
The landscape of metabolic health treatment has witnessed a remarkable transformation with the introduction of tirzepatide, a groundbreaking medication that represents a significant leap forward in how researchers and clinicians approach Type 2 diabetes and weight management. What Is Tirzepatide (Mounjaro)? This innovative pharmaceutical compound has captured widespread attention for its unique dual-receptor mechanism and impressive clinical outcomes, establishing itself as a subject of intense scientific investigation across multiple therapeutic areas.
Tirzepatide stands apart from conventional treatments through its unprecedented ability to simultaneously activate two critical metabolic pathways, offering researchers valuable insights into complex hormonal regulation systems. As the first approved single-molecule dual agonist of its kind, this peptide has opened new avenues for understanding glucose homeostasis, appetite regulation, and metabolic syndrome management in laboratory settings.
Key Takeaways
- Tirzepatide is a dual-action peptide that simultaneously activates both GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1) receptors, making it the first and only approved medication with this unique mechanism
- Two distinct formulations exist: Mounjaro (approved for Type 2 diabetes management) and Zepbound (approved for chronic weight management and obstructive sleep apnea)
- Multiple metabolic effects include enhanced insulin secretion, reduced glucagon production, delayed gastric emptying, and decreased appetite through central nervous system pathways
- Research-grade tirzepatide is available from specialized suppliers like PEPTIDE PRO for scientific investigation and laboratory applications
- Clinical significance extends beyond glucose control to include substantial weight reduction, improved insulin sensitivity, and potential cardiovascular benefits
Understanding What Is Tirzepatide (Mounjaro): The Science Behind the Dual-Action Mechanism
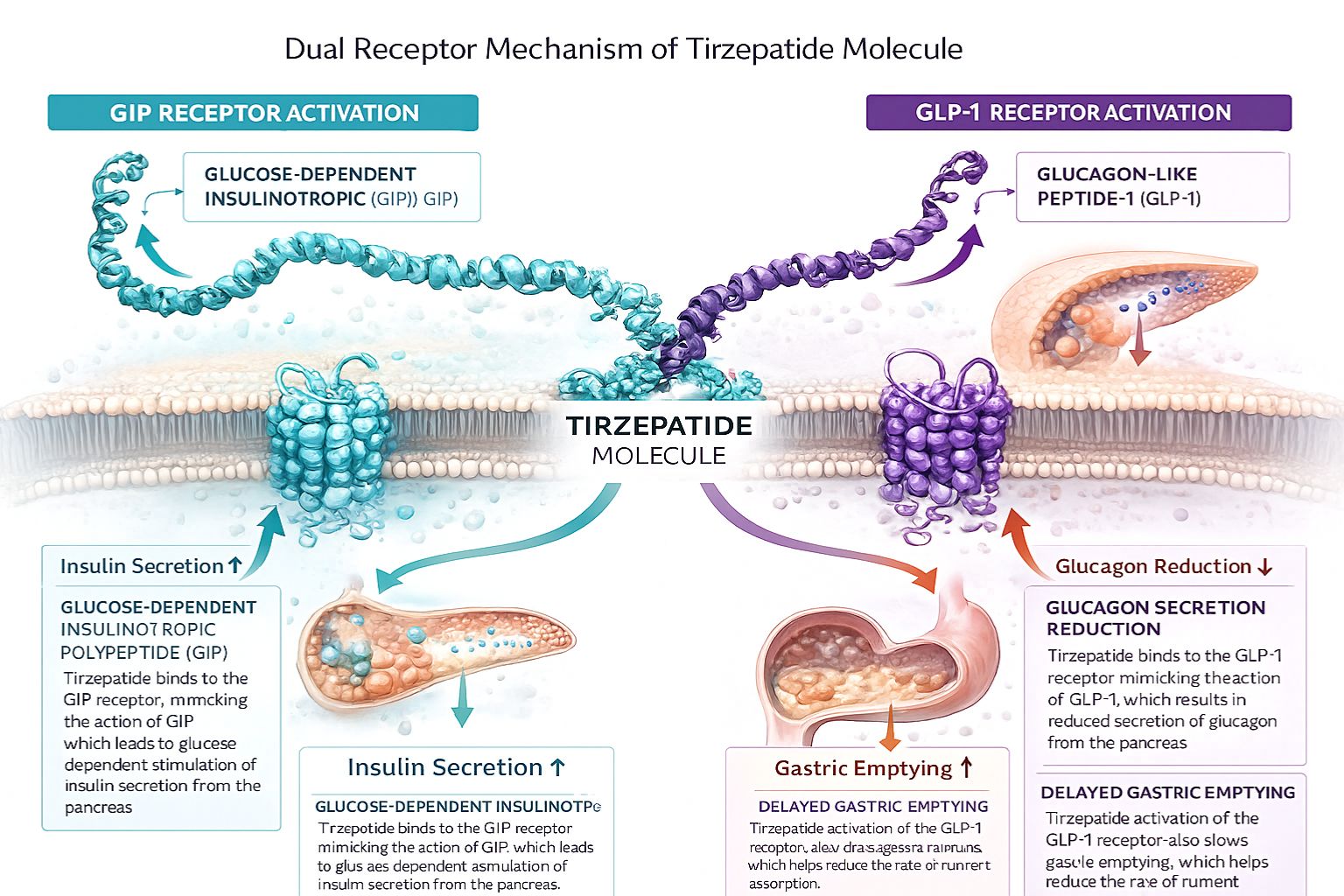
The Molecular Foundation of Tirzepatide
Tirzepatide represents a sophisticated advancement in peptide pharmacology, functioning as a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. This synthetic peptide was engineered to mimic the action of naturally occurring incretin hormones—signaling molecules produced in the gastrointestinal tract that play crucial roles in metabolic regulation.[1]
The molecular structure of tirzepatide consists of 39 amino acids, carefully designed to maintain stability in biological systems while retaining high affinity for both target receptors. This dual-receptor activation strategy distinguishes tirzepatide from earlier single-receptor agonists, providing researchers with a powerful tool for investigating synergistic hormonal pathways.
Unlike traditional GLP-1 receptor agonists such as semaglutide, tirzepatide’s unique architecture allows it to engage both the GIP and GLP-1 receptor systems simultaneously. This dual activation creates a complementary effect that appears to enhance overall metabolic outcomes beyond what either receptor activation achieves independently.
How Tirzepatide Activates GIP and GLP-1 Receptors
The GIP receptor activation component of tirzepatide’s mechanism triggers several important metabolic responses:
- 🔬 Enhanced insulin secretion: GIP receptors on pancreatic beta cells respond to tirzepatide binding by amplifying glucose-dependent insulin release
- 📊 Improved beta-cell function: Sustained GIP receptor activation may support pancreatic beta-cell health and insulin production capacity
- 🧬 Lipid metabolism effects: GIP influences adipose tissue function and may contribute to improved lipid profiles
- ⚡ Energy expenditure modulation: GIP receptor signaling appears to influence metabolic rate and energy utilization
The GLP-1 receptor activation aspect contributes complementary mechanisms:
- 💉 Glucose-dependent insulin secretion: GLP-1 receptors enhance first- and second-phase insulin release in response to elevated blood glucose
️ Glucagon suppression: Tirzepatide reduces inappropriate glucagon secretion, thereby decreasing hepatic glucose production
️ Gastric emptying delay: Activation of GLP-1 receptors slows the rate at which food moves from the stomach to the intestines
- 🧠 Central appetite regulation: GLP-1 receptors in brain regions controlling satiety receive signals that reduce hunger and food intake
This dual-receptor approach creates a synergistic effect that researchers continue to investigate. Studies suggest that GIP receptor activation may enhance the insulinotropic effects of GLP-1 receptor activation while potentially mitigating some of the gastrointestinal side effects associated with GLP-1-only agonists.[2]
The Incretin System and Metabolic Regulation
To fully appreciate what is tirzepatide (Mounjaro) and how it functions, understanding the incretin system proves essential. Incretins are hormones secreted by intestinal cells in response to nutrient intake, particularly glucose and fats. These hormones account for a substantial portion of insulin secretion following meals—a phenomenon known as the “incretin effect.”
In healthy individuals, the incretin effect contributes approximately 50-70% of total insulin secretion after oral glucose intake. However, individuals with Type 2 diabetes often exhibit a diminished incretin effect, with reduced GLP-1 secretion and impaired beta-cell responsiveness to incretin signals.[3]
Key components of the incretin system include:
| Incretin Hormone | Primary Source | Main Functions | Effect Duration |
|---|---|---|---|
| GIP | K-cells (upper small intestine) | Insulin secretion, beta-cell preservation, lipid metabolism | Rapidly degraded (minutes) |
| GLP-1 | L-cells (lower small intestine) | Insulin secretion, glucagon suppression, gastric emptying delay, appetite reduction | Rapidly degraded (minutes) |
| Tirzepatide | Pharmaceutical administration | Dual GIP/GLP-1 receptor activation with extended half-life | Days (weekly dosing) |
The natural incretin hormones GIP and GLP-1 are rapidly degraded by the enzyme dipeptidyl peptidase-4 (DPP-4), limiting their therapeutic utility. Tirzepatide’s molecular design incorporates modifications that confer resistance to DPP-4 degradation and extend its half-life to approximately 5 days, enabling once-weekly administration in clinical settings.[4]
For researchers interested in investigating incretin-based therapies, PEPTIDE PRO offers high-purity research-grade peptides with comprehensive documentation and quality assurance protocols suitable for laboratory applications.
What Is Tirzepatide (Mounjaro) Used For? Clinical Applications and Research Directions
Mounjaro: Type 2 Diabetes Management
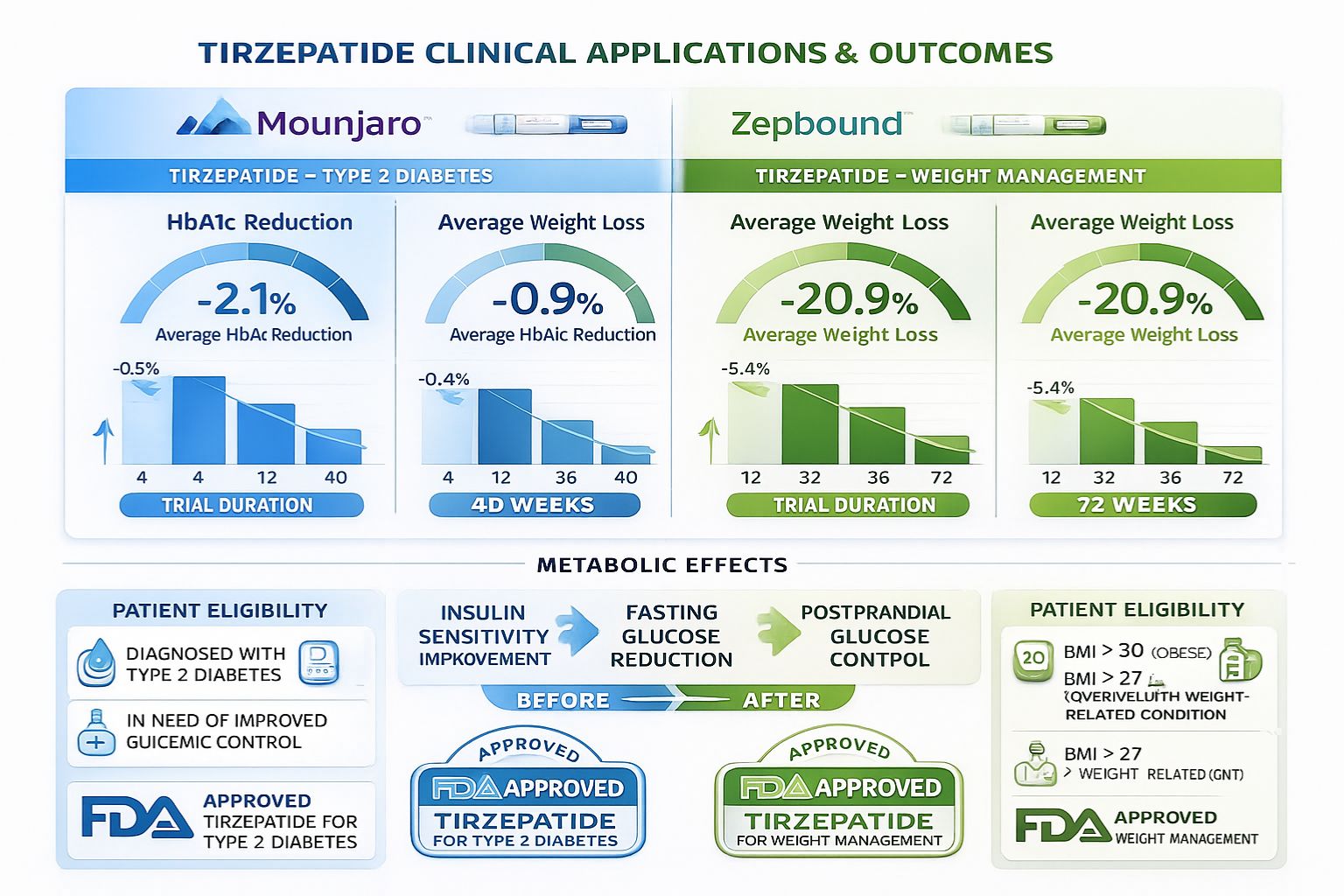
Tirzepatide under the brand name Mounjaro received FDA approval in May 2022 for improving glycemic control in adults with Type 2 diabetes mellitus, as an adjunct to diet and exercise. This approval followed extensive clinical investigation demonstrating superior glucose-lowering efficacy compared to existing treatment options.
Primary mechanisms supporting diabetes management:
- ✅ Enhanced first-phase insulin secretion: Tirzepatide restores the rapid insulin response that typically occurs within minutes of glucose elevation—a response often impaired in Type 2 diabetes
- ✅ Sustained second-phase insulin release: The medication supports prolonged insulin secretion that continues as long as glucose levels remain elevated
- ✅ Glucagon suppression: By reducing inappropriate glucagon secretion, tirzepatide decreases hepatic glucose production, particularly during fasting states
- ✅ Improved insulin sensitivity: Clinical studies utilizing hyperinsulinemic euglycemic clamp methodology—the gold standard for measuring insulin sensitivity—demonstrated significant improvements after 28 weeks of tirzepatide treatment[5]
The SURPASS clinical trial program, which enrolled thousands of participants across multiple studies, established tirzepatide’s efficacy in reducing HbA1c (a measure of average blood glucose over 2-3 months). Participants receiving tirzepatide achieved HbA1c reductions ranging from 1.87% to 2.59%, depending on dosage, with many achieving HbA1c levels below the diagnostic threshold for diabetes.[6]
Comparative effectiveness data from head-to-head trials showed tirzepatide achieving superior glycemic control compared to:
- Semaglutide 1 mg (a GLP-1 receptor agonist)
- Insulin degludec (a long-acting basal insulin)
- Insulin glargine (another basal insulin formulation)
These findings have generated substantial interest in the research community regarding the potential advantages of dual incretin receptor activation over single-receptor approaches.
Zepbound: Weight Management and Metabolic Health
In November 2023, tirzepatide received FDA approval under the brand name Zepbound for chronic weight management in adults with obesity (BMI ≥30 kg/m²) or overweight (BMI ≥27 kg/m²) with at least one weight-related comorbidity, such as hypertension, dyslipidemia, or Type 2 diabetes.
Weight loss mechanisms include:
- Central appetite suppression: Tirzepatide activates receptors in hypothalamic and brainstem regions that regulate hunger, satiety, and food reward
- Gastric emptying delay: Slower stomach emptying creates prolonged feelings of fullness after meals
- Reduced food cravings: The medication appears to diminish “food noise”—persistent thoughts about eating—through modulation of dopamine signaling pathways[7]
- Potential energy expenditure effects: Emerging research suggests possible increases in resting metabolic rate, though this mechanism requires further investigation
The SURMOUNT clinical trial program evaluated tirzepatide specifically for weight management in individuals without diabetes. Results demonstrated average weight reductions of:
- 15.0% of initial body weight with 5 mg weekly dosing
- 19.5% with 10 mg weekly dosing
- 20.9% with 15 mg weekly dosing
These outcomes represent some of the most substantial weight reductions achieved with pharmaceutical interventions, approaching results previously seen primarily with bariatric surgical procedures.[8]
Beyond weight reduction, participants experienced improvements in multiple cardiometabolic risk factors, including blood pressure, lipid profiles, inflammatory markers, and liver fat content—findings that continue to drive research into tirzepatide’s broader metabolic effects.
Emerging Research Applications
The scientific community has identified several additional research directions for tirzepatide beyond its approved indications:
🔬 Cardiovascular outcomes: Ongoing trials are investigating whether tirzepatide reduces major adverse cardiovascular events (heart attack, stroke, cardiovascular death) in high-risk populations
🔬 Non-alcoholic fatty liver disease (NAFLD): Preliminary data suggest significant reductions in liver fat content and improvement in markers of liver inflammation
🔬 Obstructive sleep apnea: In December 2023, the FDA approved Zepbound for moderate-to-severe obstructive sleep apnea in adults with obesity, based on evidence of reduced apnea-hypopnea events
🔬 Polycystic ovary syndrome (PCOS): Researchers are exploring tirzepatide’s potential effects on metabolic and reproductive parameters in PCOS
🔬 Neuroprotection: Preclinical studies suggest GLP-1 and GIP receptor activation may offer neuroprotective benefits, prompting investigation in neurodegenerative conditions
Laboratories conducting peptide research can access research-grade tirzepatide through specialized suppliers that provide comprehensive quality documentation and appropriate handling guidance for investigational applications.
The Metabolic Effects of Tirzepatide: A Deep Dive into Physiological Mechanisms
Glucose Homeostasis and Insulin Dynamics
What is tirzepatide (Mounjaro) doing at the cellular level to achieve its metabolic effects? The answer involves multiple coordinated physiological processes that collectively improve glucose regulation.
Pancreatic beta-cell function enhancement represents a critical mechanism. Tirzepatide binding to GIP and GLP-1 receptors on pancreatic beta cells triggers intracellular signaling cascades that:
- Increase cyclic AMP (cAMP) production, a second messenger that amplifies insulin secretion
- Enhance calcium influx into beta cells, directly triggering insulin granule release
- Activate protein kinase A (PKA) pathways that sensitize the secretory machinery
- Potentially support beta-cell survival and proliferation through anti-apoptotic signaling
These effects are glucose-dependent, meaning they occur primarily when blood glucose levels are elevated. This glucose dependency provides an important safety feature, reducing the risk of hypoglycemia (dangerously low blood sugar) compared to insulin or sulfonylurea medications that stimulate insulin release regardless of glucose levels.
Hepatic glucose production suppression occurs through multiple pathways:
| Mechanism | Effect | Clinical Significance |
|---|---|---|
| Glucagon reduction | Decreased hepatic gluconeogenesis and glycogenolysis | Lower fasting glucose levels |
| Improved insulin sensitivity | Enhanced hepatic insulin signaling | Better suppression of glucose output |
| Direct hepatic effects | Possible direct GIP/GLP-1 receptor effects on liver metabolism | Reduced liver fat accumulation |
Research utilizing continuous glucose monitoring has demonstrated that tirzepatide reduces both fasting glucose (glucose levels before meals) and postprandial glucose (glucose levels after meals). The reduction in postprandial glucose appears particularly robust, with peak glucose excursions substantially blunted compared to baseline measurements.[9]
Gastric Emptying and Satiety Mechanisms
The gastric emptying delay induced by tirzepatide represents a key mechanism contributing to both improved glucose control and weight reduction. GLP-1 receptor activation in the gastric wall and vagal nerve pathways slows the rate at which stomach contents move into the small intestine.
Physiological consequences of delayed gastric emptying:
- 🕐 Prolonged nutrient absorption: Slower delivery of glucose to the bloodstream reduces postprandial glucose spikes
- 🕐 Extended satiety signals: Continued stomach distension maintains activation of stretch receptors that signal fullness
- 🕐 Reduced meal size: Participants in clinical trials consistently report feeling satisfied with smaller food portions
- 🕐 Altered eating patterns: Many individuals naturally reduce eating frequency and eliminate snacking behaviors
Interestingly, the gastric emptying effect appears to undergo some adaptation over time. Initial treatment typically produces more pronounced slowing, which may contribute to early gastrointestinal side effects such as nausea. As treatment continues, partial tachyphylaxis (reduced response) to the gastric emptying effect occurs, though appetite suppression and weight loss continue, suggesting that central nervous system effects become increasingly important for sustained weight management.[10]
Central Nervous System and Appetite Regulation
Perhaps the most fascinating aspect of what is tirzepatide (Mounjaro) involves its effects on brain regions controlling appetite, food reward, and eating behavior. Both GIP and GLP-1 receptors are expressed in multiple brain areas, including:
- Hypothalamus: The primary regulatory center for energy balance, containing neurons that sense nutritional status and coordinate feeding behavior
- Brainstem: Houses the area postrema and nucleus tractus solitarius, which integrate peripheral satiety signals
- Ventral tegmental area (VTA): Part of the brain’s reward circuitry, involved in food motivation and pleasure
- Hippocampus: Contributes to memory formation related to food experiences and may influence eating patterns
Appetite suppression mechanisms include:
- POMC/CART neuron activation: Tirzepatide stimulates pro-opiomelanocortin (POMC) and cocaine- and amphetamine-regulated transcript (CART) neurons in the hypothalamus—these are “satiety neurons” that reduce food intake
- NPY/AgRP neuron inhibition: The medication suppresses neuropeptide Y (NPY) and agouti-related peptide (AgRP) neurons—these are “hunger neurons” that normally drive feeding behavior
- Dopamine pathway modulation: GLP-1 receptor activation in the VTA appears to reduce dopamine release in response to food cues, diminishing the reward value of eating[11]
- Reduced food cravings: Many individuals report decreased preoccupation with food and reduced intensity of cravings, particularly for highly palatable, calorie-dense foods
This multi-level approach to appetite regulation explains why tirzepatide produces sustained weight loss rather than the plateau effect commonly seen with earlier weight management medications. The combination of homeostatic (energy balance) and hedonic (food reward) pathway modulation creates a comprehensive effect on eating behavior.
Researchers investigating appetite regulation and metabolic peptides can find comprehensive resources and high-purity research compounds through specialized suppliers committed to supporting scientific advancement.
Tirzepatide Formulations, Dosing, and Administration Considerations
Pharmaceutical Formulations: Mounjaro vs. Zepbound
While Mounjaro and Zepbound contain the same active pharmaceutical ingredient (tirzepatide), they are marketed under different brand names for distinct therapeutic indications:
Mounjaro (Type 2 Diabetes)
- Approved indication: Improving glycemic control in Type 2 diabetes mellitus
- Available doses: 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, 15 mg
- Prescribing considerations: Often used in combination with other diabetes medications
- Coverage: Typically covered by insurance for diabetes diagnosis
Zepbound (Weight Management)
- Approved indications: Chronic weight management; moderate-to-severe obstructive sleep apnea with obesity
- Available doses: 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, 15 mg
- Prescribing considerations: Requires BMI ≥30 or BMI ≥27 with weight-related comorbidity
- Coverage: Variable insurance coverage; often requires prior authorization
Both formulations utilize a single-dose pen injector designed for subcutaneous administration. The pen device features a hidden needle and automated injection mechanism intended to simplify administration.
Clinical Dosing Protocols
Standard clinical protocols employ gradual dose escalation to optimize tolerability while achieving therapeutic effects. The typical escalation schedule follows this pattern:
| Week | Dose | Purpose |
|---|---|---|
| 1-4 | 2.5 mg once weekly | Initiation dose; allows physiological adaptation |
| 5-8 | 5 mg once weekly | First therapeutic dose for many patients |
| 9-12 | 7.5 mg once weekly | Optional intermediate escalation |
| 13-16 | 10 mg once weekly | Higher therapeutic dose |
| 17-20 | 12.5 mg once weekly | Optional intermediate escalation |
| 21+ | 15 mg once weekly | Maximum approved dose |
The 2.5 mg starting dose serves primarily as an initiation dose to minimize gastrointestinal side effects rather than as a therapeutic dose. Most clinical trial participants required doses of 5 mg or higher to achieve substantial metabolic benefits.
Dose escalation decisions in clinical practice consider:
- ✔️ Tolerability: Gastrointestinal side effects may necessitate slower escalation or remaining at lower doses
- ✔️ Efficacy: HbA1c reduction and weight loss response guide whether dose increases are needed
- ✔️ Individual goals: Target HbA1c levels or weight loss objectives influence optimal dosing
- ✔️ Concomitant medications: Other diabetes medications may be reduced or discontinued as tirzepatide doses increase
Administration Technique and Injection Sites
Tirzepatide is administered via subcutaneous injection, meaning the medication is injected into the fatty tissue layer beneath the skin. Approved injection sites include:
- 💉 Abdomen: Most common site; avoid the area immediately around the navel
- 💉 Thigh: Front and outer portions provide adequate subcutaneous tissue
- 💉 Upper arm: Back of the upper arm (may require assistance for self-administration)
Best practices for administration:
- Site rotation: Alternate injection sites with each dose to prevent lipohypertrophy (fatty lumps) or lipoatrophy (fat loss) at injection sites
- Timing flexibility: Can be administered at any time of day, with or without meals
- Consistency: Administer on the same day each week when possible
- Missed doses: If a dose is missed, administer within 4 days; if more than 4 days have passed, skip the missed dose and resume the regular schedule
- Storage: Unused pens should be refrigerated (2-8°C); may be kept at room temperature for up to 21 days
For research applications, PEPTIDE PRO provides research-grade peptides with detailed reconstitution and handling guidance appropriate for laboratory settings, ensuring optimal stability and experimental consistency.
Safety Profile, Side Effects, and Contraindications of Tirzepatide
Common Gastrointestinal Side Effects
The most frequently reported adverse effects associated with tirzepatide involve the gastrointestinal system, consistent with the medication’s mechanism of action on gastric emptying and GI tract signaling:
Most common side effects (occurring in >5% of participants):
� Nausea: Reported by 12-29% of participants, typically most prominent during dose escalation
� Diarrhea: Affects 12-16% of users, usually transient
� Decreased appetite: While therapeutically desired, can occasionally be excessive
� Vomiting: Occurs in 5-9% of participants, often associated with rapid eating or large meals
� Constipation: Affects 6-7% of users, possibly related to reduced food intake
� Dyspepsia: Indigestion or upper abdominal discomfort in 7-9% of participants
� Abdominal pain: Reported by 6-9% of users
Management strategies for GI side effects:
| Side Effect | Management Approach |
|---|---|
| Nausea | Eat smaller, more frequent meals; avoid high-fat foods; stay hydrated; consider anti-nausea medications if severe |
| Diarrhea | Maintain hydration; consume easily digestible foods; may resolve spontaneously within days to weeks |
| Vomiting | Eat slowly; stop eating when feeling full; avoid lying down immediately after meals |
| Constipation | Increase fiber intake gradually; maintain adequate hydration; consider fiber supplements or stool softeners |
Most gastrointestinal side effects are dose-dependent and time-limited, with the highest incidence occurring during the first few weeks after initiating treatment or increasing doses. Symptoms typically diminish over 4-8 weeks as physiological adaptation occurs.[12]
Serious Adverse Events and Warnings
While generally well-tolerated, tirzepatide carries several important warnings and precautions:
⚠️ Thyroid C-cell tumors: In rodent studies, GLP-1 receptor agonists caused thyroid C-cell tumors at clinically relevant exposures. While human relevance remains uncertain, tirzepatide carries a boxed warning and is contraindicated in patients with:
- Personal or family history of medullary thyroid carcinoma (MTC)
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
⚠️ Pancreatitis: Acute pancreatitis has been reported with GLP-1 receptor agonists. Patients should discontinue tirzepatide and seek medical attention if experiencing severe abdominal pain that may radiate to the back, with or without vomiting.
⚠️ Hypoglycemia: When used as monotherapy, tirzepatide carries low hypoglycemia risk due to its glucose-dependent mechanism. However, when combined with insulin or insulin secretagogues (sulfonylureas), hypoglycemia risk increases. Dose reduction of concomitant medications may be necessary.
⚠️ Acute kidney injury: Cases have been reported, typically in the setting of severe gastrointestinal side effects leading to dehydration. Patients should maintain adequate hydration, particularly during dose escalation.
⚠️ Diabetic retinopathy complications: Rapid improvement in glycemic control has been associated with temporary worsening of diabetic retinopathy. Patients with a history of diabetic retinopathy should be monitored.
⚠️ Gallbladder disease: Increased incidence of cholelithiasis (gallstones) and cholecystitis (gallbladder inflammation) has been observed, likely related to rapid weight loss.
⚠️ Hypersensitivity reactions: Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported. Discontinue tirzepatide if suspected.
Contraindications and Special Populations
Absolute contraindications:
- Personal or family history of medullary thyroid carcinoma
- Multiple Endocrine Neoplasia syndrome type 2
- Known serious hypersensitivity to tirzepatide or any excipients
Special populations requiring careful consideration:
🔬 Pregnancy and lactation: Tirzepatide is not recommended during pregnancy due to potential risks. Animal studies showed fetal harm at exposures below human therapeutic doses. Discontinue at least 2 months before planned pregnancy. Insufficient data exist regarding presence in breast milk.
🔬 Pediatric population: Safety and efficacy have not been established in individuals under 18 years of age.
🔬 Geriatric population: No dose adjustment is required based solely on age, though elderly patients may have increased sensitivity to gastrointestinal effects.
🔬 Renal impairment: No dose adjustment is recommended for mild to moderate renal impairment. Limited data exist for severe renal impairment or end-stage renal disease; use with caution.
🔬 Hepatic impairment: No dose adjustment is required for hepatic impairment based on pharmacokinetic studies.
Researchers working with peptide compounds should consult comprehensive safety data sheets and follow appropriate laboratory safety protocols. PEPTIDE PRO provides detailed safety information and handling guidance with all research-grade materials.
Comparing Tirzepatide to Other Metabolic Peptides and Medications
Tirzepatide vs. GLP-1 Receptor Agonists
Understanding what is tirzepatide (Mounjaro) requires context within the broader landscape of incretin-based therapies. How does this dual agonist compare to established GLP-1 receptor agonists?
Head-to-head comparison: Tirzepatide vs. Semaglutide
| Parameter | Tirzepatide | Semaglutide |
|---|---|---|
| Mechanism | Dual GIP/GLP-1 receptor agonist | GLP-1 receptor agonist only |
| Dosing frequency | Once weekly | Once weekly (injectable); once daily (oral) |
| HbA1c reduction | 1.87-2.59% (dose-dependent) | 1.5-1.8% (dose-dependent) |
| Weight loss | 15-21% of body weight | 10-15% of body weight |
| GI side effects | Nausea: 12-29% | Nausea: 15-44% |
| Cardiovascular outcomes | Under investigation (SURPASS-CVOT) | Proven cardiovascular benefit (SUSTAIN-6) |
Clinical trials directly comparing tirzepatide to semaglutide (SURPASS-2) demonstrated superior glycemic control and weight reduction with tirzepatide across all dose levels tested. The 15 mg tirzepatide dose achieved significantly greater HbA1c reductions (-2.46%) compared to semaglutide 1 mg (-1.86%), with a treatment difference of -0.60%.[13]
Other GLP-1 receptor agonists in context:
- Dulaglutide: Once-weekly GLP-1 agonist; tirzepatide showed superior efficacy in SURPASS-3 and SURPASS-5 trials
- Liraglutide: Daily GLP-1 agonist; generally less weight loss than weekly formulations
- Exenatide: Twice-daily or once-weekly formulations; older generation with more modest efficacy
The enhanced efficacy of tirzepatide compared to GLP-1-only agonists suggests that GIP receptor co-activation provides additive or synergistic metabolic benefits. Proposed mechanisms for this enhanced effect include:
✨ GIP-mediated enhancement of insulin secretion complementing GLP-1 effects ✨ Potential GIP effects on adipose tissue metabolism and energy expenditure
✨ Possible reduction in GLP-1-associated nausea through GIP receptor activation ✨ Synergistic central nervous system effects on appetite regulation
Tirzepatide vs. Other Weight Management Medications
In the weight management landscape, tirzepatide represents one of the most effective pharmaceutical options currently available:
Comparative weight loss efficacy:
| Medication | Mechanism | Average Weight Loss |
|---|---|---|
| Tirzepatide 15 mg | Dual GIP/GLP-1 agonist | 20.9% |
| Semaglutide 2.4 mg | GLP-1 agonist | 14.9% |
| Liraglutide 3.0 mg | GLP-1 agonist | 8.0% |
| Phentermine/topiramate | Sympathomimetic/anticonvulsant | 9.3% |
| Naltrexone/bupropion | Opioid antagonist/antidepressant | 5.0% |
| Orlistat | Lipase inhibitor | 3.0% |
Tirzepatide’s weight loss efficacy approaches that of bariatric surgery procedures, which typically achieve 20-30% weight loss. This has generated substantial interest in whether pharmacotherapy might serve as an alternative or bridge to surgical interventions for some patients.
Triple Agonist Peptides: The Next Frontier
Research continues to advance beyond dual agonists, with triple agonist peptides currently under investigation. These compounds activate GIP, GLP-1, and glucagon receptors simultaneously, adding glucagon’s effects on energy expenditure and hepatic fat metabolism.
Retatrutide, a GIP/GLP-1/glucagon triple agonist, has shown even greater weight loss in early clinical trials—approximately 24% at 48 weeks—though with a potentially different side effect profile.[14] This represents an active area of metabolic peptide research that may yield additional therapeutic options.
Researchers investigating novel metabolic peptides can access a comprehensive range of research-grade compounds through specialized suppliers committed to supporting cutting-edge scientific investigation.
Research Applications and Laboratory Considerations for Tirzepatide
Tirzepatide in Metabolic Research
Beyond clinical applications, what is tirzepatide (Mounjaro) contributing to basic and translational metabolic research? The compound serves as a valuable tool for investigating multiple aspects of metabolic physiology:
🔬 Incretin biology: Tirzepatide enables researchers to dissect the relative contributions of GIP versus GLP-1 receptor activation to various metabolic outcomes
🔬 Beta-cell function: The compound provides insights into mechanisms of glucose-dependent insulin secretion and potential beta-cell preservation strategies
🔬 Energy homeostasis: Studies utilizing tirzepatide help elucidate the complex neural circuits regulating appetite, satiety, and energy expenditure
🔬 Metabolic disease models: Animal models treated with tirzepatide contribute to understanding obesity, diabetes, and metabolic syndrome pathophysiology
🔬 Cardiovascular-metabolic interactions: Research explores connections between metabolic improvements and cardiovascular outcomes
🔬 Hepatic steatosis: Investigations examine mechanisms by which dual incretin activation reduces liver fat accumulation
Quality Considerations for Research-Grade Tirzepatide
Laboratories conducting peptide research require high-purity compounds with consistent quality to ensure reproducible, reliable results. Key quality parameters include:
Purity specifications:
- ≥98% purity by HPLC (high-performance liquid chromatography)
- Minimal presence of related impurities or degradation products
- Appropriate peptide content verification by mass spectrometry
Handling and storage requirements:
- Lyophilized (freeze-dried) form for optimal stability
- Storage at -20°C or below in original sealed containers
- Protection from light and moisture
- Desiccant inclusion to prevent moisture absorption
Documentation standards:
- Certificate of Analysis (COA) with each batch
- HPLC chromatogram demonstrating purity
- Mass spectrometry confirmation of molecular weight
- Recommended reconstitution protocols
- Stability data under specified storage conditions
PEPTIDE PRO specializes in providing research-grade peptides that meet rigorous quality standards, with comprehensive documentation and expert technical support to ensure optimal experimental outcomes.
Reconstitution and Experimental Preparation
For laboratory applications, lyophilized tirzepatide requires reconstitution prior to use. Standard protocols include:
Reconstitution procedure:
- Solvent selection: Sterile water, phosphate-buffered saline (PBS), or bacteriostatic water depending on application
- Concentration calculation: Determine desired working concentration based on experimental design
- Gentle mixing: Add solvent slowly down the pen peptide wall; swirl gently rather than vortexing to prevent peptide aggregation
- Complete dissolution: Allow adequate time for complete reconstitution; may require 5-10 minutes
- Storage of working solution: Refrigerate at 2-8°C for short-term use (days to weeks) or aliquot and freeze at -20°C or below for longer storage
Stability considerations:
- Reconstituted peptide solutions are generally less stable than lyophilized powder
- Freeze-thaw cycles should be minimized to prevent degradation
- Aliquoting into single-use portions prevents repeated freeze-thaw
- Addition of carrier proteins (e.g., 0.1% BSA) may enhance stability in dilute solutions
Experimental dosing:
Research applications require careful dose calculation based on:
- Subject species and body weight
- Route of administration (subcutaneous, intraperitoneal, intravenous)
- Desired receptor occupancy or physiological effect
- Pharmacokinetic parameters specific to the experimental model
Researchers can access detailed reconstitution guidance and technical resources through specialized peptide suppliers committed to supporting high-quality scientific investigation.
The Future of Tirzepatide Research and Clinical Development
Ongoing Clinical Trials and Investigations
The tirzepatide research landscape continues to expand, with numerous ongoing investigations exploring additional therapeutic applications:
SURPASS-CVOT (Cardiovascular Outcomes Trial)
- Evaluating whether tirzepatide reduces major adverse cardiovascular events (MACE)
- Enrolling >12,000 participants with Type 2 diabetes and established cardiovascular disease
- Expected completion: 2025
- Potential impact: If positive, would establish tirzepatide as a cardioprotective therapy similar to some GLP-1 agonists
SURMOUNT-OSA
- Investigating tirzepatide for obstructive sleep apnea in adults with obesity
- Completed with positive results leading to FDA approval in December 2023
- Demonstrated significant reductions in apnea-hypopnea index (AHI)
SURMOUNT-MMO (Metabolic Dysfunction-Associated Steatohepatitis)
- Examining tirzepatide’s effects on liver histology in MASH (formerly NASH)
- Assessing liver fat reduction, inflammation resolution, and fibrosis improvement
- Could expand tirzepatide’s indications to include liver disease
Additional research directions:
- Combination therapies with other metabolic agents
- Pediatric obesity and Type 2 diabetes applications
- Polycystic ovary syndrome (PCOS) metabolic effects
- Cognitive function and neurodegenerative disease prevention
- Heart failure with preserved ejection fraction (HFpEF)
Potential Expanded Indications
Based on emerging evidence, what is tirzepatide (Mounjaro) may become in the future could extend well beyond current approved uses:
� Metabolic dysfunction-associated steatotic liver disease (MASLD): Substantial liver fat reductions observed in clinical trials suggest potential therapeutic benefit
� Cardiovascular disease prevention: If SURPASS-CVOT demonstrates cardiovascular benefit, tirzepatide could be indicated for cardiovascular risk reduction
� Chronic kidney disease: GLP-1 agonists have shown renal protective effects; tirzepatide’s dual mechanism may offer similar or enhanced benefits
� Heart failure: Metabolic improvements and potential direct cardiac effects warrant investigation in heart failure populations
� Alzheimer’s disease and cognitive decline: Preclinical evidence suggests GLP-1 and GIP receptor activation may offer neuroprotective effects
Challenges and Considerations
Despite promising developments, several challenges remain:
⚡ Cost and accessibility: High medication costs limit access for many patients; biosimilar development may eventually improve affordability
⚡ Long-term safety: While clinical trial data extend to 2+ years, longer-term safety data (5-10+ years) will be important as use expands
⚡ Treatment duration: Optimal treatment duration remains unclear; weight regain typically occurs upon discontinuation, suggesting chronic therapy may be necessary
⚡ Individual variability: Response varies substantially between individuals; predictive biomarkers would help identify optimal candidates
⚡ Supply constraints: High demand has periodically created supply limitations, affecting patient access
⚡ Insurance coverage: Variable coverage policies, particularly for weight management indications, create access barriers
The research community continues to address these challenges through ongoing investigation, health economics research, and development of next-generation compounds with potentially improved profiles.
Practical Guidance for Researchers Working with Tirzepatide
Sourcing High-Quality Research Peptides
For laboratories conducting tirzepatide research, peptide quality directly impacts experimental validity and reproducibility. Key considerations when sourcing research-grade tirzepatide include:
Supplier evaluation criteria:
✅ Quality documentation: Comprehensive Certificates of Analysis (COA) with each batch, including HPLC purity analysis and mass spectrometry confirmation
✅ Regulatory compliance: Suppliers should clearly label products “For Research Use Only” and maintain appropriate documentation
✅ Storage and handling: Proper storage conditions maintained throughout supply chain, with temperature-controlled shipping options
✅ Technical support: Access to knowledgeable staff who can provide guidance on reconstitution, storage, and handling
✅ Consistency: Batch-to-batch consistency in purity and peptide content for reproducible experimental results
✅ Transparency: Clear product information including sequence, molecular weight, storage recommendations, and any modifications
PEPTIDE PRO exemplifies these quality standards, providing research-grade peptides with comprehensive documentation, rigorous quality control, and expert technical support to ensure optimal experimental outcomes.
Experimental Design Considerations
Researchers designing studies involving tirzepatide should consider:
Dose selection:
- Review published literature for species-specific dosing
- Consider pharmacokinetic differences between species
- Include multiple dose levels to establish dose-response relationships
- Account for differences between acute and chronic administration
Route of administration:
- Subcutaneous administration most closely mimics clinical use
- Intraperitoneal administration may be acceptable for some rodent studies
- Route affects pharmacokinetics and should be consistent within studies
Treatment duration:
- Acute studies (single dose): Useful for mechanistic investigations
- Short-term studies (days to weeks): Examine initial metabolic responses
- Chronic studies (weeks to months): Required for weight loss and sustained metabolic effects
Control groups:
- Vehicle-treated controls (same injection volume and frequency)
- Positive controls (established comparator compounds)
- Pair-fed controls (for weight loss studies to distinguish direct effects from caloric restriction effects)
Outcome measures:
- Glucose homeostasis: Fasting glucose, glucose tolerance tests, insulin levels, HbA1c (in appropriate species)
- Body composition: Body weight, fat mass, lean mass (via imaging or carcass analysis)
- Food intake: Daily food consumption, meal patterns, feeding behavior
- Energy expenditure: Indirect calorimetry, activity monitoring
- Tissue-specific effects: Pancreatic histology, liver fat content, adipose tissue characteristics
Regulatory and Ethical Considerations
Research involving tirzepatide must adhere to appropriate regulatory and ethical frameworks:
🔒 Institutional oversight: Studies involving animals require IACUC (Institutional Animal Care and Use Committee) approval with detailed protocols
🔒 Research use only: Tirzepatide for research applications must be clearly labeled and documented as “For Research Use Only—Not for Human or Animal Consumption”
🔒 Proper documentation: Maintain detailed records of peptide sourcing, storage conditions, reconstitution procedures, and administration protocols
🔒 Safety protocols: Follow appropriate laboratory safety procedures when handling peptide compounds, including PPE use and proper waste disposal
🔒 Data integrity: Maintain rigorous experimental records, including batch numbers, reconstitution dates, and storage conditions that might affect peptide stability
Researchers can access comprehensive regulatory guidance and quality documentation through specialized peptide suppliers committed to supporting compliant, high-quality research.
Conclusion: The Significance of Tirzepatide in Metabolic Science and Medicine
What is tirzepatide (Mounjaro)? As this comprehensive examination reveals, tirzepatide represents far more than simply another diabetes or weight loss medication. This innovative dual GIP/GLP-1 receptor agonist embodies a paradigm shift in how researchers and clinicians approach metabolic disease management—moving from single-target interventions to sophisticated multi-pathway modulation that more closely mimics the body’s natural regulatory systems.
Key insights from this exploration:
� Unprecedented mechanism: Tirzepatide’s dual-receptor activation strategy creates synergistic metabolic effects that exceed those achieved by single-receptor agonists, establishing a new benchmark for incretin-based therapies
� Comprehensive metabolic impact: Beyond glucose lowering and weight reduction, tirzepatide influences multiple interconnected systems—pancreatic function, hepatic metabolism, gastric motility, central appetite regulation, and potentially cardiovascular health
� Clinical transformation: The substantial efficacy demonstrated in clinical trials—with HbA1c reductions exceeding 2% and weight loss approaching 21%—represents outcomes previously achievable primarily through surgical interventions
� Research applications: Tirzepatide serves as a valuable investigational tool for dissecting incretin biology, energy homeostasis, and metabolic disease mechanisms, driving scientific understanding forward
� Evolving landscape: Ongoing clinical trials continue to expand potential therapeutic applications, while next-generation compounds build upon tirzepatide’s foundation to explore triple-agonist and other novel approaches
Actionable Next Steps
For researchers and laboratory professionals:
- Evaluate experimental applications: Consider how tirzepatide might serve as a tool in your metabolic research, whether investigating incretin biology, appetite regulation, or metabolic disease models
- Source high-quality peptides: Partner with reputable suppliers like PEPTIDE PRO that provide research-grade compounds with comprehensive quality documentation and technical support
- Design rigorous protocols: Incorporate appropriate controls, dose-response relationships, and outcome measures to generate reproducible, publication-quality data
- Stay current with literature: Monitor ongoing clinical trials and emerging research to inform experimental design and identify knowledge gaps requiring investigation
- Consider collaborative opportunities: The complexity of metabolic regulation often benefits from multidisciplinary approaches combining expertise in endocrinology, neuroscience, pharmacology, and other fields
For those seeking to understand tirzepatide’s role in metabolic health:
- Recognize the multifaceted mechanism: Tirzepatide’s effects extend well beyond simple appetite suppression or insulin stimulation, involving coordinated action across multiple organ systems
- Appreciate individual variability: Response to tirzepatide varies substantially between individuals, influenced by genetic factors, baseline metabolic status, lifestyle factors, and other variables
- Understand the research context: While approved for specific clinical indications, tirzepatide continues to be investigated for numerous additional applications that may expand its therapeutic utility
- Monitor emerging evidence: The tirzepatide research landscape evolves rapidly, with new findings regularly published regarding mechanisms, applications, and long-term outcomes
The Broader Impact
Tirzepatide exemplifies how sophisticated understanding of physiological regulatory systems can translate into transformative therapeutic interventions. The compound’s success has validated the dual-agonist approach, inspiring development of triple-agonist and other multi-target peptides that may offer even greater metabolic benefits.
As obesity, Type 2 diabetes, and related metabolic conditions continue to affect hundreds of millions globally, innovations like tirzepatide provide hope for more effective management strategies. The medication’s ability to produce substantial, sustained metabolic improvements challenges previous assumptions about the limits of pharmaceutical intervention in metabolic disease.
For the research community, tirzepatide represents both a powerful investigational tool and proof-of-concept that sophisticated multi-pathway modulation can achieve outcomes previously considered unattainable with single-target drugs. This success will likely influence drug development strategies across multiple therapeutic areas, extending well beyond metabolic disease.
Final Thoughts
Understanding what is tirzepatide (Mounjaro) requires appreciating both its immediate clinical applications and its broader significance for metabolic science. This dual GIP/GLP-1 receptor agonist has fundamentally altered the landscape of diabetes and obesity management while simultaneously advancing scientific understanding of incretin biology and metabolic regulation.
As research continues to unveil additional mechanisms, applications, and long-term outcomes, tirzepatide’s full impact will become increasingly apparent. Whether serving clinical needs or research objectives, this innovative peptide exemplifies the potential of translating sophisticated physiological understanding into meaningful therapeutic advancement.
For researchers seeking high-purity tirzepatide and other research-grade peptides, PEPTIDE PRO provides the quality, documentation, and support necessary for rigorous scientific investigation—delivered with the professionalism and reliability that cutting-edge research demands.
References
[1] Nauck MA, Quast DR, Wefers J, Meier JJ. GLP-1 receptor agonists in the treatment of type 2 diabetes – state-of-the-art. Mol Metab. 2021;46:101102.
[2] Frias JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N Engl J Med. 2021;385(6):503-515.
[3] Nauck MA, Meier JJ. The incretin effect in healthy individuals and those with type 2 diabetes: physiology, pathophysiology, and response to therapeutic interventions. Lancet Diabetes Endocrinol. 2016;4(6):525-536.
[4] Coskun T, Sloop KW, Loghin C, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept. Mol Metab. 2018;18:3-14.
[5] Heise T, Mari A, DeVries JH, et al. Effects of subcutaneous tirzepatide versus placebo or semaglutide on pancreatic islet function and insulin sensitivity in adults with type 2 diabetes: a multicentre, randomised, double-blind, parallel-arm, phase 1 clinical trial. Lancet Diabetes Endocrinol. 2022;10(6):418-429.
[6] Rosenstock J, Wysham C, Frías JP, et al. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet. 2021;398(10295):143-155.
[7] Borner T, Workinger JL, Tinsley IC, et al. GIP receptor agonism attenuates GLP-1 receptor agonist-induced nausea and emesis in preclinical models. Diabetes. 2021;70(11):2545-2553.
[8] Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387(3):205-216.
[9] Thomas MK, Nikooienejad A, Bray R, et al. Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes. J Clin Endocrinol Metab. 2021;106(2):388-396.
[10] Urva S, Coskun T, Loghin C, et al. The Novel Dual Glucose-Dependent Insulinotropic Polypeptide and Glucagon-Like Peptide-1 (GLP-1) Receptor Agonist Tirzepatide Transiently Delays Gastric Emptying Similarly to Selective Long-Acting GLP-1 Receptor Agonists. Diabetes Care. 2020;43(8):1661-1664.
[11] Adriaenssens AE, Biggs EK, Darwish T, et al. Glucose-Dependent Insulinotropic Polypeptide Receptor-Expressing Cells in the Hypothalamus Regulate Food Intake. Cell Metab. 2019;30(5):987-996.
[12] Dahl D, Onishi Y, Norwood P, et al. Effect of Subcutaneous Tirzepatide vs Placebo Added to Titrated Insulin Glargine on Glycemic Control in Patients With Type 2 Diabetes: The SURPASS-5 Randomized Clinical Trial. JAMA. 2022;327(6):534-545.
[13] Frías JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N Engl J Med. 2021;385(6):503-515.
[14] Jastreboff AM, Kaplan LM, Frías JP, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity – A Phase 2 Trial. N Engl J Med. 2023;389(6):514-526.
