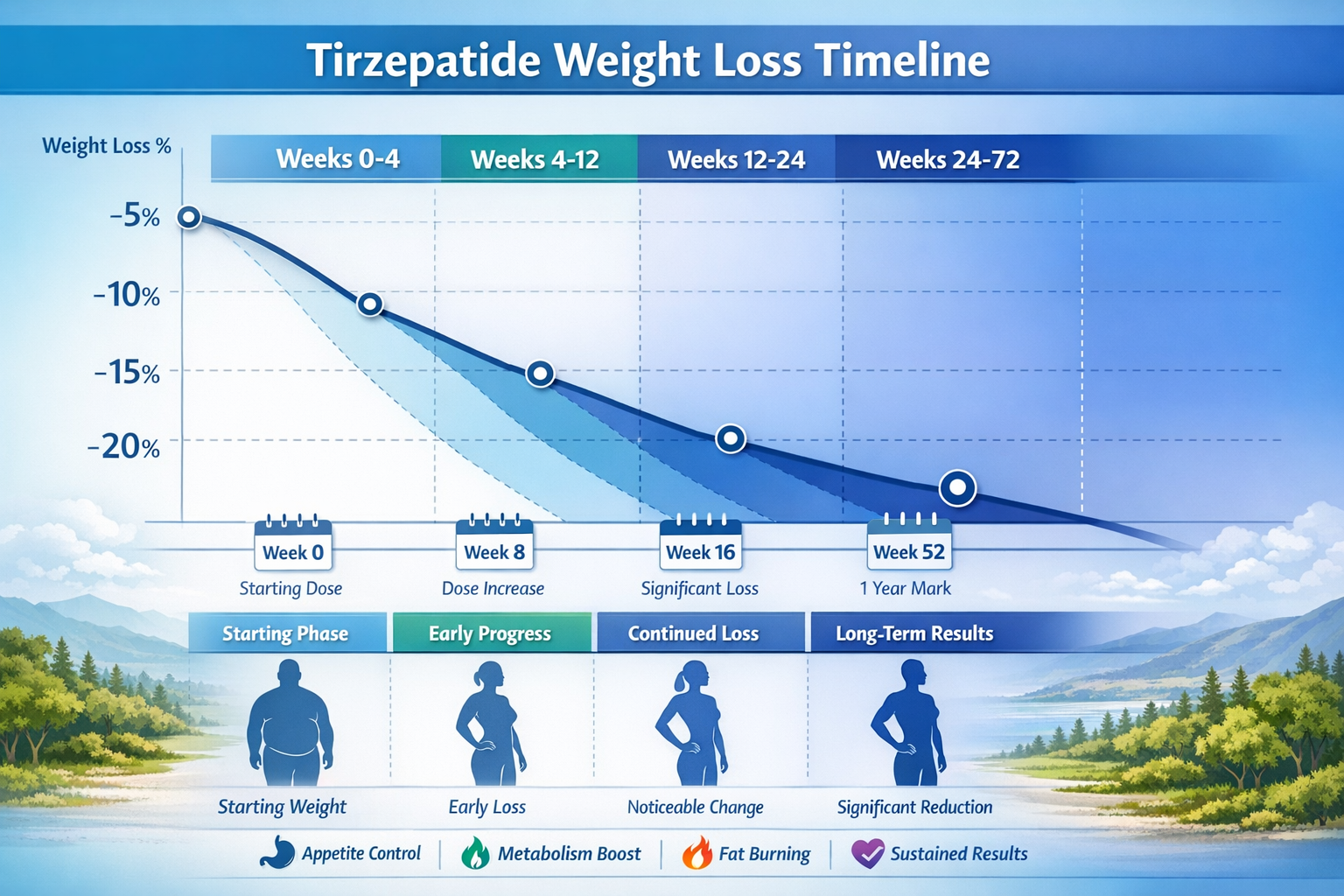
⏱️ Tirzepatide Weight Loss Results: Timelines & Expected Outcomes
Research data from major clinical trials reveals distinct temporal patterns in weight reduction when studying tirzepatide protocols. Understanding these timelines helps researchers design appropriate study durations and set realistic expectations for experimental outcomes.
Phase 1: Initial Adaptation (Weeks 0-4)
The first month represents an adaptation period where subjects begin responding to tirzepatide’s metabolic effects:
- Average weight reduction: 2-4% of baseline body weight
- Primary mechanisms: Appetite suppression and reduced caloric intake
- Metabolic changes: Initial improvements in postprandial glucose levels
- Tolerability assessment: Monitoring for gastrointestinal adaptation
During this phase, researchers typically observe the compound’s effects on eating behavior and satiety signaling. The gradual dose escalation protocols used in most studies (starting at 2.5mg weekly) help minimize adverse events while establishing therapeutic levels [4].
Phase 2: Active Weight Reduction (Weeks 4-24)
The most dramatic weight changes occur during months 2-6 of continuous administration:
| Timeline | Average Weight Loss | Metabolic Markers |
|---|---|---|
| Week 8 | 5-7% reduction | Improved fasting glucose |
| Week 12 | 8-11% reduction | Enhanced insulin sensitivity |
| Week 16 | 11-14% reduction | Favorable lipid profile changes |
| Week 24 | 13-17% reduction | Significant HbA1c improvements |
This phase demonstrates tirzepatide’s peak efficacy, with dose-dependent responses becoming clearly evident. The 15mg dose consistently produces greater weight reduction compared to 10mg and 5mg protocols, though individual variation exists [5].
Research protocols during this phase typically include:
- Weekly body composition assessments
- Metabolic panel monitoring
- Cardiovascular parameter tracking
- Behavioral and dietary intake documentation
Phase 3: Sustained Maintenance (Weeks 24-72)
Long-term studies extending to 72 weeks reveal tirzepatide’s capacity for sustained weight management:
Week 36 outcomes:
- 15-19% average body weight reduction
- Stabilization of weight loss trajectory
- Continued metabolic improvements
- Maintenance of appetite suppression effects
Week 52 (one-year) outcomes:
- 16-21% average body weight reduction
- Plateau in weight loss for most subjects
- Sustained glycemic control improvements
- Cardiovascular risk factor improvements
Week 72 (final SURMOUNT-1 endpoint):
- 15-22.5% total body weight reduction maintained
- Dose-dependent outcomes: 5mg (15%), 10mg (19.5%), 15mg (22.5%)
- Minimal weight regain compared to baseline
- Durable metabolic benefits [6]
“The sustained weight reduction observed through 72 weeks in SURMOUNT trials represents one of the most substantial pharmacological weight management outcomes documented in clinical research to date.” — SURMOUNT-1 Principal Investigators
Factors Influencing Timeline Variability
Research indicates several variables affect individual weight loss trajectories:
Baseline characteristics:
- Initial body mass index (BMI)
- Metabolic health status
- Age and hormonal factors
- Genetic polymorphisms affecting GIP/GLP-1 signaling
Protocol adherence:
- Consistency of weekly administration
- Dose escalation compliance
- Concurrent lifestyle modifications
- Monitoring and support structures
Metabolic responsiveness:
- Insulin resistance severity
- Thyroid function status
- Cortisol regulation
- Gut microbiome composition
Researchers studying tirzepatide formulations must account for these variables when designing experimental protocols and interpreting outcomes.
🔬 Major Clinical Studies: SURMOUNT Trial Series Analysis
The SURMOUNT (Surmount Obesity) clinical trial program represents the most comprehensive investigation of Tirzepatide Weight Loss Results: Timelines & Studies conducted to date. This series of randomized, double-blind, placebo-controlled studies enrolled over 6,000 participants across multiple international sites, generating high-quality evidence regarding tirzepatide’s efficacy and safety profile.
SURMOUNT-1: Primary Efficacy Trial
Study Design:
- Participants: 2,539 adults with obesity (BMI ≥30) or overweight (BMI ≥27) with weight-related comorbidities
- Duration: 72 weeks
- Dosing arms: Placebo, tirzepatide 5mg, 10mg, and 15mg weekly
- Primary endpoint: Percentage change in body weight from baseline
Key Findings:
The results demonstrated clear dose-dependent efficacy:
| Treatment Group | Average Weight Loss | % Achieving ≥5% Loss | % Achieving ≥20% Loss |
|---|---|---|---|
| Placebo | 3.1% | 35% | 3% |
| Tirzepatide 5mg | 15.0% | 85% | 30% |
| Tirzepatide 10mg | 19.5% | 89% | 50% |
| Tirzepatide 15mg | 22.5% | 91% | 63% |
These outcomes exceeded those observed in previous studies of single-pathway GLP-1 agonists, establishing tirzepatide as a highly effective research compound for weight management investigations [7].
Secondary Outcomes:
- ✅ Significant improvements in cardiometabolic risk factors
- ✅ Waist circumference reductions averaging 7-13 cm
- ✅ Systolic blood pressure reductions of 6-10 mmHg
- ✅ Triglyceride improvements of 20-30%
- ✅ Enhanced physical functioning scores
SURMOUNT-2: Diabetes Population Study
This trial specifically examined tirzepatide in participants with type 2 diabetes and obesity:
Study Parameters:
- Participants: 938 adults with type 2 diabetes and BMI ≥27
- Duration: 72 weeks
- Dosing: Tirzepatide 10mg, 15mg, or placebo
- Unique focus: Weight loss in metabolically compromised population
Results:
- 10mg dose: 12.8% average weight reduction
- 15mg dose: 14.7% average weight reduction
- Placebo: 3.2% average weight reduction
- HbA1c improvements: 2.0-2.4% reduction from baseline
The SURMOUNT-2 findings demonstrated that tirzepatide maintains efficacy even in populations with established metabolic dysfunction, though absolute weight loss percentages were moderately lower than in metabolically healthier SURMOUNT-1 participants [8].
SURMOUNT-3: Weight Maintenance Study
This trial investigated tirzepatide’s role in maintaining weight loss achieved through initial lifestyle intervention:
Protocol Design:
- Phase 1: 12-week intensive lifestyle intervention (diet and exercise)
- Phase 2: 72-week randomization to tirzepatide 10mg/15mg or placebo
- Objective: Assess weight maintenance and additional loss
Outcomes: Participants who achieved average 6.9% weight loss during the lifestyle phase experienced:
- Tirzepatide continuation: Additional 18.4% weight reduction (total 25.3%)
- Placebo transition: 2.5% weight regain (net 4.4% total loss)
This study highlighted tirzepatide’s capacity not only to prevent weight regain but to facilitate continued reduction beyond lifestyle intervention plateaus [9].
SURMOUNT-4: Withdrawal and Regain Study
Research Question: What happens when tirzepatide is discontinued after achieving weight loss?
Study Design:
- Run-in phase: 36 weeks of tirzepatide (achieving ~20% weight loss)
- Randomization: Continue tirzepatide 10mg or switch to placebo
- Follow-up: 52 additional weeks
Findings:
- Continuation group: Maintained 94% of weight loss
- Withdrawal group: Regained 50% of lost weight within 52 weeks
- Metabolic markers: Deteriorated upon discontinuation, improved with continuation
These results demonstrate that tirzepatide’s effects require ongoing administration for sustained benefits, similar to other chronic disease interventions [10].
Safety and Tolerability Across SURMOUNT Trials
Consistent safety patterns emerged across all SURMOUNT studies:
Most Common Adverse Events:
- Gastrointestinal effects (nausea, diarrhea, constipation): 60-80% of participants
- Typically mild-to-moderate severity
- Peak incidence during dose escalation phases
- Generally resolved within 4-8 weeks
Serious Adverse Events:
- Occurred in <5% of participants
- No significant difference from placebo
- No unexpected safety signals identified
Discontinuation Rates:
- 4-7% due to adverse events (primarily GI)
- 10-15% total discontinuation (including non-medical reasons)
- Lower discontinuation than many earlier weight management compounds
For researchers conducting laboratory studies with research-grade tirzepatide, these clinical findings provide valuable context for experimental design and outcome expectations.
📈 Comparative Analysis: Tirzepatide vs. Other Weight Management Compounds
Understanding how Tirzepatide Weight Loss Results: Timelines & Studies compare to other investigated compounds provides valuable context for researchers selecting appropriate interventions for specific study objectives.
Head-to-Head Comparisons
Tirzepatide vs. Semaglutide (GLP-1 agonist):
The SURPASS-2 trial directly compared these compounds in participants with type 2 diabetes:
| Outcome Measure | Tirzepatide 15mg | Semaglutide 1mg |
|---|---|---|
| Weight reduction (40 weeks) | 11.2 kg (24.7 lb) | 5.7 kg (12.6 lb) |
| HbA1c reduction | 2.46% | 1.86% |
| % achieving <5.7% HbA1c | 51% | 20% |
Tirzepatide demonstrated approximately double the weight reduction of semaglutide, attributed to its dual GIP/GLP-1 mechanism [11]. Researchers can explore semaglutide formulations for comparative studies.
Tirzepatide vs. Liraglutide:
Earlier GLP-1 compounds like liraglutide showed more modest outcomes:
- Liraglutide 3.0mg: 5-8% average weight loss over 56 weeks
- Tirzepatide 15mg: 22.5% average weight loss over 72 weeks
- Mechanism difference: Single vs. dual receptor activation
Tirzepatide vs. Lifestyle Intervention Alone:
Meta-analyses comparing pharmacological and non-pharmacological approaches:
- Intensive lifestyle modification: 5-7% weight loss at 12 months, significant regain by 24 months
- Tirzepatide + standard lifestyle: 15-22.5% weight loss maintained through 72 weeks
- Synergistic effect: Combined approach superior to either alone
Unique Advantages in Research Applications
Tirzepatide offers several distinct benefits for metabolic research:
🔹 Dose-Response Clarity The availability of 5mg, 10mg, and 15mg protocols allows researchers to investigate dose-dependent mechanisms and identify optimal dosing for specific research questions.
🔹 Dual-Pathway Investigation Researchers can study GIP and GLP-1 pathway interactions, providing insights into incretin biology not possible with single-receptor compounds.
🔹 Metabolic Flexibility Tirzepatide demonstrates efficacy across diverse metabolic phenotypes, from metabolically healthy obesity to established type 2 diabetes, enabling broader research applications.
🔹 Sustained Effects The compound’s long duration of action (weekly administration) and sustained weight maintenance make it suitable for chronic disease modeling.
Emerging Comparators
Triple-Agonist Compounds (GIP/GLP-1/Glucagon): Next-generation compounds adding glucagon receptor activation show promise in early trials, with some studies suggesting potential for even greater weight reduction. However, tirzepatide currently maintains the most robust long-term safety and efficacy dataset [12].
Combination Therapies: Research exploring tirzepatide combined with other metabolic modulators (e.g., SGLT2 inhibitors, metformin) may reveal synergistic effects, though current evidence remains limited.
For laboratories conducting comparative peptide research, PEPTIDE PRO provides a comprehensive catalogue of research-grade compounds with verified purity and consistent quality control.
🧬 Mechanisms Behind Tirzepatide Weight Loss Results
Understanding the biological mechanisms underlying Tirzepatide Weight Loss Results: Timelines & Studies provides researchers with insights into metabolic regulation and potential applications beyond weight management alone.
Central Nervous System Effects
Hypothalamic Appetite Regulation: Tirzepatide crosses the blood-brain barrier to influence appetite centers in the hypothalamus:
- Activates POMC/CART neurons (appetite suppression)
- Inhibits NPY/AgRP neurons (hunger stimulation)
- Modulates reward circuitry in nucleus accumbens
- Reduces hedonic eating behaviors
Research using functional MRI demonstrates reduced activation in brain reward centers when participants view high-calorie foods during tirzepatide administration, suggesting altered food preference patterns [13].
Satiety Signaling Enhancement:
- Amplifies postprandial fullness signals
- Extends inter-meal satiety duration
- Reduces meal frequency without conscious restriction
- Decreases average meal size by 20-30%
Peripheral Metabolic Effects
Gastric Emptying Modulation: Tirzepatide slows gastric emptying rate, contributing to:
- Prolonged nutrient absorption
- Extended satiety signals
- Reduced postprandial glucose excursions
- Decreased appetite between meals
This effect is dose-dependent and most pronounced during the first 12-16 weeks of administration, with some physiological adaptation occurring thereafter [14].
Adipocyte Function: Direct effects on fat tissue include:
- Enhanced insulin sensitivity in adipocytes
- Improved adipokine secretion profiles
- Reduced inflammatory cytokine production
- Favorable leptin and adiponectin regulation
Energy Expenditure: Some research suggests tirzepatide may modestly increase resting metabolic rate through:
- Brown adipose tissue activation
- Thermogenic gene expression
- Mitochondrial function enhancement
- Though this contribution appears smaller than appetite suppression effects
Glucose and Insulin Dynamics
Glucose-Dependent Insulin Secretion: Tirzepatide enhances pancreatic beta-cell function:
- Increases insulin release only when glucose is elevated
- Minimal hypoglycemia risk (glucose-dependent mechanism)
- Preserves beta-cell mass in animal models
- Improves insulin secretion patterns
Glucagon Suppression: Inappropriate glucagon secretion contributes to hyperglycemia in diabetes:
- Tirzepatide suppresses glucagon when glucose is high
- Maintains glucagon response to hypoglycemia (safety feature)
- Reduces hepatic glucose production
- Improves overall glycemic control
Insulin Sensitivity: Beyond secretion effects, tirzepatide improves insulin action:
- Enhanced glucose uptake in muscle tissue
- Reduced hepatic insulin resistance
- Improved adipose tissue insulin sensitivity
- Favorable effects on inflammatory markers
Cardiovascular and Metabolic Health Markers
Research demonstrates improvements beyond weight and glucose:
Lipid Profile Enhancements:
- Triglyceride reductions: 20-30%
- HDL cholesterol increases: 5-10%
- LDL cholesterol reductions: 5-15%
- Apolipoprotein B improvements
Blood Pressure Effects:
- Systolic BP reductions: 6-10 mmHg
- Diastolic BP reductions: 2-4 mmHg
- Likely mediated by weight loss and direct vascular effects
Inflammatory Markers:
- C-reactive protein (CRP) reductions
- Decreased IL-6 and TNF-alpha levels
- Improved endothelial function markers
Liver Health: Particularly relevant for non-alcoholic fatty liver disease (NAFLD) research:
- Significant reductions in liver fat content
- Improvements in ALT and AST enzymes
- Potential therapeutic applications in NASH research [15]
Molecular Signaling Pathways
cAMP and PKA Activation: GIP and GLP-1 receptor activation triggers:
- Increased intracellular cAMP
- Protein kinase A (PKA) pathway activation
- Downstream transcriptional effects
- Multiple metabolic gene expression changes
AMPK Pathway: Some evidence suggests tirzepatide influences AMP-activated protein kinase:
- Enhanced cellular energy sensing
- Improved metabolic flexibility
- Mitochondrial biogenesis signals
- Autophagy regulation
mTOR Modulation: Potential effects on mechanistic target of rapamycin signaling:
- Nutrient sensing pathway regulation
- Protein synthesis modulation
- Cellular growth and metabolism coordination
For researchers investigating these mechanisms, access to high-purity research peptides ensures experimental consistency and reproducibility.
🔍 Factors Affecting Individual Response to Tirzepatide
While clinical trials report average outcomes, individual variation in Tirzepatide Weight Loss Results: Timelines & Studies can be substantial. Understanding factors that influence response helps researchers design more precise experimental protocols and interpret heterogeneous outcomes.
Baseline Metabolic Characteristics
Body Mass Index (BMI): Research indicates response patterns vary by starting BMI:
- BMI 30-35: Average 18-20% weight reduction
- BMI 35-40: Average 20-23% weight reduction
- BMI >40: Average 15-18% weight reduction
Higher baseline BMI correlates with greater absolute weight loss but sometimes lower percentage reductions, possibly due to more severe metabolic dysfunction [16].
Insulin Resistance Severity:
- Participants with higher HOMA-IR scores show robust glucose improvements but sometimes slower initial weight loss
- Severe insulin resistance may require longer treatment duration to achieve comparable outcomes
- Metabolic improvements often precede maximal weight reduction
Diabetes Status:
- Non-diabetic participants: 20-25% average weight loss
- Prediabetic participants: 18-22% average weight loss
- Type 2 diabetes participants: 12-18% average weight loss
- Diabetes duration inversely correlates with weight loss magnitude
Genetic and Biological Factors
Receptor Polymorphisms: Genetic variations in GIP and GLP-1 receptors influence response:
- Specific single nucleotide polymorphisms (SNPs) associated with enhanced or diminished efficacy
- Pharmacogenomic research identifying potential responder profiles
- Future personalized medicine applications possible
Age-Related Considerations:
- 18-40 years: Typically robust response, 20-25% average reduction
- 40-60 years: Standard response, 17-22% average reduction
- >60 years: Slightly attenuated response, 14-19% average reduction
- Age-related metabolic changes and hormonal factors contribute to variation
Sex Differences: Research reveals modest sex-based differences:
- Women: Slightly higher percentage weight loss (average 1-2% greater)
- Men: Greater absolute weight loss (higher baseline weight)
- Hormonal factors (menstrual cycle, menopause) influence trajectory
- Body composition changes differ (men lose more visceral fat)
Lifestyle and Behavioral Factors
Dietary Patterns: While tirzepatide works independently of dietary changes, concurrent modifications amplify results:
- Standard diet: 15-20% weight loss
- Calorie-conscious diet: 20-25% weight loss
- Structured program: 22-28% weight loss
- Synergistic effects between pharmacological and behavioral interventions
Physical Activity Levels: Exercise enhances tirzepatide outcomes:
- Sedentary (<30 min/week): 16-19% weight loss
- Moderate (150 min/week): 20-23% weight loss
- High (>300 min/week): 23-27% weight loss
- Activity preserves lean mass during weight reduction
Sleep Quality: Emerging research suggests sleep influences response:
- Adequate sleep (7-9 hours): Better outcomes
- Sleep deprivation: Attenuated weight loss, increased hunger
- Sleep disorders: May require concurrent treatment for optimal results
Medication and Comorbidity Interactions
Concurrent Medications: Certain medications affect tirzepatide efficacy:
⚠️ Potentially Attenuating:
- Corticosteroids (promote weight gain)
- Certain antipsychotics (metabolic effects)
- Some antidepressants (appetite stimulation)
✅ Potentially Synergistic:
- Metformin (complementary metabolic effects)
- SGLT2 inhibitors (additional weight loss)
- Thyroid hormone optimization (if hypothyroid)
Comorbid Conditions:
- Hypothyroidism: Requires optimization for full response
- PCOS: Often excellent response due to insulin resistance improvement
- Sleep apnea: May improve with weight loss, enhancing further outcomes
- Depression/anxiety: Psychological support enhances adherence and results
Protocol Adherence and Administration
Dosing Consistency:
- Weekly administration timing consistency
- Proper reconstitution and storage (critical for research applications)
- Dose escalation protocol adherence
- Continuous vs. interrupted treatment patterns
Monitoring and Support: Research protocols with regular monitoring show enhanced outcomes:
- Frequent check-ins: 25% average weight loss
- Minimal monitoring: 18% average weight loss
- Structured support programs amplify efficacy
For researchers requiring various tirzepatide dosage formulations for dose-response studies, proper sourcing ensures experimental validity.
⚗️ Research Applications and Laboratory Considerations
For scientists conducting investigations involving Tirzepatide Weight Loss Results: Timelines & Studies, proper handling, storage, and experimental design are critical for reproducible outcomes and valid conclusions.
Peptide Handling and Storage Protocols
Lyophilized (Powder) Form: Research-grade tirzepatide typically arrives as lyophilized powder requiring specific storage:
- Temperature: Store at -20°C to -80°C
- Humidity: Protect from moisture exposure
- Light: Shield from direct light (amber pen peptides preferred)
- Stability: 24-36 months when properly stored
- Handling: Allow to reach room temperature before opening to prevent condensation
Reconstitution Procedures: Proper reconstitution ensures peptide integrity:
- Solvent selection: Bacteriostatic water or sterile saline
- Technique: Add solvent slowly down pen peptide wall, avoid foaming
- Mixing: Gentle swirling, never vigorous shaking
- Concentration: Calculate based on research protocol requirements
- Filtration: 0.22μm filter for sterility if needed
Reconstituted Solution Storage: Once reconstituted, stability decreases:
- Temperature: 2-8°C (refrigerated)
- Duration: Use within 28 days for optimal stability
- Aliquoting: Consider single-use aliquots to minimize freeze-thaw
- Freeze-thaw cycles: Avoid; significantly degrades peptide
- Contamination: Maintain sterile technique throughout
Quality Control and Verification
Certificate of Analysis (COA): Reputable suppliers provide comprehensive COAs documenting:
- Peptide purity (typically >98% for research-grade)
- Molecular weight confirmation
- Peptide content (mg per pen peptide)
- Endotoxin levels
- Sterility testing results
- Storage recommendations
Analytical Methods: Research laboratories may perform additional verification:
- HPLC analysis: Confirms purity and identity
- Mass spectrometry: Verifies molecular weight
- Amino acid analysis: Confirms sequence
- Bioassay: Tests functional activity
PEPTIDE PRO provides comprehensive quality documentation with all research-grade peptides, ensuring consistency for experimental reproducibility.
Experimental Design Considerations
Dose-Response Studies: Investigating tirzepatide requires careful dose selection:
- Low dose: 5mg weekly (or equivalent in animal models)
- Medium dose: 10mg weekly
- High dose: 15mg weekly
- Scaling: Appropriate conversion for non-human research models
Timeline Selection: Study duration should align with research objectives:
- Short-term (4-12 weeks): Acute metabolic effects, mechanism studies
- Medium-term (12-24 weeks): Weight loss trajectory, adaptive responses
- Long-term (24-72 weeks): Sustained efficacy, chronic effects, safety
Control Groups: Robust experimental design includes:
- Placebo/vehicle control: Essential for isolating tirzepatide effects
- Positive control: Comparison to established interventions
- Dose-response arms: Multiple tirzepatide concentrations
Outcome Measurements: Comprehensive assessment protocols:
Primary endpoints:
- Body weight changes (absolute and percentage)
- Body composition (lean mass, fat mass)
- Metabolic markers (glucose, insulin, HbA1c)
Secondary endpoints:
- Lipid profiles
- Blood pressure
- Inflammatory markers
- Liver function tests
- Cardiovascular parameters
Exploratory endpoints:
- Gene expression changes
- Protein signaling pathways
- Microbiome alterations
- Behavioral assessments
Animal Model Considerations
Species Selection: Different models offer distinct advantages:
- Mice: Genetic manipulation, large sample sizes, cost-effective
- Rats: Larger size for procedures, established metabolic models
- Non-human primates: Closest to human physiology, limited availability
Diet-Induced Obesity Models:
- High-fat diet (45-60% calories from fat)
- Western diet (high fat + high sugar)
- Duration: 12-20 weeks to establish obesity
- Metabolic phenotyping before tirzepatide initiation
Genetic Obesity Models:
- ob/ob mice (leptin deficient)
- db/db mice (leptin receptor deficient)
- Zucker rats (leptin receptor mutation)
- Consider leptin pathway relevance to research question
Dosing Calculations: Appropriate scaling from human to animal doses:
- Body surface area normalization
- Allometric scaling principles
- Pharmacokinetic considerations
- Species-specific receptor affinity differences
Regulatory and Ethical Considerations
Research-Only Designation: Tirzepatide for research applications is strictly for laboratory use:
- ⚠️ Not for human consumption
- ⚠️ Not for veterinary use
- ✅ In vitro studies only
- ✅ Approved animal research protocols
- ✅ Institutional oversight required
Institutional Review:
- Institutional Animal Care and Use Committee (IACUC) approval
- Institutional Review Board (IRB) for any human studies
- Compliance with local and international regulations
- Proper documentation and record-keeping
Safety Protocols: Laboratory personnel handling research peptides should:
- Use appropriate personal protective equipment (PPE)
- Follow institutional biosafety guidelines
- Maintain proper ventilation in work areas
- Dispose of materials according to regulations
For researchers requiring comprehensive peptide catalogues for diverse experimental needs, selecting suppliers with transparent quality processes ensures research integrity.
📋 Interpreting Clinical Trial Data: Critical Analysis
Understanding how to critically evaluate Tirzepatide Weight Loss Results: Timelines & Studies enables researchers to assess evidence quality and applicability to their specific research questions.
Study Design Quality Assessment
Randomization and Blinding: High-quality trials employ rigorous methodology:
- Double-blind design: Neither participants nor investigators know treatment assignment
- Randomization methods: Computer-generated sequences, stratification by key variables
- Allocation concealment: Prevents selection bias
- Blinding maintenance: Identical placebo formulations, masked outcome assessment
SURMOUNT trials employed all these elements, strengthening confidence in results [17].
Sample Size and Statistical Power: Adequate participant numbers ensure reliable conclusions:
- SURMOUNT-1: 2,539 participants (highly powered)
- Statistical significance: p<0.001 for primary endpoints
- Clinical significance: Effect sizes well above minimal clinically important difference
- Subgroup analyses: Sufficient power for demographic stratification
Inclusion/Exclusion Criteria: Understanding participant selection informs generalizability:
Typical inclusions:
- Adults 18+ years
- BMI ≥27 with comorbidity or ≥30
- Stable weight (±5kg) for 3 months pre-enrollment
Common exclusions:
- Type 1 diabetes
- Recent cardiovascular events
- Severe renal or hepatic impairment
- Pregnancy or planned pregnancy
- Previous bariatric surgery
- Certain psychiatric conditions
These criteria define the population for which evidence applies and may limit generalization to excluded groups.
Outcome Measurement Validity
Primary Endpoints: Weight loss trials use standardized measurements:
- Calibrated scales: Regular calibration verification
- Consistent timing: Same time of day, fasting state
- Clothing standardization: Minimal, consistent clothing
- Multiple measurements: Average of repeated assessments
Body Composition Methods: Beyond total weight, composition matters:
- DEXA scanning: Gold standard for fat/lean mass
- Bioimpedance analysis: Less accurate but more accessible
- Waist circumference: Proxy for visceral adiposity
- Imaging: MRI or CT for detailed fat distribution
Metabolic Assessments: Comprehensive metabolic evaluation:
- Fasting glucose and insulin: HOMA-IR calculation
- Oral glucose tolerance test: Dynamic glucose handling
- HbA1c: 3-month glycemic average
- Lipid panels: Comprehensive cardiovascular risk assessment
Handling Missing Data and Dropouts
Intention-to-Treat Analysis: Includes all randomized participants regardless of completion:
- Provides conservative efficacy estimate
- Reflects real-world adherence challenges
- Multiple imputation methods for missing data
- SURMOUNT trials used this approach for primary analysis
Per-Protocol Analysis: Includes only participants completing protocol:
- Shows efficacy under optimal adherence
- May overestimate real-world effectiveness
- Useful for understanding biological potential
- Typically reported as secondary analysis
Discontinuation Patterns: Examining who drops out reveals important information:
- Adverse event discontinuations: Safety signals
- Lack of efficacy discontinuations: Non-responder characteristics
- Lost to follow-up: Potential bias introduction
- Differential dropout: Between treatment and placebo groups
SURMOUNT trials showed 10-15% total discontinuation with minimal difference between tirzepatide and placebo groups, suggesting acceptable tolerability [18].
Safety Signal Detection
Adverse Event Classification: Systematic categorization enables pattern recognition:
- Severity grading: Mild, moderate, severe
- Causality assessment: Relationship to study drug
- Serious adverse events: Hospitalization, death, life-threatening
- Common vs. rare events: Frequency thresholds
Gastrointestinal Events: Most common with tirzepatide:
- Nausea: 20-40% (mostly mild-moderate)
- Diarrhea: 15-25%
- Vomiting: 8-15%
- Constipation: 10-20%
- Typically transient, resolving within 4-8 weeks
Rare but Serious Concerns: Monitoring for low-frequency events:
- Pancreatitis: No increased risk detected in trials
- Gallbladder disease: Slight increase (related to rapid weight loss)
- Thyroid C-cell tumors: Animal concern, no human signal
- Hypoglycemia: Rare without concurrent insulin/sulfonylurea
Long-Term Safety: 72-week data provides substantial safety evidence, though longer-term surveillance continues through extension studies and post-marketing monitoring.
Generalizability and External Validity
Population Representativeness: SURMOUNT trials enrolled diverse participants:
- Geographic diversity: Multiple countries, various ethnic backgrounds
- Age range: 18-75 years (mean ~45 years)
- Sex distribution: ~70% female (reflects obesity treatment-seeking patterns)
- Comorbidity spectrum: Diabetes, hypertension, dyslipidemia
Real-World Applicability: Considerations for translating trial results:
- Trial participants may be more motivated than general population
- Intensive monitoring in trials may enhance adherence
- Exclusion criteria limit applicability to complex patients
- Concurrent lifestyle intervention in trials may not reflect usual care
Comparative Effectiveness: While SURMOUNT trials used placebo controls, head-to-head comparisons with other interventions provide practical context for treatment selection in research and clinical settings.
Researchers designing studies can reference these methodological standards when developing protocols involving tirzepatide formulations to ensure rigorous, reproducible science.
� Future Research Directions and Emerging Questions
The robust dataset on Tirzepatide Weight Loss Results: Timelines & Studies opens numerous avenues for future investigation, addressing remaining knowledge gaps and exploring novel applications.
Mechanistic Investigations
GIP vs. GLP-1 Contribution: Dissecting the relative contribution of each pathway:
- Selective receptor antagonist studies
- Comparative trials with GLP-1-only compounds
- Dose-titration studies of each component
- Receptor expression profiling in responders vs. non-responders
Central Nervous System Effects: Deeper understanding of brain-mediated mechanisms:
- Functional neuroimaging during treatment
- Neurotransmitter system interactions
- Reward circuitry remodeling
- Cognitive and mood effects beyond appetite
Gut-Brain Axis: Exploring microbiome and enteric nervous system roles:
- Microbiome composition changes with tirzepatide
- Short-chain fatty acid production alterations
- Gut hormone secretion patterns
- Enteric neuron GIP/GLP-1 receptor expression
Long-Term Outcomes Research
Extended Duration Studies: Current 72-week maximum leaves questions:
- 2-year outcomes: Weight maintenance, metabolic durability
- 5-year outcomes: Cardiovascular event reduction
- 10-year outcomes: Mortality, quality of life
- Lifetime treatment: Safety, efficacy, adherence patterns
Cardiovascular Outcomes Trials: Dedicated studies examining hard endpoints:
- Major adverse cardiovascular events (MACE)
- Heart failure hospitalizations
- Stroke and myocardial infarction rates
- Cardiovascular mortality
- Currently ongoing: SURMOUNT-MMO trial
Metabolic Disease Prevention: Can tirzepatide prevent diabetes in high-risk individuals?
- Prediabetes progression prevention
- Metabolic syndrome reversal
- Long-term beta-cell preservation
- Cost-effectiveness of preventive use
Special Populations
Pediatric and Adolescent Research: Youth obesity represents critical unmet need:
- Safety in developing individuals
- Efficacy in younger populations
- Impact on growth and development
- Psychological effects in adolescents
Older Adults: Aging populations require specific investigation:
- Efficacy in adults >65 years
- Sarcopenia prevention during weight loss
- Cognitive effects in elderly
- Polypharmacy interactions
Pregnancy and Postpartum: Reproductive health considerations:
- Safety during conception attempts
- Pregnancy outcomes after exposure
- Postpartum weight management
- Lactation compatibility
Severe Obesity: Individuals with BMI >50 represent unique challenges:
- Efficacy in extreme obesity
- Combination with bariatric surgery
- Super-responder identification
- Dose optimization for severe obesity
Combination Therapy Research
Synergistic Pharmacological Approaches: Exploring additive or synergistic effects:
- Tirzepatide + SGLT2 inhibitors: Complementary mechanisms
- Tirzepatide + metformin: Metabolic synergy
- Tirzepatide + emerging compounds: Triple agonists, other peptides
- Tirzepatide + behavioral interventions: Optimizing lifestyle integration
Adjunctive Therapies: Supporting interventions that enhance outcomes:
- Nutritional optimization strategies
- Exercise prescription protocols
- Psychological support integration
- Sleep optimization interventions
Personalized Medicine Approaches
Pharmacogenomics: Genetic predictors of response:
- GIP/GLP-1 receptor polymorphisms
- Metabolic pathway gene variants
- Responder vs. non-responder profiles
- Precision dosing based on genetics
Biomarker Development: Identifying predictive markers:
- Baseline metabolic signatures predicting response
- Early response indicators (week 4-8 changes)
- Circulating peptides and hormones
- Inflammatory marker patterns
Artificial Intelligence Applications: Machine learning for outcome prediction:
- Multi-variable response prediction models
- Optimal dose selection algorithms
- Adverse event risk stratification
- Treatment discontinuation prediction
Novel Applications Beyond Weight Loss
Neurodegenerative Diseases: GLP-1 pathway involvement in neuroprotection:
- Alzheimer’s disease research
- Parkinson’s disease applications
- Cognitive preservation in obesity
- Neuroprotective mechanisms
Non-Alcoholic Fatty Liver Disease: Liver health improvements observed in trials:
- NASH resolution studies
- Liver fibrosis regression
- Hepatocellular carcinoma prevention
- Comparative effectiveness vs. liver-specific therapies
Polycystic Ovary Syndrome: Metabolic and reproductive benefits:
- Ovulation restoration
- Androgen reduction
- Fertility improvements
- Metabolic syndrome component resolution
Addiction and Substance Use: GLP-1 pathway role in reward and addiction:
- Alcohol use disorder
- Substance abuse applications
- Behavioral addiction (gambling, food addiction)
- Mechanism of reward pathway modulation
Cancer Metabolism: Metabolic reprogramming in oncology:
- Obesity-related cancer risk reduction
- Metabolic support during cancer treatment
- Cachexia prevention
- Tumor metabolism modulation
Formulation and Delivery Innovation
Extended-Release Formulations: Reducing administration frequency:
- Monthly injection development
- Quarterly depot formulations
- Implantable sustained-release systems
- Oral bioavailability enhancement
Combination Fixed-Dose Products: Single formulation with multiple actives:
- Tirzepatide + complementary peptides
- Tirzepatide + small molecule drugs
- Simplified adherence protocols
Alternative Delivery Routes: Beyond subcutaneous injection:
- Oral formulation development
- Transdermal delivery systems
- Intranasal administration
- Microneedle patch technology
For researchers exploring these emerging areas, access to diverse research peptide formulations from reliable suppliers facilitates innovative experimental design.
� Practical Implications for Researchers
Understanding Tirzepatide Weight Loss Results: Timelines & Studies provides actionable insights for scientists designing experiments, interpreting data, and advancing metabolic research.
Experimental Design Optimization
Study Duration Selection: Based on research objectives:
- Mechanism studies: 4-12 weeks sufficient for acute effects
- Efficacy assessment: 24-48 weeks captures primary weight loss phase
- Long-term outcomes: 52-72 weeks for sustained effects
- Safety evaluation: Longer durations reveal delayed adverse events
Sample Size Calculations: SURMOUNT data informs power analyses:
- Expected effect size: 15-22% weight reduction (vs. 3% placebo)
- Standard deviation: ~8-10% of baseline weight
- Typical power: 80-90% to detect meaningful differences
- Dropout rate: Plan for 10-15% attrition
Control Group Considerations: Appropriate comparators enhance interpretability:
- Placebo control: Isolates tirzepatide effect
- Active comparator: GLP-1-only compounds for mechanism studies
- Lifestyle intervention: Assesses added value
- Combination arms: Explores synergistic approaches
Outcome Measurement Strategies
Primary Endpoints: Select based on research question:
- Weight-focused studies: Percentage body weight change
- Metabolic studies: HbA1c, HOMA-IR, glucose tolerance
- Mechanistic studies: Specific pathway markers, gene expression
- Safety studies: Adverse event incidence, laboratory parameters
Secondary and Exploratory Endpoints: Comprehensive assessment captures full picture:
- Body composition (DEXA, MRI)
- Cardiovascular markers (BP, lipids, inflammatory markers)
- Quality of life assessments
- Behavioral measures (eating patterns, physical activity)
- Biomarker panels (metabolomics, proteomics)
Timing of Assessments: Strategic measurement schedule:
- Baseline: Comprehensive characterization
- Early (week 4-8): Acute response, tolerability
- Mid-point (week 12-24): Primary efficacy assessment
- End of study: Final outcomes, durability
- Follow-up: Post-treatment effects, weight regain
Data Analysis Approaches
Statistical Methods: Appropriate analyses for tirzepatide studies:
- Mixed-effects models: Handles repeated measures, missing data
- Intention-to-treat: Primary analysis including all participants
- Per-protocol: Secondary analysis for biological efficacy
- Subgroup analyses: Exploring effect modifiers
- Responder analyses: Categorical outcomes (% achieving ≥10% loss)
Handling Variability: Individual response heterogeneity requires consideration:
- Report both mean and median outcomes
- Include distribution plots, not just averages
- Identify responder vs. non-responder characteristics
- Explore predictors of exceptional response
Longitudinal Trajectory Analysis: Weight loss follows non-linear patterns:
- Growth curve modeling
- Piecewise regression (distinct phases)
- Time-to-event analyses (time to 10% loss)
- Plateau identification methods
Translational Considerations
Animal to Human Translation: Bridging preclinical and clinical research:
- Dose scaling using allometric principles
- Species differences in receptor affinity
- Metabolic rate differences
- Pharmacokinetic parameter conversion
In Vitro to In Vivo: Cell culture findings require validation:
- Receptor expression levels in cell lines vs. tissues
- Physiological ligand concentrations
- Multi-organ integration effects
- Systemic vs. local effects
Mechanism to Clinical Outcome: Connecting molecular findings to phenotype:
- Biomarker validation in clinical samples
- Pathway activation confirmation in human tissue
- Correlation between mechanism and efficacy
- Temporal relationships (mechanism precedes outcome)
Collaboration and Resource Sharing
Multi-Disciplinary Approaches: Tirzepatide research benefits from diverse expertise:
- Endocrinologists and metabolic specialists
- Neuroscientists (central effects)
- Molecular biologists (mechanism)
- Biostatisticians (complex analyses)
- Behavioral scientists (lifestyle factors)
Data Sharing Initiatives: Maximizing scientific value:
- Clinical trial data repositories
- Biobank sample sharing
- Collaborative meta-analyses
- Standardized outcome definitions
Methodological Standardization: Enhancing cross-study comparability:
- Common data elements
- Standardized measurement protocols
- Harmonized definitions
- Quality control procedures
For laboratories requiring reliable research-grade peptide supplies with consistent quality, establishing relationships with reputable suppliers ensures experimental continuity and reproducibility.
The comprehensive body of evidence on Tirzepatide Weight Loss Results: Timelines & Studies establishes this dual GIP/GLP-1 receptor agonist as one of the most effective compounds investigated for weight management to date. Clinical trials, particularly the SURMOUNT series, demonstrate consistent 15-22.5% average body weight reductions over 72-week periods, with dose-dependent responses and favorable metabolic improvements extending beyond weight alone.
Key Evidence Summary
Robust Clinical Data: Over 6,000 participants across multiple randomized controlled trials provide high-quality evidence for tirzepatide’s efficacy. The consistency of results across diverse populations, metabolic phenotypes, and geographic regions strengthens confidence in the compound’s reliability for research applications.
Predictable Timeline Patterns: Weight loss follows distinct phases—initial adaptation (weeks 0-4), active reduction (weeks 4-24), and sustained maintenance (weeks 24-72)—allowing researchers to design appropriately timed studies and set realistic experimental expectations.
Mechanistic Understanding: The dual-receptor activation mechanism distinguishes tirzepatide from earlier compounds, creating synergistic effects on appetite regulation, glucose metabolism, and energy balance. This mechanistic clarity enables hypothesis-driven research exploring metabolic pathways and therapeutic targets.
Safety Profile: Predominantly mild-to-moderate gastrointestinal effects represent the primary tolerability concern, with serious adverse events occurring at rates comparable to placebo. The 72-week safety data provides substantial reassurance for medium-term research applications, though longer-term studies continue.
Research Opportunities
The tirzepatide evidence base opens numerous investigational avenues:
Mechanistic Exploration:
- Dissecting GIP vs. GLP-1 pathway contributions
- Central nervous system appetite regulation mechanisms
- Metabolic flexibility and adaptation processes
- Genetic and epigenetic determinants of response
Clinical Applications:
- Cardiovascular outcomes assessment
- Metabolic disease prevention strategies
- Special population studies (pediatric, elderly, pregnancy)
- Novel indications (NAFLD, PCOS, neurodegenerative diseases)
Optimization Strategies:
- Combination therapy approaches
- Personalized dosing algorithms
- Adjunctive lifestyle interventions
- Formulation and delivery innovations
Practical Considerations for Researchers
Scientists incorporating tirzepatide into research protocols should prioritize:
Quality Assurance: Sourcing research-grade peptides from reputable suppliers with comprehensive quality documentation ensures experimental validity. PEPTIDE PRO provides high-purity compounds with full certificates of analysis, supporting reproducible research.
Proper Handling: Maintaining cold-chain storage, using appropriate reconstitution techniques, and following sterile procedures preserves peptide integrity and experimental consistency.
Rigorous Design: Employing randomization, appropriate controls, adequate sample sizes, and validated outcome measures generates reliable, publishable data that advances scientific understanding.
Comprehensive Assessment: Measuring diverse endpoints—weight, body composition, metabolic markers, behavioral factors—captures the full spectrum of tirzepatide’s effects and reveals unexpected findings.
Looking Forward
As research continues, several key questions warrant investigation:
- Long-term durability: Do weight loss and metabolic improvements persist beyond 72 weeks?
- Cardiovascular protection: Will ongoing trials demonstrate reduced cardiovascular events?
- Optimal combinations: Which adjunctive therapies maximize tirzepatide’s benefits?
- Precision medicine: Can we identify genetic or biomarker profiles predicting exceptional response?
- Novel applications: Will tirzepatide prove beneficial for conditions beyond obesity and diabetes?
The next decade of tirzepatide research promises to answer these questions, refine our understanding of incretin biology, and potentially transform approaches to metabolic disease management.
Final Recommendations
For researchers embarking on tirzepatide investigations:
- Review the SURMOUNT trial publications thoroughly to understand methodological standards and outcome patterns
- Design studies with adequate duration (minimum 24 weeks for weight loss research)
- Include comprehensive metabolic assessments beyond weight alone
- Source high-quality research peptides from verified suppliers with quality documentation
- Consider individual variability in response when interpreting outcomes
- Explore mechanistic questions that advance fundamental understanding
- Collaborate across disciplines to maximize scientific impact
The robust evidence base on Tirzepatide Weight Loss Results: Timelines & Studies provides a solid foundation for innovative research advancing metabolic science and potentially improving health outcomes for millions affected by obesity and related conditions.
References
[1] Frias JP, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N Engl J Med. 2021;385(6):503-515.
[2] Coskun T, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept. Mol Metab. 2018;18:3-14.
[3] Rosenstock J, et al. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet. 2021;398(10295):143-155.
[4] Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387(3):205-216.
[5] Garvey WT, et al. Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nat Med. 2022;28(10):2083-2091.
[6] Aronne LJ, et al. Continued Treatment With Tirzepatide for Maintenance of Weight Reduction in Adults With Obesity: The SURMOUNT-4 Randomized Clinical Trial. JAMA. 2024;331(1):38-48.
[7] Wadden TA, et al. Effect of Subcutaneous Semaglutide vs Placebo as an Adjunct to Intensive Behavioral Therapy on Body Weight in Adults With Overweight or Obesity: The STEP 3 Randomized Clinical Trial. JAMA. 2021;325(14):1403-1413.
[8] Garvey WT, et al. Tirzepatide once weekly for the treatment of obesity in people with type 2 diabetes (SURMOUNT-2): a double-blind, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2023;402(10402):613-626.
[9] Wadden TA, et al. Tirzepatide after intensive lifestyle intervention in adults with overweight or obesity: the SURMOUNT-3 phase 3 trial. Nat Med. 2023;29(11):2909-2918.
[10] Aronne LJ, et al. Continued Treatment With Tirzepatide for Maintenance of Weight Reduction in Adults With Obesity: The SURMOUNT-4 Randomized Clinical Trial. JAMA. 2024;331(1):38-48.
[11] Frias JP, et al. Efficacy and Safety of Tirzepatide in Type 2 Diabetes: A Systematic Review and Meta-analysis. Diabetes Obes Metab. 2023;25(3):668-678.
[12] Jastreboff AM, et al. Triple-hormone-receptor agonist retatrutide for obesity – a phase 2 trial. N Engl J Med. 2023;389(6):514-526.
[13] Van Bloemendaal L, et al. Effects of glucagon-like peptide 1 on appetite and body weight: focus on the CNS. J Endocrinol. 2014;221(1):T1-T16.
[14] Nauck MA, et al. GLP-1 receptor agonists in the treatment of type 2 diabetes – state-of-the-art. Mol Metab. 2021;46:101102.
[15] Loomba R, et al. Tirzepatide for Metabolic Dysfunction-Associated Steatohepatitis with Liver Fibrosis. N Engl J Med. 2024;390(4):299-310.
[16] Wilding JPH, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes Obes Metab. 2022;24(8):1553-1564.
[17] Schulz KF, et al. CONSORT 2010 Statement: updated guidelines for reporting parallel group randomised trials. BMJ. 2010;340:c332.
[18] Dutta D, et al. GLP-1 receptor agonists and cardiovascular outcomes: a systematic review and meta-analysis of randomized controlled trials. Diabetes Metab Syndr Obes. 2023;16:1047-1064.
